ABSTRACT
There is an increasing evidence for the role of cysteine proteases cathepsin B, L and S in cancer progression. Cathepsin S was identified and isolation from breast cancerous tissue by using the techniques of electrophoresis and chromatography (Gel filtration and reverse phase HPLC). Purity of cathepsin have been observed by 8-25 percent polyacrylamide gradient gel on phast system which gave a single band of approximately 25,000 dalton. Charge gel electrophoresis on 10 percent polyacrylamide gel showed the neutrality of cathepsin.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2000.508.510
URL: https://scialert.net/abstract/?doi=pjbs.2000.508.510
INTRODUCTION
Cathepsin are lysosomal enzyme (digestive enzyme) and include members of diverse class of proteases such as cysteine proteases (Cath B, L, H, S, O, C, K, W), aspartyl proteases (Cath D), serine proteases (Cath G) and metalloproteases (thermolysin). Like cysteine proteases, cathepsin S synthesizes as preproenzyme which is processed to the corresponding proenzyme and is targeted to lysosome by mannose 6-phosphate signal. The enzyme is further processed to mature forms, which consist of single polypeptide chain. In some cases, precursors of these lysosomal proteases escape from the processing pathway, continue along the secretary route, enter the storage granules and are finally released into extracellular space (Petanceska and Devi, 1992).
These cysteine proteases including cathepsin S are implicated in many diverse cellular events, including receptor internalization, protein turnover, neuronal degeneration as well as regeneration during development (Kornfeld and Mellmann, 1989) and antigen processing (Golde et al., 1992), bone remodeling (Tezuka et al., 1994) and prohormone processing. In addition these appear to be involved in a variety of pathological processes such as glomerulonephritis, Alzheimer's disease, cancer invasion and metastasis (Sloane, 1990).
It is hypothesized that protease secreted from transformed cells, degrade extracellular matrix and thus contribute to tumor invasion. Tumor invasion and degradation is though to occur in extracellular environment where pH is neutral. It is observed that cathepsin S protease and plays a role in matrix degradation (Portnoy et al., 1986). Different tumors seem to use distinct proteases or in combination with other proteases act to invade adjacent tissue. Possibility exists that beside cathepsin B and L, cathepsin S may be produced by breast tumor.
Purpose of study is to isolate, and sequence align cathepsin S with other human cathepsin and also construct a model of cathepsin S to identify the active site, specify the mode of binding of substrate and elucidate the enzymatic mechanism of cathepsin S.
MATERIALS AND METHODS
Human tissue was obtained from surgery department of Sir Ganga Ram Hospital Lahore. Tissue was characterized as malignant. Extraction and autolysis was carried out following Dalet-Fumeron et al. (1991) method. The crude extract was purified employing column chromatography (Bromme et al., 1989; Dolenc et al., 1992). Protein estimation and activity of enzyme crude extract was carried out using the techniques of Bollag et al. (1996a) and Maciewicz and Etherington (1988). Inhibitory assay was also performed to check the inhibition of cathepsin using egg white cystatin following the method of Shi et al. (1992). Slab gel/phast gel electrophoresis was performed in both reducing and non-reducing condition according to the method of Laemmli (1970). Gels were silver stained according to the method of Swain and Ross (1995). Amino acid analysis of purified cathepsin S was performed using the technique of Bollag et al. (1996b).
RESULTS AND DISCUSSION
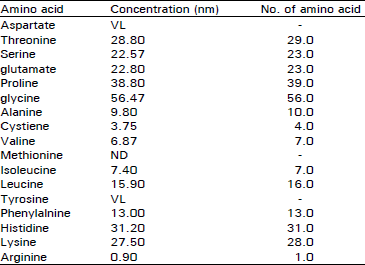
Total protein content of crude sample was found to be 7 mg/ml. Protein concentration of fractions 1, 2 and 3 of gel permeation chromatography was 4.0, 2.0 and 1.0 mg/ml respectively. Activity of cathepsin S was 0.2 units/ml in fraction 2 of gel filtration column (Sephadex G-75). Activity in active fractions (peak 2 and 3) of HPLC gel filtration columns (TSK G 3000SW, TSK G 2000 SW) was 0.25 units/ml and 0.3 units/ml.
| Table 1: | Amino acid analysis of N-terminal blocked cathepsin S from breast cancerous tissue |
 | |
 | |
| Fig. 1(a,b): | Electrophorsis of curde tissue extract was carried out using 12.5% polyacrylamide gel under reducing and non reducing condition |
 | |
| Fig. 2: | Gel permation chromatography of crude tissue extract was applied on Sephadex G-75 |
 | |
| Fig. 3: | Fractions of peak 1,2 and 3 of gel permeation chromatography were analyzed by 12.5 percent SDS polyacrylamide gel electrophhoresis |
 | |
| Fig. 4: | HPLC gel permeation of fraction 2 containing cathepsin S activity. Col: TSK G 3000 SW |
Inhibitory assay showed that cystatin inhibit approximately 90 % activity of cathepsin S instead of 100 % as reported. The reason may be sample impurity or slow inhibition kinetics exhibited by cystatin (Fox et al., 1992).
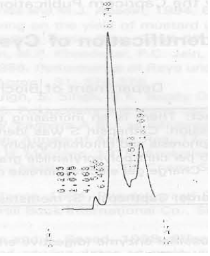 | |
| Fig. 5: | HPLC gel permeation (Col: TSK G 2000 SW) of fraction (peak 3) containing cathepsin S activity after TSK G 3000 SW column |
 | |
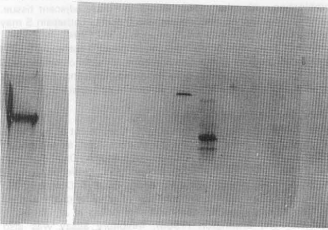
| Fig. 6a: | Fraction of TSK G 3000 SW were analyzed by 8-25% polyacrylamide gradient gel electrophoresis A) Dye standard marker. B) Fraction containing cathepsin S activity |
 | |
| Fig. 6b: | Fraction of TSK G 2000 SW containing cathepsin S activity was analyzed by 10 percent polyacrylaamide gel under non reducing condition and reducing condition |
Twining (1994) proposed the mechanism of enzyme inhibition is that enzyme inhibitor complex is mainly formed by hydrophobic interaction between chicken cystatin and the complementary active site of cathepsin S.
Amino acid analysis (Table 1) shows the neutrality of enzyme as the ratio of (lys+arg) (glu+asp) >1. Among hydrophobic residues those of glycine, alanine leucine are more prominent. In hydrophilic residue those of serine, threonine and glutamate are observed.
Pre-fractionation of crude tissue extract on gel filtration (Sephadex G-75) resulted in 3 peaks (Fig. 1). Fractions of these peaks were pooled and concentrated in vacuo. The concentrated fraction of peak 2 containing cathepsin S activity (active fraction) was chromatographed on a TSK G 3000SW and resulted in 7 peaks (Fig. 2). Concentrated active fraction of peak 3 from TSK G 3000SW column was chromatographed on a TSK G 2000SW column and resulted in 4 peaks (Fig. 3). Activity of cathepsin S was found to be in fraction of peak 2.
Purity of cathepsin S was observed by 8-25% polyacrylamide gradient gel on phast system which give a single band of approximately 25,000 dalton (Fig. 4). Charge gel electrophoresis on 10% polyacrylamide gel shows the neutrality of cathepsin S (Fig. 5).
Cysteine proteases are remarkably widespread and are present in all forms of life. They all belong to papain super family. The papain super family is divided into 2 families; the papain family and calpain family. There may be 2 branches of papain family the one including cathepsin L. H and papain and the other containing cathepsin B and helminth cysteine protease. The enzyme range in size of the first group is shorter than the second group (Kominami et al., 1992). In order to confirm that human cathepsin is a cystiene protease, homology with other cystiene proteases and a three dimensional structure of cathepsin S has to be constructed (Fig. 6a, b).
REFERENCES
- Petanceska, S. and L. Devi, 1992. Sequence analysis, tissue distribution and expression of rat cathepsin S. J. Biol. Chem., 267: 26038-26043.
Direct Link - Golde, T.E., S. Estus, L.H. Younkin, D.J. Selkoe and S.G. Younkin, 1992. Processing of the amyloid protein precursor to potentially amyloidogenic derivatives. Science, 255: 728-730.
CrossRefDirect Link - Sloane, B.F., 1990. Cathepsin B and cystatins: Evidence for a role in cancer progression. Semin. Cancer Biol., 1: 137-152.
PubMedDirect Link - Dalet-Fumeron, V., N. Guinec and M. Pagano, 1991. High-performance liquid chromatographic method for the simultaneous purification of cathepsins B, H and L from human liver. J. Chromatogr. B: Biomed. Sci. Applic., 568: 55-68.
CrossRefDirect Link - Bromme, D., A. Steinert, S. Friebe, S. Fittkau, B. Wiederanders and H. Kirschke, 1989. The specificity of bovine spleen cathepsin S. A comparison with rat liver cathepsins L and B. Biochem. J., 264: 475-481.
CrossRefDirect Link - Dolenc, I., A. Ritonja, A. Colic, M. Podobnik, T. Ogrinc and V. Turk, 1992. Bovine cathepsins S and L: Isolation and amino acid sequences. Biol. Chem. Hoppe-Seyler, 373: 407-412.
CrossRefDirect Link - Maciewicz, R.A. and D.J. Etherington, 1988. A comparison of four cathepsins (B, L, N and S) with collagenolytic activity from rabbit spleen. Biochem. J., 256: 433-440.
CrossRefDirect Link - Shi, G.P., J.S. Munger, J.P. Meara, D.H. Rich and H.A. Chapman, 1992. Molecular cloning and expression of human alveolar macrophage cathepsin S, an elastinolytic cysteine protease. J. Biol. Chem., 267: 7258-7262.
Direct Link - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - Swain, M. and N.W. Ross, 1995. A silver stain protocol for proteins yielding high resolution and transparent background in sodium dodecyl sulfate‐polyacrylamide gels. Electrophoresis, 16: 948-951.
CrossRefDirect Link - Fox, T., E. de Miguel, J.S. Mort and A.C. Storer, 1992. Potent slow-binding inhibition of cathepsin B by its propeptide. Biochemistry, 31: 12571-12576.
CrossRefPubMedDirect Link - Twining, S.S., 1994. Regulation of proteolytic activity in tissues. Crit. Rev. Biochem. Mol. Biol., 29: 315-383.
CrossRefDirect Link - Kominami, E., K. Ishido, D. Muno and N. Sato, 1992. The primary structure and tissue distribution of cathepsin. Biol. Chem. Hoppe-Seyler, 373: 367-374.
CrossRefDirect Link - Tezuka, K., Y. Tezuka, A. Maejima, T. Sato and K. Nemoto et al., 1994. Molecular cloning of a possible cysteine proteinase predominantly expressed in osteoclasts. J. Biol. Chem., 269: 1106-1109.
PubMedDirect Link - Portnoy, D.A., A.H. Erickson, J. Kochan, J.V. Ravetch and J.C. Unkeless, 1986. Cloning and characterization of a mouse cysteine proteinase. J. Biol. Chem., 261: 14697-14703.
PubMedDirect Link








