Research Article
Regeneration of Spend Activated Carbon
Department of Chemistry, University of Agriculture, Faisalabad, Pakistan
Haq Nawaz
Department of Chemistry, University of Agriculture, Faisalabad, Pakistan
Activated carbon is an amorphous form of carbon with large internal surfaces and pores. On account of this adsorption, activated carbon is extensively used in the purification of water, sewerage treatments, dry cleaning, refrigeration, food and beverage industries, pharmaceutical industries etc. (Foster and Leslie, 1980).
During removal and adsorption of various impurities, active carbon loses its sites which are responsible for adsorption. The spent carbon may be regenerated by off-site and on-site regeneration (Jahangir et al., 1995). Over a long period of time, a number of regeneration processes have been tested, evaluated and applied. These include the thermal, chemical and biological process (Clark and Dorsey, 1987).
Spent carbon is a valuable renewable resource. Under strict quality controls that parallel those used to originally active carbon, spent carbon can be reactivated to its original activity (±10%). This capability has been perfected to the levels of quality and safety associated with "Food grade" and drinking water standards (Anonymous, 1985).
Keeping in view the significance of the utilization of spent carbon, the present project is undertaken. The purpose of present study is to investigate the extent of chemical regeneration on granular activated carbon.
The sample of spent activated carbon was procured from CPC-maize product. The moisture contents of the sample were determined by drying it in an oven at 105°C for an hour. The percentage purity was estimated by determining its adsorptive capacity using methylene blue and iodine as standard solution (Foster and Hilton, 1980). The dried spent activated carbon was washed with water and treated with different concentrations (0.5 N 0.8 N) with HCI and H3PO4. The treated samples of spent carbon were dried in an electric oven for half and hour. After drying the samples, they were subjected to chemical reactivation in the absence of an air in an electric furnace (7000-1000°C) for an hour.
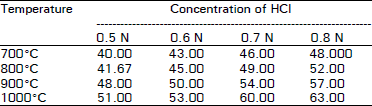
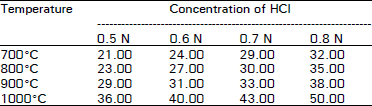
The results pertaining to the effect of concentrations of HCI and H3PO4 and temperature on the adsorption capacity of regenerated spent activated carbon are shown in Table 1-4 respectively. The results revealed that the adsorption capacity of regenerated spent activated carbon for both iodine and methylene blue solution increased with increasing acid concentration. The minimum adsorption capacity was recorded for both methylene blue and iodine with 0.5 N acid concentration. While the maximum adsorption capacities for both the solutions were observed with 0.8 N acid concentration. O’Grady and Wennerberg (1986), Beg and Usmani (1988) and Chughtai et al. (1992) reported an increase in the adsorption capacity of an activated carbon with the increase in the concentration of activated carbon.
| Table 1: | Adsorption capacity expressed as mg of methylene blue adsorbed by 1 regenerated spent activated carbon with hydrochloric acid |
 | |
| Table 2: | Adsorption capacity expressed as mg of methylene blue adsorbed by 1 regenerated spent activated carbon with phosphoric acid |
 | |
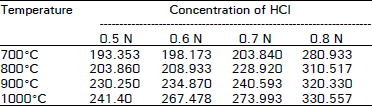
| Table 3: | Adsorption capacity expressed as mg of methylene blue adsorbed by 1 regenerated spent activated carbon with HCI |
 | |
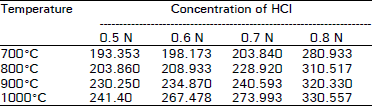
| Table 4: | Adsorption capacity expressed as mg of iodine adsorbed by 1 regenerated spent activated carbon with HCI |
 | |
Moreover it was found that the regenerated spent carbon treated with HCI as more adsorption capacity then H3PO4.
The data regarding the effect of different temperatures on the adsorption capacity of regenerated spent activated carbon revealed that the adsorption capacity increase with increasing temperature. Minimum adsorption capacity was observed at 7000°C. While the maximum adsorption capacity was observed at 1000°C.
The results are in close agreement with the findings of Khan et al. (1985), Beg and Usmani (1985), Chughtai et al. (1987) and Cannon et al. (1993) who reported an increase in the adsorption capacity of activated carbon with an increase in temperature. Moreover, the spent carbon treated with HCI showed more adsorption capacity at 1000°C than that treated with H3PO4 at the same temperature.