Research Article
The Antinociceptive Effects of Extracts from Stichopus chloronotus Brandt
Not Available
M.Z. Zarina
Not Available
Kaswandi M.A.
Not Available
M. Nadirah
Not Available
A.F. Shamsuddin
Not Available
Intoduction
Sea cucumbers are locally known as "bat" or "balat" among the ethnics of Sabah. In the Peninsular of Malaysia, they are referred to as "gamat", a name given to one particular species which is widely used in traditional medicine. However, other species appear to have similar medicinal properties (Ridzwan et al., 1995). Like haruan, Channa stratus (Mat Jais et al.,1997), sea cucumbers promote wound healing (Ridzwan et al.,1990) whereas their extracts being antibacteria (Ridzwan et al., 1995) and contain antioxidant properties (Hawa et al.,1999) and fatty acids (Fredalina et al., 1999). Relief from pain and irritation is one of the reported benefits in those taking sea cucumbers based remedies for the treatment of skin conditions such as eczema and arthritis. However, despite the widespread use of preparations from sea cucumbers for medicinal purposes, there have been hardly any study to establish the scientific basis for its claimed benefits, especially, the analgestic tendency of sea cucumbers, hence the present study. Four preparations of sea cucumber, Stichopus chloronotus; water, phosphate buffer solution (PBS), ethanol and methanol, were investigated for their antinociceptive effects in mice with a view to establishing whether or not the reported pain-relieving activities of sea cucumber have a scientific basis.
Animals: Male albino mice averaging 30 g b.w. were used in accordance with the Institute Medical Research (IMR), Malaysia guidelines on animal experimentation. The animals were obtained from IMR and kept in the animal house for at least 72 h before used. Food and water were supplied ad libitum. Each mice was used only once. All experiments were performed during the period between 9:30 h and 12:00 h.
Preparation of the extracts: The sea cucumbers Stichopus chloronotus Brandt were collected from the coastal areas of Peninsular Malaysia. Identification of species was based on Ridzwan (1993). The samples were kept in labeled plastic bags before they were stored in a freezer at -80°C.
Extraction of sea cucumbers: The animals were thawed from deep freezer, thoroughly washed with distilled water, cut up longitudinally along the bodyline to remove the visceral organs. The remaining body walls were dried on blotting papers before being homogenized to a fine texture. The extracts were prepared according to Shimada (1961) in which 50 g of the homogenized tissues for each extract was placed in a 250 ml conical flask to which 100 ml of each of the following solvents; distilled water, PBS (pH 7.2), methanol (BDH 99.8%) and ethanol (BP 95%) were added. Each of the flask containing the blended tissues in their corresponding solvents was shaken in a water-bath shaker at 80 rev/min at room temperature for 72 h in methanol and ethanol,fand 4 h in PBS and distilled water. The resulting mixtures were centrifuged at 3,000 rpm for 20 min. The supernatants were then collected. For PBS and distilled water extraction, the samples were freeze-dried using a freeze-dryer (Model Heto FD3, ID 87154), and for methanol and ethanol extraction, the extracts undergo drying process using a rotary vacuum evaporator (Eyela SB-35 Tokyo Rikakikae) until all the samples dried. The powder form of each extract was then stored in labelled sterile bottles at 4 °C before being assayed for the present of antinociceptive agents conducted on mice.
Preparation of samples: For antinociceptive determination test, 100 mg of PBS and distilled water extracts were solubilized in 1 ml sterile PBS (pH 7.26) and sterile distilled water, respectively. The ethanolic and methanolic extracts were solubilized in dimethylsulphoxide (DMSO, Merck). To speed up the solubilisation process, the extracts were sonicated in the water for 30 min at room temperature using a sonicator (Model Bronson 2200).
Drugs: Morphine sulphate (Delta West, USA) was dissolved in a physiological saline solution to such concentrations that requisite doses were administered in volume of 10 ml/kg. The dose of morphine used in each test was adopted from ED50 values previously established in our lab setting, being 0.8 mg/kg for the abdominal constriction test and 1.5 mg/kg for the tail-flick test (Dambisya and Lee, 1994, 1995). The sea cucumber extracts and morphine were administered 10 min after the extract, at a different site. In all experiments, saline was used as the control.
Antinociceptive assays: The acetic acid abdominal constriction test was used with local adaptations as described by Dambisya and Lee (1995). Acetic acid (0.6%) was administered intraperitoneally (i.p.) in a volume of 10 ml/kg with the observations and other modifications of this test for our lab setting as described elsewhere (Dambisya and Lee, 1995). Low concentrations of the respective extracts were used in the morphine-sea cucumber extract interaction study. The acetic acid was administered (i.p.) 25-30 min after the sea cucumber extract (s.c.) or 30 min after morphine, being times we established to be the period for peak antinociceptive effects. Analgesia was calculated as the percentage inhibition of the abdominal constrictions.
The hot tail-flick test (D'Amour and Smith, 1941) was used with modification as described by Dambisya and Lee (1995). The screening cut-off time was 5s, while the test cut-off time was 10s. A single concentration level of holothuroid extract was used in this assay to gauge the effects of the extracts on tail-flick latency while lower concentration levels were used in the morphine-sea cucumber extracts interaction study as for the abdominal constriction assay above.
Statistical analysis: The results were presented as mean ± S.E.M. The Student's t-test was used to analyze and compare the data with p < 0.05 as the limit of significance.
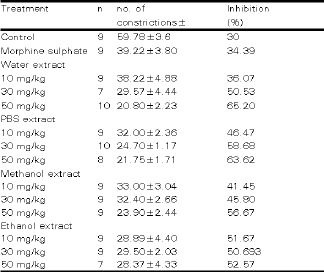
Various concentrations (0, 10, 30 and 50 mg/kg) of the 30 min different extracts of sea cucumber were studied in order to established the concentration-response profile. The results are shown in Table 1. There was a demonstrable dose response (p < 0.05) pattern between 10 mg/kg using water extract with the 50 mg/kg giving 65.20% inhibition. Treatment using water extract exhibited an increased in percentage of inhibition (36.07-65.20%) as the dose was increased.
Abdominal constriction response: There was an increment in the percentage of inhibition of abdominal constriction response for treatment using water extract at dose 10, 30, 50 mg/kg from 36.07, 50.53 and 65.20%. Comparison with normal saline as negative control showed a significant increased (p < 0.05) in percentage of inhibition. Nevertheless, comparison study with positive control (morphine sulphate 34.39%) did not produce a significant difference at dose 10 mg/kg and 30mg/kg.
Treatment using PBS extract also showed an increase in percentage of inhibition from 46.47 % at a dose 10 mg/kg to 63.62 % at a dose 50mg/kg. This increment is significant (p < 0.05) in comparison with treatment using a normal saline whereas for treatment using morphine sulphate, this increment was only significant (p < 0.05) at a dose 50 mg/kg of PBS extract.
Hot tail-flick: Treatment using saline and morphine showed a remarkable antinociceptive activities (Table 2). When extracts of PBS and ethanol were used the effects were not so satisfactory. On the other hand, treatment using water extract produced an obvious effect only after 30 min after administration. The first sign of positive effect was observed 15 min after the mice was treated followed by a fall in the activity at 45 min (Table 2).
| Table 1: | The effects of Stichopus chloronotus extracts administered intraperitoneally on the abdominal constriction responses in mice. |
 | |
Discussion
The results showed that all the extracts of Stichopus chloronotus have dose-dependent antinociceptive effects as assessed by the abdominal constriction test. However, there was no demonstrable effects in the tail-flick test except on the water extract.
| Table 2: | Effects of various extracts (30 mg/kg i.p.) of Stichopus chloronotus on tail-flick response using hot plate. Normal saline and morphine sulphate being negative and positive control, respectively. The number in bracket indicate percentage of inhibition. |
 | |
The abdominal constriction test is very sensitive and can detect antinociceptive effects of compounds/dose levels that maybe inactive in the tail-flick assay (Collier et al., 1968). The abdominal constriction response is thought to involve, in part, local peritoneal receptors (Bentley et al., 1983) while the tail-flick response is essentially a spinal reflex. These differences in sensitivity of the two tests and the mechanisms involved may explain the apparent lack of effect of these effects in the tail-flick test. Nevertheless, the finding of antinociceptive action in one test and the indication using water extract for the tail-flick test, seem to support the common observation among the local as well as the ethnics in Malaysia that utilizing sea cucumber offers pain relief, especially, in arthritis and burning.
The research was funded by the Ministry of Science and Technology Malaysia through grant IRPA RM 09-02-02-021.