Research Article
Histological Assessment of Ghost Crab`s Ovary (Ocypode saratan) in Chabahar Bay
Faculty of Fisheries, Islamic Azad University, Science and Research Branch, P.O. Box 1415/755, Tehran, Iran
In this research, the histology of Flat crab`s ovary (Ocypode saratan) is assessed in Chabahar Bay of Oman Sea during the period of December 2005 to December 2006. Ten ovaries of such species were sampled and fixed in the bouin solution. The samples were undergone the histology, preparation, dehydration, clearing and paraffination according to standard procedure. Then sections of 5 μ were prepared and after coloration, they were studied under an electronic microscope. Gonad maturity stages of such species were determined in 6 microscopic levels. In first and second stages, the crab is immature and third to sixth stages, the crab is mature. Oocyte diameter sizes in ovary through ovulation process are: Immature (<20 μm), rudimentary (20-40 μm), developing (40-70 μm), intermediate (70-90 μm), advanced (90-160 μm) and resting (>160 μm). Maximum dimension of oocyte frequency is 152.3±0.5 micron. Salinity has complementary effect on temperature and increase in salinity results in complete maturity of gonads. Female sex has almost during May and September. The peak has been registered in May 4.12.
INTRODUCTION
Ghost crab has always been taken into consideration for various aspects such as environmental pollution index, nutritional value, scavenger, remedial traits and tourist attraction (Burggren and McMahon, 1988). It is also written in the list of preserved animals. However, in some countries (such as Greece), extreme increase in its population has caused a serious environmental problem because of its harmful consequences (Thanos, 1999). Over exploitation of resources especially in African countries has threatened such organisms. Therefore, if using the timing for fishing season is considered as a tool to preserve the resources and responsible exploitation of such species, achieving more information and findings are required about their reproduction biology and physiology. In most cases, histological researches account a great deal for achieving information about the above-mentioned matters (Poosti, 2004). In this study, it`s been tried to assess how ovulation and its reproduction pattern function through showing the ovary tissue sections of the Ghost crab. Such species has been identified on Iran`s southern coastline (Bahmani, 1994; Saeedpoor, 1994). Paying attention to being a new-born science, the ovary histology of flat crabs in Iran has not been well-studied yet. A thus perfect histological study has been done in addition to external indices relevant to the reproduction of such crab. Considering the results, such studies can cause some methodologies in controlling and optimal management measures on the coastline.
MATERIALS AND METHODS
Three times a month, the temperature was measured and averaged out by a thermometer with 0.1°C accuracy as well as water salinity with AgNO3 titration. Monthly 10 samples were collected at dusk (because this species is nocturnal) most often with butterfly-catching net from 6000 m2 of Chabahar bay coast (with northern 60° 31` longitude and 25° 26` latitude) from December 2005 to December 2006 and then, female sexes were separated after measuring the total weight and ovary weight with digital scale with 0.01 g accuracy. In order to provide the tissue sections, first the ovary was fixed in Bouin solution, then cleared, dehydrated and paraffinated according to Poostis method Poosti (2004). Five micron sections were prepared with microtome, colorated with Hematoxylin and Eosin method and assesses by a light microscope. The identifying key is the ovary development considering its appearance such as color, weight, occupied space and the ovum (Mouton and Felder, 1995; Goodman, 1964). To calculate the oocytes` diameters, first they should be separated from the abdomen (in order to separated the fiberous tissues with Gilson`s solution, they are kept in dark dishes for one month and shaken slowly every week) and then after preparation, 5 oocyte masses are harvested and their diameters are determined with micrometer under the microscope. The real diameters of the oocytes are measured using the coefficients related to microscope and are used in the result after averaging out. Moreover, in order to assess the maturity as well as other factors, the maturity index is determined with this formula which is an indirect method in estimating the species spawning season:
Gonad index GSI = (Gonad weight/bode weight)x100 |
To assess statistically, (ANOVA) SPSS 13 program and Excel program are be used.
RESULTS
Figure 1 and 2 represent the fluctuations of environmental factor such as temperature and salinity during 12 month. The temperature peak in the inhabitant of Ocypode saratan was 31.3°C in Chabahar Bay during June. In addition, the salinity peak in Tune was 40.12 ppm. The result achieved from the assessment of Ghost crab ovary tissue show 6 gonad stages.
Immature stage: Consists of Juvenile and immature crabs with whit color and narrow ovaries which occupy little space of abdomen. Such stage had an ascending procedure from November to January (Fig. 5). The ovary tissue is corrugated a lot and the immature ova are embedded among these corrugations. The cytoplasm is extremely alkalophile which becomes purplish during coloration (Fig. 7). The ovary morphology is similar to stage 2. The only difference between these two stages is the tissue because it is not maturely distinctive and the oocytes` diameters average is <20 μm (Fig. 3).
Rudimentary stage: The ovary is milky white to light cream and its volume increase is completely obvious. At this stage, the crab is immature. Immature ova can be seen with high density. Reproductive centers and embedded among the lobs and consist of oogonia. The nucleus is large and has 1 or 2 nucleoli which have irregular external surfaces (Fig. 8). The peak of such stage was seen in November and December (Fig. 5). The averages of oocytes`s diameters are 20 to 40 μ (Fig. 3).
Developing stage: The ovary color is darker orange compared to the previous stage and ranges from cream to light orange. Entering such stage indicates the ovary maturity which can be seen during the whole year except December (Fig. 5). Two distinct Ova can be seen, one with larger diameter and one in first maturity stage. There are Follicular cells round the large ova and yolk formation
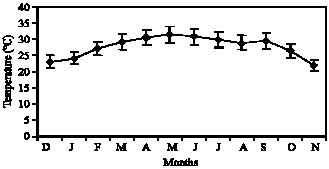 | |
Fig. 1: | Mean month fluctuations of water temperature (°C) in Chabahar bay |
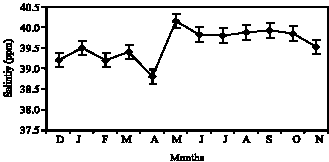 | |
Fig. 2: | Mean month fluctuations of water salinity in chabahr bay |
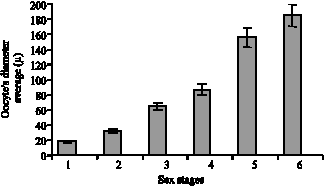 | |
Fig. 3: | Mean oocyte`s diameter±standard error (μ) of sex stages |
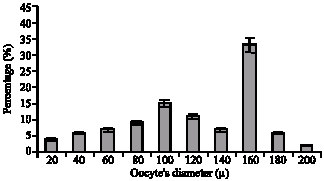 | |
Fig. 4: | Frequency distribution oocyte`s diameter (mean±SE) in Ocypode saratan |
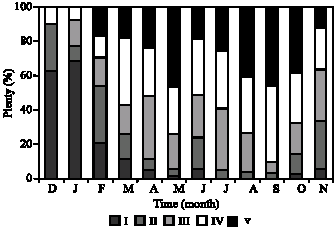 | |
Fig. 5: | Plenty of sex stage`s female in ocypode saratan |
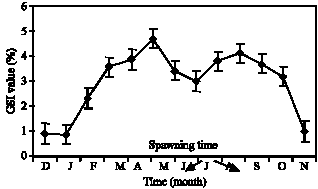 | |
Fig. 6: | Monthly fluctuations of gonadosomatic index (mean±SE) ocypode saratn |
process can be seen through vitilin increase in cytoplasm (Fig. 9). The average of oocytes` diameters is between 40 to 70 μ (Fig. 3).
Intermediated stage: The color changes to dark orange and granularity of the ovary appearance is the remarkable indication of such stage. The ovary occupies most of the abdomen. Two peaks happen in April and May. April peak is more (Fig. 5). At this stage, the crab is mature. Immature oocytes are seen with alkalophil cytoplasm and mature oocytes are seen with acidophil cytoplasm with are darker red. The majority is with mature oocytes (Fig. 10). The average of oocytes` diameters is between 70 to 90 μ.
Advanced stage: The bright orange color and the granular appearance are completely obvious. This stage is exactly before the spawning and can be considered as ultimate limit of the ovary maturity and gonads occupy abdomen anterior region as well as Carapace cavity. This stage consists of 2 peaks in October and June. The peak of June is greater. Yolk vacuoles has increased, while the number of empty vacuoles, which are the places of fat storage,
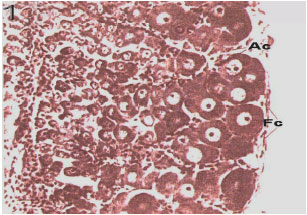 | |
Fig. 7: | First stage of Ocypode saratan ovary (Hematoxylin-Eosin-x180) |
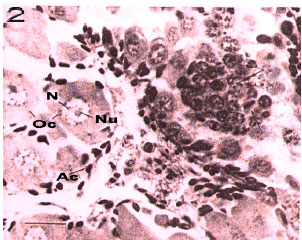 | |
Fig. 8: | Second stage of Ocypode saratan ovary in immature female (x180) |
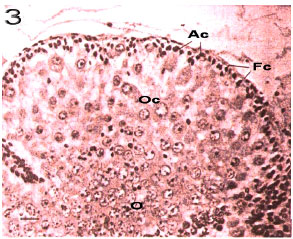 | |
Fig. 9: | Third stage of Ocypode saratan ovary in immature female. Oogonium and immature oocyte are seen (x180) |
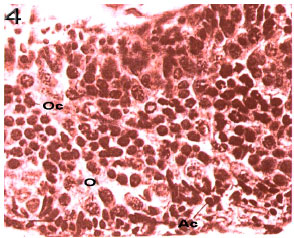 | |
Fig. 10: | Fourth stage of Ocypode saratan ovary. At pre-yolk-formation stage in a mature female. Accessory cells inside the lobe participate in surrounding process of the mature oocytes (x180). Ac: Accessory cells, Fc: Fibrous capsule, N: Nucleus, Nu: Nucleolus, Oc: Oocyte, O: Oogonium and Yg: Yolk globule |
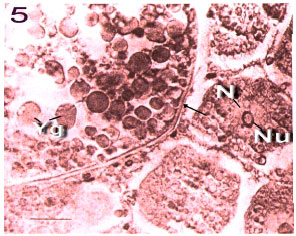 | |
Fig. 11: | Fifth stage of Ocypode saratan ovary with a choronic membrane (arrow). Big yolk globule round the nucleus and smaller grains further (x180) |
has decreased. The mature oocytes have reached their maximum number (Fig. 11). The average of oocytes` diameters is between 90 to 160 μ (Fig. 3)
Resting stage: The ovary situation is in one of the stages 2 to 5 (mainly corroded and in dark cream color) and the oocytes are stuck to ventral feet. Two kinds of cells can be seen in the ovary tissue section: one is small immature ova and another is large mature ova which have polygonal cell walls and absolutely degenerate. Yolk vacuole and fat particles are mixed with each other and cell wall is destroyed. The average of oocytes diameters is >160 μ (Fig. 3).
The assessment of oocytes` diameters showed that maximum frequency oocytes, diameters in Ghost crab are 152.3±0.5 microns (Fig. 4). In addition, Gonad index was determined which is an indirect method to estimate the species` spawning season. Maximum amount of such index in female sex is registered 4.12±0.45 in June and then 4.70±0.45 in October (Fig. 6).
DISCUSSION
Salinity has complementary effect on temperature and increase in salinity results in complete maturity of gonads. Temperature peak in Chabahar Bay is in accordance with maturity peak. Decrease in temperature and salinity causes the spawning in autumn. Crabs can have reproductions during the whole year along with a lot spawning peaks when the salinities are high and fixed. In lower and alternative salinities, the number of the peaks decreases (Burggren and McMahon, 1988). Two environmental factors such as temperature and salinity can have the most effect on gametogenesis (Cobo and Fransozo, 2003). Bed situation, available food amount, amount of density in each region, salinity variation, temperature and waves` intensity can all be the influential factors in immortality, growth and reproduction (Smith et al., 1999). Considering gonads recognition indices such as color, ovum size and the amount of body cavity occupation, researchers have regarded some keys. Such divisions are various in 5 to 6 stages (Mouton and Felder, 1995; Goodman, 1964). Assessing the histology of ghost crab ovary, it is proved that above-mentioned species has 6 main maturity stages. The crab is immature at first and second maturity stage almost consists of the most percentage of the observation. Some crabs enter such stage for the first time in summer and winter. In autumn and spring, most of the crabs spawn and have empty ovaries. Since such stage is the beginning of yolk formation and some oocytes can also be seen at developed stages, this species has Batch fecundity (Fig. 8) which has been proved by other researchers (Dias and Fransozo, 2003). At third maturity stage, we face to increasing procedure of cytoplasm amount in most cells (Fig. 9). At the fourth maturity stage, yolk-information activity increases and a lot of fat vacuoles can be seen which are often non-condensed inside the nucleus, therefore; they are seen orbicularly at this stage (Fig. 10). However, at the fifth maturity stage, the secondary cells, which can be seen round each oocyte, become clear as much as they cause the isolation of the oocyte from other parts of the cytoplasm (Fig. 11). Such stage can be seen in females during the whole year except November, December, January and August (Fig. 5) which implies the spawning of this crab during the year. These stages are almost in accordance with Bonchenary (2000) researches in Bandare Abas region about the species Portunus pelagicus. This stage has two October and June peaks during the year. June peak is greater. These two months coincide with the maximum amount of the first and second maturity stages and crab`s spawning. At this stage, the ascending procedure of the yolk formation continues and the numbers of mature oocytes reach its maximum amount. The procedure of ova diameters fast growth from stage 4 to stage 5 is because of ova dehydration and ova metabolic need to a proper condition for protein-Formation. Considering the results, the female sex of the species Ocypode saratan almost has two peaks of gonad increase during the whole year and June peak is greater because of the most temperature and salinity complementary effect (Fig. 1, 2, 4). Therefore, it has two spawning season. Spring spawning is from March to the end of June and then autumn spawning is from the end of August to the first of November. Gonad maturity has been seen in high percentage of ghost crab females during all months which are in accordance with recent studies on Brazilian coastline. This shows the accuracy of the studies (Reigada and Fransozo, 2000). Continuous reproduction all over the year in the regions which have limited seasonal variation such as sea depths or some parts of tropical regions is something predictable (Giese and Pears, 1974).
Gonad index changes (Fig. 6) have shown increase during March, April, May, June, September and November and the maximum was in June. Such increase coincided with the increase in ovary weight, the number of ova and also ovum diameter. On Oman coast the females carry the oocytes to the ocean during June and July (Vannini and et al., 1989). Such stage happens in summer in Carolina (Dennis, 2003). On the Easter coast of America, spawning has been reported in June (Anonymous, 2000). Such differences depend on geographical situation changes and especially. Temperature and salinity of the regions under study (Weinstein, 2003). In Chabahar Bay regions maximum frequency of ghost crab oocytes` diameters was 152.3±0.5 μ and the average of oocyte diameters was 150±0.5 μ (Fig. 3, 4). On Brazilian coastline, the average of oocyte diameter in females Ocypode quadrata was 146.17 μ (Adilson et al., 2003). Histological evidence shows the different procedures of the ovum maturity in the ovary during a certain time. Therefore, the ovary was of Asynchronous kind. The existence of such ovary in other crabs of ocypodidea family has also been proved (Adilson and Bertini, 2003). Same as the other species depends on temperature, salinity and all physical and chemical factors of the environment and the achieved results of a region can not be used for other regions.