ABSTRACT
Investigations were carried out on the non-target effects of two new fungicides, Amistar and Score (Syngenta, Mumbai, India) in terms of phytotoxicity. These fungicides are found to be generally non phytotoxic at or below the recommended dose for field application (2.2 μg (a.i.) mL-1). At higher concentrations, the extent of phytotoxicity of Azoxystrobin and Difenoconazole varied with host genotype. Among the test plants, Vigna catjung was most sensitive to both the fungicides for all the studied parameters like seed germination, shoot elongation, root elongation, number of lateral roots initiated and may serve as good indicator of phytotoxicity of these fungicides. Both the fungicides at their different concentrations significantly decreased community respiration and gross primary productivity. However, the net primary productivity was significantly increased by Azoxystrobin treatment up to 0.0073 μg (a.i.) mL-1 and Difenoconazole up to 0.0014 μg (a.i.) mL-1 concentrations. Treatment of leaf tissue with Azoxystrobin resulted in electrolyte leakage as measured by increased electrical conductivity (EC). The increase in EC was pronounced with the increase in fungicide concentration and incubation period. The negative EC values obtained in the Difenoconazole treatment may be due to fast and efficient uptake of the fungicide from the ambient solution by the leaf tissue.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ajpp.2006.89.98
URL: https://scialert.net/abstract/?doi=ajpp.2006.89.98
INTRODUCTION
The damage caused to the biological communities in the soil and aquatic environments due to agrochemical application is of serious concern. Plants are the main recipients of pesticides through direct application of pesticides or through the uptake from soil, water and air drift. Phytotoxicity of agrochemicals on crop plants such as ornamentals, vegetables, cereals, oil seeds and fruits may be caused by misuse or misapplication of chemicals and therefore, phytotoxicity is an essential element of biological evaluation (Sahni, 1983).
Fungicides reach aquatic systems by direct application, spray drift from ground or aerial spraying, atmospheric fall out, run-off from agricultural land, discharge of effluent from chemical factories and from sewage. As the progressive use of organic fungicides reached massive proportions in the past century, it becomes increasingly important to study their detrimental effects on aquatic forms of life.
A number of fungicides are being routinely used for crop protection but their phytotoxic effects have been often ignored (Vyas, 1993). Application of systemic fungicides as coatings on the seeds has been suggested as an effective method of controlling pathogenic fungi in contrast to conventional foliar application (Sithanantham, 1973). Fungicides used for seed treatment often interfere with germination and seedling growth (Mitra et al., 1970; Das and Chandrika, 1972; Rao and Rao, 1980; Rajamani et al., 1987). Knowledge on the effect of fungicides on the germination of seeds and the growth of seedlings is essential in choosing desirable fungicides for coating on seeds.
Phytoplanktons play a key role in freshwater ecosystem, as they are the primary producers in the food chain (Kumar and Singh, 2000). They are important in maintaining a proper equilibrium between biotic and abiotic components in an aquatic ecosystem. The most commonly measured functional attributes of an aquatic ecosystem are gross and net primary productivity (GPP and NPP) and community respiration (CR). These two matrices have been shown to be sensitive indicators of ecosystem stress (Jindal and Kaur, 2000). A reduction in GPP and NPP may be due to the impact of pesticides on algal growth and their pigmentation as reported by Anand and Veerappan (1980) and Tarar and Shewale (1984).
Amistar (azoxystrobin 25% EC) and Score (difenoconazole 25% EC) are the two broad spectrum, foliar, systemic fungicides, yet to be released to the farmers and planters in India by Syngenta India Limited, Mumbai. The objective of the present study was to investigate their non-target effects in terms of phytotoxicity at concentrations ranging from 0.44 - 22000 μg (a.i.) mL-1. For both the fungicides, manufacturer’s recommended dose for foliar spray is 2.2 μg (a.i.) mL-1.
MATERIALS AND METHODS
Test Plants
The test plants used include certain legumes (Cicer arietinum Linn., Dolichos biflorus Roxb., Phaseolus aureus Roxb., Phaseolus mungo Linn., Vigna catjang Walp), non-legumes (Brassica juncea Coss., Capsicum frutescens Linn., Lycopersicum esculentum Mill., Sesamum indicum Linn., Trigonella foenumgraecum Linn.) and cereals (Pennesetum typhoides Linn., Sorghum vulgare Pers., Triticum aestivum Lamk, Zea mays Linn.). All the seeds were obtained from the seed section of Tamil Nadu Agricultural University, Coimbatore. All sets of measurement were repeated by conducting a separate set of measurements on a separately executed experiment.
Seed Germination Assay
Seed germination assay was used to evaluate the phytotoxicity, if any, of the fungicides Azoxystrobin and Difenoconazole. Prior to treatment with fungicides, the seeds of the test plants were surface sterilized in 0.1% mercuric chloride for 1-2 min followed by repeated rinsing with distilled water. The surface sterilized seeds were then soaked overnight (12 h) in different concentrations of the fungicides.
One hundred treated seeds were spread in a petri dish (15 cm dia.), lined with moist filter paper and allowed to germinate. Germination was observed for seven days. Triplicate plates were maintained for each treatment. Alteration in germination percentage of the test seeds due to fungicide treatment was calculated as follows:
Parameters like shoot and root elongation besides the number of lateral roots initiated, were also noted.
Effect on Membrane Stability (Tripathi et al., 1982)
The effect of different fungicide concentrations on the membrane stability of plant tissue was studied using the third unfolded leaves of Coccinea indica L. The fresh leaves were washed in tap water followed by distilled water. Leaf discs of 1 cm diameter and uniform thickness were cut using a cork borer. The discs were blotted and 10 pieces were floated in 250 mL of fungicide concentrations taken in 500 mL beakers and incubated at 25°C in dark with minimum agitation for 3, 6 and 12 h. After incubation, leaf segments were removed and the ambient solutions were used to measure the ion efflux by conductivity measurements using a Digitalmeter (CyberScan pH/Ion 510, Eutech Instruments, Singapore). Blanks with fungicide concentrations in which leaf discs were not incubated, were also used to account for any conductivity due to fungicide itself, which were tared from the treatment values.
Effect on Primary Production
Primary production was determined using the light and dark bottle method of Wallenweider (1975). Different concentrations of Azoxystrobin and Difenoconazole were prepared and 1mL was added into the initial oxygen bottle (IB), light bottle (LB) and dark bottle (DB) before the water sample is collected. One of the simple methods of estimating primary production is the determination of dissolved oxygen evolved during photosynthesis. The increase in dissolved oxygen of water as a result of photosynthesis is measured in a BOD bottle (light bottle) containing a sample of water under study. Simultaneously, the decrease in oxygen content in a darkened bottle was measured to estimate the respiration alone in the sample of water. From the data obtained thus, gross and net photosynthesis and the respiration of the community are calculated.
Three BOD bottles [two clear bottles i.e., initial oxygen bottle (IB) and light bottle (LB) and one dark bottle (DB)] were filled with the water sample collected from Singanallur pond, Coimbatore. The light bottle (LB) and dark bottle (DB) were suspended to a depth of ½ feet in the pond by means of a thread and the amount of oxygen in the sample was fixed in the initial oxygen bottle (IB) for the estimation of dissolved oxygen using a stirred oxygen electrode chamber (Orion Research, model 290A, USA). The samples (LB and DB) were allowed to remain in the location for 2 h so that photosynthesis and respiration take place. To maintain uniformity, the experiment was carried out every time between 10 am and 12 pm. After incubation time, the LB and DB samples were removed from the pond and the oxygen content in the sample bottles was fixed and the amount of dissolved oxygen was calculated. Three replicate bottles were maintained for each treatment.
Calculation
The primary productivity was calculated as follows: -
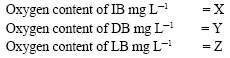 |
Then,
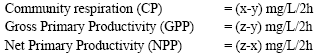 |
RESULTS
Effect on Seed Germination
Azoxystrobin and Difenoconazole enhanced percent seed germination in S. indicum and P. mungo at all concentrations studied. In six other test plants (C. frutescens, C. arietinum, L. esculentum, P. mungo, S. vulgare and Z. mays), the former increased the percent seed germination up to the recommended dose (2.2 μg (a.i.) mL-1) beyond which a steady decline in the percent seed germination was noted. Similar observation was made with Difenoconazole also in the test plants S. indicum, P. typhoides and S. vulgare. In all other test plants, there was a gradual decrease in percent seed germination with increasing concentrations of the said fungicides. A total inhibition of seed germination was not noted in any of the test plants treated with Azoxystrobin. However, higher concentrations (4400 and 22000 μg (a.i.) mL-1) of Difenoconazole resulted in 100% inhibition of seed germination in D. biflorus, B. juncea, S. indicum and S. vulgare (Table 1).
Effect on Shoot Elongation
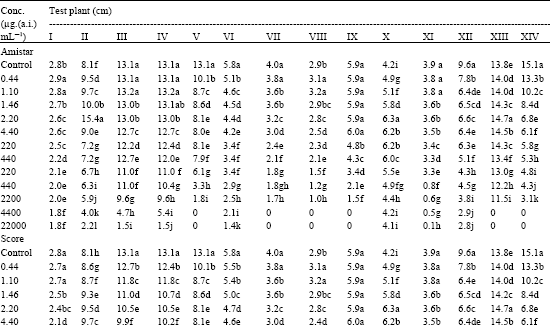
Of the 14 test plants treated with Azoxystrobin, five viz., C. arietinum, D. biflorus, S. indicum, T. foenumgraecum and T. aestivum showed gradual increase in shoot length up to the recommended dose. Beyond the recommended dose (Table 2), the shoot elongation was progressively decreased with increasing concentrations of both the fungicides.
Effect on Root Elongation
Of the 14 test plants, only four viz., V. catjung, S. indicum, P. typhoides and S. vulgare showed progressive decrease in root length with increasing concentrations of Azoxystrobin (Table 3). In all the other test plants, the root length increased with the fungicide concentration to certain levels beyond which progressive decrease with concentration was noted. Difenoconazole treatment also promoted root elongation in certain plant species (C. arietinum, D. biflorus, P. aureus, P. mungo, B. juncea, C. frutescens and T. foenumgraecum) in the lower concentrations, but the changes were not as pronounced as in Azoxystrobin.
Effect on Lateral Root Initiation
Lower concentrations of Azoxystrobin promoted lateral root initiation in D. biflorus, S. indicum, P. typhoides and T. aestjvum. In other test plants, gradual decrease in the number of lateral roots was observed with increasing concentrations (Table 4). Among the Difenoconazole treated seedlings, C. arietinum, D. biflorus, S. indicum, P. typhoides, S. vulgare and Z. mays registered enhanced lateral root initiation at lower concentrations. In the other plants, the lateral root initiation was progressively decreased with increasing concentrations of Difenoconazole.
Lateral roots were not initiated within seven days of germination in certain test plants (B. juncea, C. frutescens, L. esculentum and T. foenumgraecum). So, it was not possible to study the effect of fungicides on lateral root initiation in these species.
Effect on Membrane Stability
The effect of Azoxystrobin and Difenoconazole on the membrane stability is expressed in terms of increases in electrolyte leakage over untreated control, from the leaf discs of Coccinea indica (Table 5). Blanks with fungicide concentrations in which leaf discs were not incubated, were also used to account for any conductivity due to fungicide itself, which were tared from the treatment values. Treatment of leaf tissue with Azoxystrobin resulted in electrolyte leakage as measured by increased electrical conductivity (EC). The increase in EC was pronounced with the increase in fungicide concentration and incubation period. However, in the case of Difenoconazole, the electrolyte leakage was not obvious even at higher fungicide concentrations and extended incubation periods. The EC of Difenoconazole treatment showed only negative values (because EC due to fungicide concentrations were tared from treatment values) due to fast and efficient uptake of the fungicide by the leaf tissue.
Effect on Primary Productivity
Table 6 shows that both the fungicides at their different concentrations significantly decreased community respiration and gross primary productivity.
| Table 1: | Effect of Amistar and Score on seed germination in various test plants (expressed as % change in germination) | |
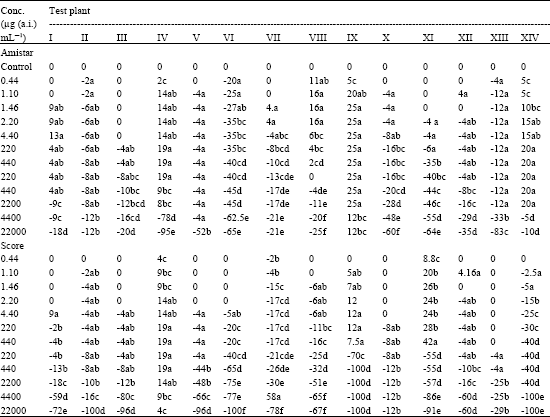 | ||
| Negative and positive values indicate respectively germination inhibition and germination promotion. I. Cicer arietinum, II. Dolichos biflorus, III. Phaseolus aureus, IV. Phaseolus mungo, V. Vigna catjung, VI. Brassica juncea, VII. Capsicum frutescens, VIII. Lycopersicum esculentum, IX. Sesamum indicum, X. Trigonella foenumgraecum, XI. Pennisetum typhoides, XII. Sorghum vulgare, XIII. Tritcum aestivum, XIV. Zea mays, In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | ||
| Table 2: | Effect of Amistar and Score on shoot length (cm / plant) of test seedlings |
 | |
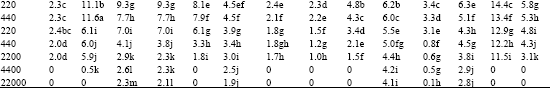 | |
| I. Cicer arietinum, II. Dolichos biflorus, III. Phaseolus aureus, V. Phaseolus mungo, V. Vigna catjung, VI. Brassica juncea, VII. Capsicum frutescens, VIII. Lycopersicum esculentum, IX. Sesamum indicum, X. Trigonella foenumgraecum, XI. Pennisetum typhoides, XII. Sorghum vulgare, XIII. Tritcum aestivum, XIV. Zea mays, ‘0’ indicates inhibition of seed germination and hence no growth. In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | |
| Table 3: | Effect of Amistar and Score on root length (cm.plant-1) of test seedlings |
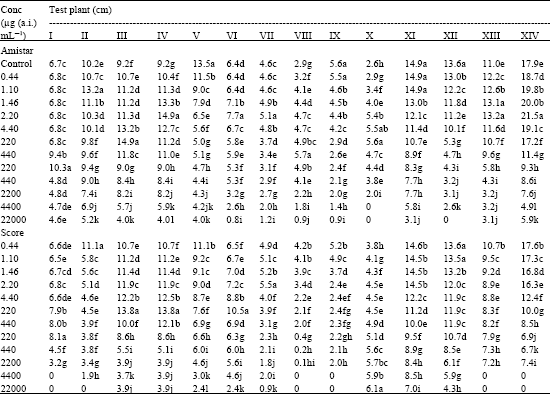 | |
| I. Cicer arietinum, II. Dolichos biflorus, III. Phaseolus aureus, VI. Phaseolus mungo, V. Vigna catjung, VI. Brassica juncea, VII. Capsicum frutescens, VIII. Lycopersicum esculentum, IX. Sesamum indicum, X. Trigonella foenumgraecum, XI. Pennisetum typhoides, XII. Sorghum vulgare, XIII. Tritcum aestivum, XIV. Zea mays, In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | |
| Table 4: | Effect of Amistar and Score on the number of lateral root (Number / plant) initiation in the test seedlings |
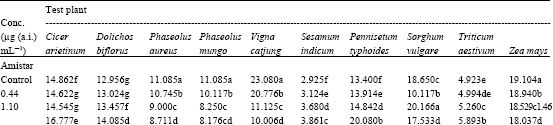 | |
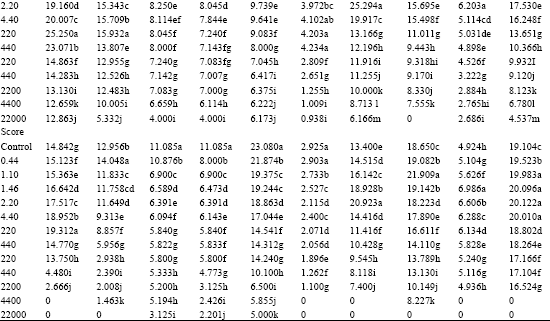 | |
| In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | |
| Table 5: | Electrolyte leakage* (mScm-1) from the leaf discs of Coccinea indica Linn. as influenced by different concentrations of the fungicides Amistar and Score |
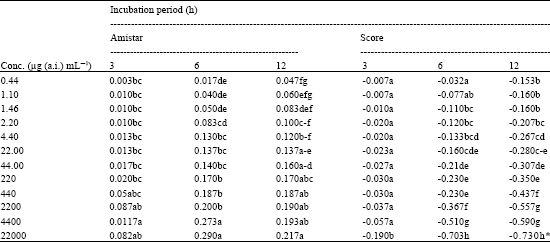 | |
| Measured as electrical conductivity (EC) of the ambient solution. The EC due to fungicide concentrations were tared, In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | |
However, the net primary productivity was significantly increased by Azoxystrobin treatment up to 0.0073 μg (a.i.) mL-1and Difenoconazole up to 0.0014 μg (a.i.) mL-1 concentrations. Higher concentrations, however, were detrimental for net primary productivity.
| Table 6: | Effect of Amistar and Score on community respiration and primary productivity (mg.O2 L h) in a pond ecosystem |
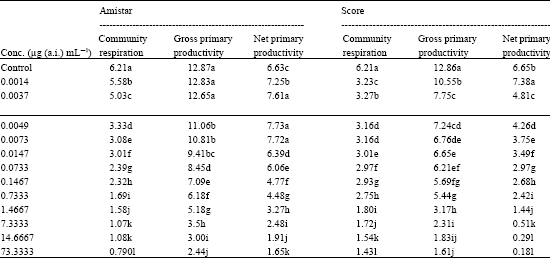 | |
| In a column, means followed by a common letter are not significantly different at the 5% level by DMRT | |
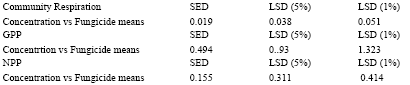 | |
DISCUSSION
One of the easiest and most favoured methods of assessing phytotoxicity is the seed germination assay (Wong and Keturi, 1990). A number of earlier workers have recommended the usefulness of seed germination assay for ecotoxicology studies (Ratsch, 1983; Wang and Williams, 1988). Ratsch (1983) concluded that inhibition of root elongation was a valid and sensitive indicator of environmental toxicity. Miller et al. (1985) and Thomas et al. (1986) used root elongation of five plant species as part of a test battery for hazard assessment of toxic waste sites.
The present study indicated that the fungicides were generally non phytotoxic at or below the recommended dose for field application (2.2 μg (a.i.) mL-1). At higher concentrations, however, both Azoxystrobin and Difenoconazole exhibited concentration dependant phytotoxicity. The systemic fungicide Carboxin and Oxycarboxin were phytotoxic to soybean seedlings at 100 μg mL-1 when incorporated in soil (Gray and Sinclair, 1970). The extent of phytotoxicity of Azoxystrobin and Difenoconazole also varied with host genotype. Among the test plants, V. catjung was more sensitive to both the fungicides for all the studied parameters and may serve as good indicator of phytotoxicity of these fungicides.
A notable observation in the present study was the stimulatory effect of Azoxystrobin and Difenoconazole, at or below the recommended dose, in terms of percent germination, shoot and root elongation and lateral root initiation in a number of test plants. Schulz (1888) first introduced the hypothesis that all poisonous substances are stimulatory in small quantities and has since become the basis of Arndt-Schulz rule (Luckey, 1959).
Only Azoxystrobin affected the membrane stability of plant tissue where the extent of damage was concentration dependent. The other fungicide Difenoconazole had no adverse effect on the integrity of membrane. The negative EC values obtained in the Difenoconazole treatment may be due to fast and efficient uptake of the fungicide from the ambient solution by the leaf tissue. Carbendazim, another systemic fungicide belonging to azole group also prevented the leakage of electrolytes and disorganization of cell organelles at lower concentrations (5 and 20 μg mL-1). However, at 100 μg mL-1 concentration, it s timulated the loss of electrolytes from the wheat leaves (Tripathi et al., 1982).
In aquatic bodies, the biocides have produced unwanted changes in the biota and also upset primary productivity (Saha and Singh, 1981; Somashekar and Sreenath, 1983). Gross and net primary productivity (GPP and NPP) and community respiration (CR) are the most sensitive indicators of ecosystem stress. In the present study, CR was significantly reduced at all concentrations studied whereas the productivity was enhanced at lower concentrations. Similar reports are available on other fungicides like Brassicol, Bavistin, Fytolan, MBC, Difolatan and Hexacap, which are known to be lethal to some nitrogen fixing blue green algae only at higher concentrations (Gangawane and Saler, 1979; Anand and Veerappan, 1980). A gradual reduction in GPP and NPP was noticed with endosulfan, monocrotophos and cumin-L pesticides at low concentration (0.25 ppm) while a noticeable reduction was observed at higher concentrations (beyond 50 ppm) of all the three pesticides (Ravindra and Zohra, 2000).
The present study confirmed that when the two fungicides, Amistar and Score are used at the manufacturer’s recommended doses, there will not be any non-target effects in terms of phytotoxicity.
REFERENCES
- Jindal, R. and B. Kaur, 2000. Effect of heavy metal toxicity on the productivity of a freshwater ecosystem. Ind. J. Ecol., 27: 27-32.
Direct Link - Miller, W.E., S.A. Peterson, J.C. Greene and C.A. Callaham, 1985. Comparative toxicology of laboratory organisms for assessing hazardous waste sites. J. Environ. Qual., 14: 569-574.
Direct Link - Tripathi, R.K., K. Tandon, E. Schlosser and W.M. Hess, 1982. Effect of fungicides on the physiology of plants. Part IV: Protection of cellular organelles of senescent wheat leaves by carbendazim. Pestic. Sci., 13: 395-400.
CrossRefDirect Link - Wang, W. and J.M. Williams, 1988. Screening and biomonitoring of industrial effluents using phytotoxicity tests. Environ. Toxicol. Chem., 7: 645-652.
CrossRefDirect Link - Wong, W. and H.P. Keturi, 1990. Comparative seed germination tests using ten plant species for toxicity assessment of a metal engraving effluent sample. Water Air Soil Pollut., 52: 369-376.
CrossRefDirect Link








