ABSTRACT
Cotton is considered a moderately salt-tolerant crop, however, cotton varieties differ in the level of salt- tolerance and high salinity reduces growth, yield and fiber quality in a different extent. The objectives of this study were to determine the response of selected CRS accessions to salt stress and to identify salt-tolerant CRS accession(s) or individual plants within these accessions that potentially could be used as parental material for further investigation of salt tolerance in cotton. Seedling of four putative salt-tolerant (M-9044-0031, M-9044-0061, M-9044-0140 and M-9044-0150) and three salt-sensitive CRS accessions (M-9044-0060, M-8744-0091 and M-8744-0175) plus Acala 1517-88, Deltapine 50 and TAM 94-L25 were grown under hydroponic culture with two different salt concentrations (150 and 200 mM NaCl) and control. The study was carried out under completely randomized experimental design. Significant differences were observed among cotton genotypes for Shoot (SL) and Root Length (RL) and Shoot Dry Weights (SDW) and Root Dry Weights (RDW) reduction with the increasing salt levels. It was concluded that CRS lines, M-9044-0060, M-9044-0061 and M-8744-0091, may provide additional insights and parental material for breeding for salt tolerance in upland cotton. In addition, selected single plants from salt-tolerant CRS lines were transferred to pots to obtain seeds for further investigation of salt tolerance in cotton.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2006.99.106
URL: https://scialert.net/abstract/?doi=ajpp.2006.99.106
INTRODUCTION
Excessive salt (NaCl) accumulation, salt stress and drought are considered to be major causes of yield reductions in crop plants (Bohnert et al., 1995). Increased salinity in soils is becoming a serious problem due to low rainfall, irrigation, poor water management and/or high evaporation, especially, in hot dry areas where extensive water loss occurs through the combination of evaporation and transpiration. About 20-40% of worldwide agriculture land is subjected to drought and 20-40% of worldwide agriculture production is affected by salinity (Boyer, 1982; Tanji, 1990; Serrano and Gaxiola, 1994). Salt-tolerant cultivars could improve productivity on these marginal lands and thus help meet the demands of the world’s increasing population (Holmberg and Bulow, 1998). Drought and salt-tolerance is expressed by morpho-physiological characteristics (Pessarakli, 2002) and molecular/biochemical (Ozturk et al., 2002) mechanisms that are induced under these stress conditions. The effects of salinity on plant growth are well documented (Hasegawa et al., 2000; Pessarakli, 2002). The primary effects of high levels of salt are caused by ion imbalance and hyperosmotic stress in plants; secondary effects involve oxidative damage to enzymatic proteins and membrane integrity (Zhu, 2001; Xiong and Zhu, 2002). In general, halophytic plants adapted to salt stress by either avoidance (preventing entry or excretion mechanisms), compartmentalization of salt ions into the vacuoles and/or neutralization by compatible osmolytes and anti-oxidants enzymes (Flowers et al., 1986; Zhu, 2001). When plants are subjected to the biotic or abiotic stresses, they produce Reactive Oxygen Species (ROS) (Allen, 1995), which can disrupt normal metabolism through oxidative damage of lipids, proteins and nucleic acids in the absence of any protective mechanisms. Salt-tolerant plants, which posses the ability to regulate ions and water movement, also have a better anti-oxidative system for effective removal of ROS than glycophytes (Rout and Shaw, 2001).
Although cotton is considered as moderately salt-tolerant crop (Soltanpour and Follett, 1995), variation in salt-tolerance has been observed among different cotton cultivars (Gosset et al., 1994; Khan et al., 1995; Leidi and Saiz, 1997). In general, salt-tolerant cotton cultivars have higher seed yield than that of salt-sensitive cultivars (Ashraf and Ahmad, 2000). Salinity delays germination and seedling emergence (Ashraf and Ahmad, 2000) and reduces root development in cotton (Zhong and Lauchli, 1993). However, low salt concentration (100 mM NaCl) has been shown to increase primary root length (Leidi, 1994) and root growth (Jafri and Ahmad, 1994). Shoot growth appears to be more sensitive to salt stress than roots (Leidi et al., 1991). Salinity also causes a decrease in the number of bolls per plant (Longenecker, 1974), cotton seed yield (Khan et al., 1995) and fiber length fiber strength and fiber maturity (Ashraf and Ahmad, 2000). Garrat et al. (2002) and Meloni et al. (2003) reported that salt-tolerant cotton cultivars counter salt-induced ROS formation by increasing anti-oxidant enzyme activity. In addition to the accumulation of anti-oxidant enzymes, high K+/Na+ and Ca++/Na+ ratios in plant tissue have been suggested as a trait-selection criterion for salt-tolerance in cotton (Gosset et al., 1994; Leidi and Saiz, 1997; Ashraf and Ahmad, 2000). According to Ashraf and Ahmad (1999), seed germination and early seedling development are the stages in plant development that are the most sensitive to salt accumulation. Therefore, selection based on phenotypic differences at the initial vegetative growth stages could lead to increased salt-tolerance for cotton. Both drought and salt stress induce similar physiological dysfunctions and their recovery mechanisms, in part, have been shown to overlap (Munns, 2002; Zhu, 2001; Tyreman et al., 2002).
The Converted Race Stocks (CRS) of upland cotton have been identified as potential sources of traits involved in drought-tolerance (Quisenberry et al., 1981; Nasirci and Smith, 1999; Basal et al., 2003). Since the day-sensitive recurrent parents of the CRS lines evolved under semi-arid conditions, it is anticipated that both drought- and salt-tolerant related genes could, theoretically, exist within these lines.
Several of these CRS lines were reported to possessed genetic diversity for root formation and production (Basal et al., 2003). In the study reported herein, 10 robust and 10 non-robust rooting CRS were evaluated for salt-tolerance during germination and during early seedling growth. The main objectives of this research were (i) to determine the response of selected CRS accessions to salt stress, (ii) to identify salt-tolerant CRS accession(s) or individual plants within these accessions that would serve as parental material for further salt-tolerant research in cotton.
MATERIALS AND METHODS
Fifty seeds of each of the 10 robust and 10 non-robust rooting CRS accessions (Basal et al., 2003), TAM94L-25, (Smith, 2003) and salt tolerant, Acala 1517-88, salt sensitive, Deltapine 50 (DPL), (Gosset et al., 1994) were treated with systemic fungicide (Delta-Coat ADTM, Wilbur-Ellis) at 50% (v/v) to prevent fungal growth during germination. Seeds were placed on a germination paper that had been soaked in NaCl solutions of 0, 75 or 150 mM. Pilot studies (data not shown) had indicated that these salt levels would discriminate salt susceptibility and tolerance. Germination tests were repeated twice (data not shown). Seeds having a radical length longer than 0.5 cm were considered germinated. Subsequently, four putative salt-tolerant accessions (M-9044-0031, M-9044-0061, M-9044-0140 and M-9044-0150) and three putative salt-sensitive CRS accessions (M-9044-0060, M-8744-0091 and M-8744-0175) were selected for further tests. Following germination, 20 uniform seedlings that possessed a radical length of 2 cm or more were selected from four putative salt-tolerant and three salt-sensitive CRS accessions and other cotton genotypes and placed under hydroponic culture in a climate controlled greenhouse. The seedlings were maintained in RO water (16L) + modified Glenn’s nutrients (Glenn et al., 1999). Seedlings were grown under these conditions for 3 days before being subjected to the increased salt stress of 0, 150 and 200 mM NaCl. Seedlings were exposed to salt stress by adding 50 mM NaCl at 12 h intervals until the final concentrations of 150 and 200 mM NaCl were reached. Nutrient solution was replaced once a week. The pH level was maintained at approximately 6.3 by adding KOH. The germination tests were done in growth champers and hydroponic experiments were performed in greenhouses that were located at the Borlaug Biotechnology Center on the campus of Texas A and M University at College Station, Texas. As reported here, two experiments were performed during 2003: 27 January-17 February (Experiment 1) and 27 May-16 June (Experiment 2). Air temperatures were regulated at 24 to 27°C for daytime and 21 to 24°C for nighttime with 16/8 h light/dark condition.
Twenty days after transplanting to hydroponic culture, plants were harvested and cut into roots and shoots. Shoot (SL) and root length (RL) and shoot (SDW) and root dry weights (RDW) were measured. Shoots and roots were dried for 48 h at 72°C and dry weight recorded. Data were analyzed by a completely randomized experimental design using the GLM procedure of SAS program (SAS Institute, Cary, NC).
RESULTS
Significant genetic variation was found among the genotypes for Root Length (RL), Root Dry Weight (RDW), Shoot Length (SL) and Shoot Dry Weight (SDW) (Table 1). A significant GenotypexExperiment interaction (Table 1) would be resulted from either the differences in ambient air temperature fluctuations at experiments (21-32°C in experiment 1 and 15.5-48°C in experiments 2) or the heterogeneous nature of the makeup of CRS accessions (Liu et al., 2000). Therefore, data from each experiment were presented and evaluated separately and putative salt tolerant single plants were selected from CRS accessions.
Genotypes varied for all characters regardless of salt concentration in both experiments. In order to compare genotypes grown with and without salt and genotypic differences at 150 and 200 mM NaCl, the reduction in the values of the measured characters affected by salt were compared (Table 2).
| Table 1: | Mean square values of 10 upland cotton genotypes grown in hydroponic culture in 3 levels of salt stress, at College Station, TX, USA 2003 |
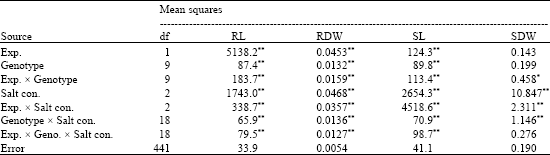 | |
| *, ** Significant at p = 0.05 and 0.01 levels, respectively | |
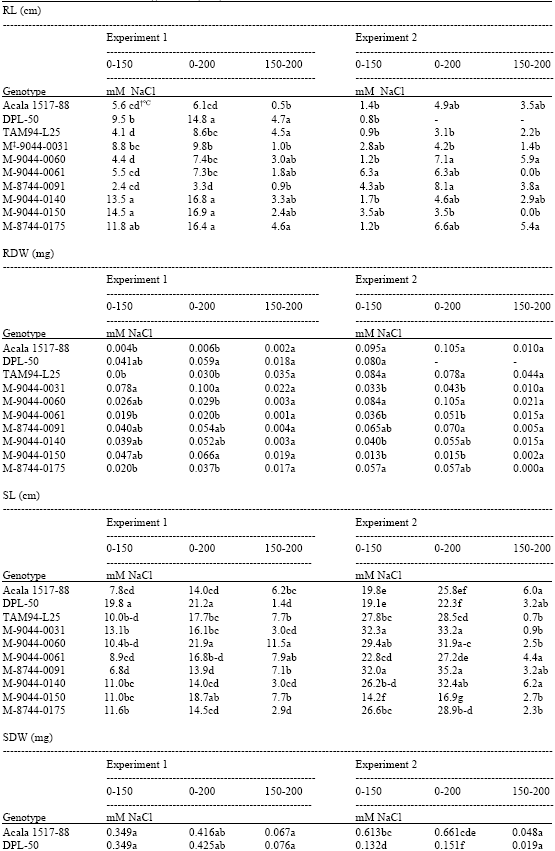
| Table 2: | Reduction in investigated characteristics in 10 genotypes of upland cotton when exposed to 3 levels of salt concentrations at College Station, TX, USA 2003 |
 | |
 | |
| † Means within columns followed by the same lower case letter are not different at k = 100 according to Bayes LSD | |
In experiment 1, the greatest reduction in RL when seedlings were grown in 150 or 200 mM NaCl was observed for 3 of the CRS lines (M-9044-0140, M-9044-0150 and M-8744-0175) along with the cultivar DPL-50 (Table 2). The salt stress germination preliminary study suggested that M-9044-0140 and M-9044-0150 were salt-tolerant based on seed germination test (data not presented). Compared across both salt concentrations, M-8744-0091 and Acala 1517-88 displayed less sensitivity to salt stress while M-9044-0060, M-9044-0061 and TAM 94L-25 appear to be intermediate in salt-tolerance relative to RL (Table 2).
In comparison, the higher ambient temperature resulted in an additional stress factor to the hydroponic grown seedlings besides the salt challenge in the second experimental run (Table 2). More RDW reduction occurred in the second experiment; therefore, the higher air temperatures experienced appears to have caused more sensitivity to salt challenge. The degree of reduction in RDW was much reduced (p = 0.01) in the second experiment relative to the first experiment. The least reduction (p = 0.05) in RDW when plants were grown for 21 days under 150 or 200 mM salt stress occurred for Acala 1517-88, TAM94L-25 and CRS lines M-9044-0060, M-9044-0061 and M-8744-0175 (Table 2). The results for CRS line M-8744-0175 were interesting since it has a relative large reduction in RDW compared with M-9044-0060 and M-9044-0061. The only consistent results when comparing genotypes in experiment one and experiment two was for CRS M-9044-0061, which exhibited lower decreases in RDW than several other genotypes.
Reduction in SL somewhat mirrored the reduction in RL across these genotypes (Table 2). CRS lines M-9044-0140, M-9044-0150 and M-8744-0175 plus DPL-50, which had exhibited larger reductions in RL in one or both experiments, all exhibited large reductions in SL. Acala 1517-88 in both experiments and TAM 94L-25 in the first experiment, along with CRS M-9044-0061 and M-8744-0091 exhibited less reduction overall. However, DPL-50 and CRS lines M-9044-0031, M-9044-0140 and M-8744-0175 displayed less sensitivity than the other genotypes when the salt concentration was increased from 150 to 200 mM NaCl in experiment 1.
In experiment 2, the same phenomenon as observed for SL for CRS M-9044-0150 and M-8744-0091 was also observed for RL. CRS line, M-9044-0150, was among the most susceptible to salt injury as measured by RL and SL in experiment 1 but less susceptible in experiment 2. However, another CRS line, M-8744-0091, was one of the most tolerant genotypes in experiment 1 as indicated by RL and SL yet one of the most susceptible in experiment 2.
No differences were observed in SDW reduction among cotton genotypes when grown without salt stress and when grown in 150 mM NaCl, nor among genotypes when grown at 150 mM and 200 mM NaCl salt levels in experiment 1 (Table 2). However, TAM 94L-25 had less SDW reduction than two CRS lines (M-9044-0031 and M-9044-0150) when salt level increased from 0 to 200 mM NaCl. However, again in experiment 2 the responses of genotypes to increasing salt levels were different from experiment 1 and significant differences were observed among cotton genotypes for SDW reduction with the increasing salt levels. The highest RDW reduction was observed for CRS line, M-8744-0091 at both salt levels, which was somewhat consistent with the reductions in SL.
The CRS, or day neutral lines, described in this study will be designated with the letter M, followed by four digits that indicate the year of release and followed by four digits of the Texas accession number of the unconverted primitive stock accessions held by the United States Department of Agriculture, Agriculture Research Service, National Plant Germplasm System.
DISCUSSION
The impact of adding an additional salt stress above the 150 mM level, i.e., the difference between RL, when grown with 150 and 200 mM NaCl, was less conclusive yet some trends were obvious. For example, the additional salt had numerically the smallest effect on Acala 1517-88 but this genotype was not different than 6 other genotypes in their response to RL reduction. TAM 94L-25 and M-9044-0060 appears to be salt-tolerant at 150 mM but less tolerant to an additional 50 mM NaCl relative to the consistent response in RL. Also, Acala 1517-88 and M-8744-0091 appear less susceptible and DPL-50, M-9044-0140, M-9044-0150 and M-8744-0175 showed a more susceptible response at 150 mM salt concentration.
The response of genotypes to different salt concentration was different at both experiments due to the higher ambient temperature in experiment 2. The 3 CRS lines that appeared to be extremely susceptible to salt, i.e., M-9044-0140, M-9044-0150 and M-8744-0175 were variable in response and not clearly susceptible, while CRS lines M-9044-0060, M-9044-0061 and M-8744-0091 appeared to be tolerant in experiment one but were not so identifiable in experiment two. The second experiment suggested that TAM 94L-25 was as salt-tolerant as Acala 1517-88 but all other conclusion based on experiment one were not supported by experiment two relative to RL. Thus the results between experiments relative to the effect of 150 and 200 mM NaCl on RL are equivocal due to the two variables as noted for experiment 2. Also, none of the CRS lines appeared to be more salt-tolerant than Acala 1517-88 and TAM 94L-25 as defined by RL.
The preliminary germination tests suggested that CRS lines M-9044-0031, M-9044-0061, M-9044-0140 and M-9044-0150 were salt-tolerant while the other CRS lines in this study were more salt-sensitive. However, among four CRS lines mentioned above only M-9044-0031 showed consistent result in RDW reduction at both experiments. These results indicated that the response of some accessions to salinity could be different at seed germination and seedling stage (Kingsburg and Epstein, 1984; Ashraf and McNeilly, 1987).
In both experiments, Acala 1517-88 appears to be the most tolerant of high salt concentrations whether measured by its lower reduction in RL or SL while TAM 94L-25 appears to be intermediate and the CRS lines appear erratic. The other interesting phenomenon that should be pointed out is the small reduction in RL observed in experiment 2 relative to experiment 1 but the opposite was observed for the reduction in SL. However, these data may support the conclusions of Brugnoli and Lauteri (1991) and Meloni et al. (2003) that salt accumulation affected shoot development more than root development due to the reduction in photosynthetic capacity, thus resulting in greater inhibition of normal function in leaves.
Among the CRS lines, only M-9044-0150 was superior to Acala 1517-88 for SDW reduction. Since salt-tolerant cotton genotypes had higher shoot biomass than that of salt- sensitive genotypes, SDW under salt treatment has been proposed to be one of the most important traits to select salt-tolerant genotypes (Ashraf and Ahmad, 2000; Munns, 2002).
If only experiment 1 is considered, where the ambient temperature was not a factor, then one can conclude that CRS M-9044-0060, M-9044-0061 and M-8744-0091 and Acala 1517-88 are the most tolerant of the genotypes tested to salt concentration up to 150 mM NaCl. At a salt concentration of 200 mM NaCl, when considering RL and SL, CRS lines M-9044-0061 and M-8744-0091 along with Acala 1517-88 are the most salt-tolerant. TAM 94L-25 would be classified as intermediate in salt-tolerance when considering RL and SL across both 150 and 200 mM NaCl. RDW reduction would support classifying M-9044-0061 as salt-tolerant across both experimental runs but not Acala 1517-88 nor TAM 94L-25. The variation in response with CRS lines could be due to the heterogeneous nature of the makeup of these cotton germ lines (Nasirci and Smith, 1999; Liu et al., 2000). Also, CRS lines M-9044-0060, M-9044-0061 and M-8744-0091 may provide additional insights and parental material for breeding for salt tolerance in upland cotton.
REFERENCES
- Allen, R.D., 1995. Dissection of oxidative stress tolerance using transgenic plants. Plant Physiol., 107: 1049-1054.
Direct Link - Ashraf, M. and S. Ahmad, 1999. Exploitation of intra-specific genetic variation for improvement of salt (NaCl) tolerance in upland cotton (Gossypium hirsutum L.). Hereditas, 131: 253-256.
Direct Link - Ashraf, M. and S. Ahmad, 2000. Influence of sodium chloride on ion accumulation, yield components and fiber characteristics in salt-tolerant and salt-sensitive lines of cotton (Gossypium hirsutum L.). Field Crops Res., 66: 115-127.
CrossRefDirect Link - Ashraf, M. and T. McNeilly, 1987. Salinity effects on five cultivars/lines of pearl millet (Pennisetum americanum L.). Plant Soil, 103: 13-19.
CrossRefDirect Link - Basal, H., P. Bebeli, C.W. Smith and P. Thaxton, 2003. Root growth parameters of converted race stocks of upland cotton (G. hirsutum L.) and two BC2F2 populations. Crop Sci., 43: 1983-1988.
Direct Link - Bohnert, H. J., D.E. Nelson and R.G. Jensen, 1995. Adaptations to environmental stress. Plant Cell, 7: 1099-1111.
Direct Link - Boyer, J.S., 1982. Plant productivity and environment. Science, 218: 443-448.
CrossRefPubMedDirect Link - Brugnoli, E. and M. Lauteri, 1991. Effect of salinity on stomatal conductance, photosynthetic capacity and carbon isotope discrimination of salt tolerant (Gossypium hirsutum L.) and salt sensitive (Phaseolus vulgaris L.) C3 non-halophytes. Plant Physiol., 95: 628-635.
Direct Link - Garratt, L.C., B.S. Janagoudar, K.C. Lowe, P. Anthony, J.B. Power and M.R. Davey, 2002. Salinity tolerance and antioxidant status in cotton culture. Free Radic. Biol. Med., 33: 502-511.
CrossRefDirect Link - Glenn, E., J.J. Brown and E. Blumwald, 1999. Salt-tolerant mechanisms and crop potential of halophytes. Critical Rev. Plant Sci., 18: 227-255.
Direct Link - Gosset, D.R., E.P. Millhollon and M.C. Lucas, 1994. Antioxidant response to NaCl stress in salt-tolerant and salt-sensitive cultivars of cotton. Crop Sci., 34: 706-714.
CrossRefDirect Link - Holmberg, N. and L. Bulow, 1998. Improving stress tolerance in plants by gene transfer. Trends Plant Sci., 3: 61-66.
CrossRefDirect Link - Jafri, A.Z. and R. Ahmad, 1994. Plant growth and ionic distribution in cotton (Gossypium hirsutum L.) under saline environment. Pak. J. Bot., 26: 105-114.
Direct Link - Kingsburg, R.W. and E. Epstein, 1984. Selection for salt-resistant spring wheat. Crop Sci., 24: 310-315.
CrossRefDirect Link - Leidi, E.O. and J.F. Saiz, 1997. Is salinity tolerance related to Na accumulation in Upland cotton (Gossypium hirsutum) seedlings? Plant Soil, 190: 67-75.
CrossRefDirect Link - Leidi, E.O., R. Nogales and S.H. Lips, 1991. Effect of salinity on cotton plants grown under nitrate or ammonium nutrition at different calcium levels. Field Crops Res., 26: 35-44.
CrossRefDirect Link - Liu, S., R.G. Cantrell, J.C. McCarty, and Jr. McD. J. Stewart, 2000. Simple sequence repeat-based assessment of genetic diversity in cotton race stock accessions. Crop Sci., 40: 1459-1469.
Direct Link - Meloni, D.A., M.A. Oliva, C.A. Martinez and J. Cambraia, 2003. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ. Exp. Bot., 49: 69-76.
CrossRefDirect Link - Munns, R., 2002. Comparative physiology of salt and water stress. Plant Cell Environ., 25: 239-250.
CrossRefDirect Link - Ozturk, Z.N., V. Talame, M. Deyhols, C.B. Michalowski and D.W. Galbraith et al., 2002. Monitoring large-scale changes in transcript abundance in drought and salt-stressed barley. Plant Mol. Biol., 48: 551-573.
Direct Link - Quisenberry, J.E., W.R. Jordan, B.A. Roark and D.W. Fryrear, 1981. Exotic cottons as genetic sources for drought resistance. Crop Sci., 21: 889-895.
CrossRefDirect Link - Rout, N.P. and B.P. Shaw, 2001. Salt tolerance aquatic macrophytes: Possible involvement of the antioxidant enzymes. Plant Sci., 160: 415-423.
CrossRefDirect Link - Serrano, R. and R. Gaxiola, 1994. Microbial models and salt tolerance in plants. Crit. Rev. Plant Sci., 13: 121-138.
Direct Link - Smith, C.W., 2003. Registration of TAM94-L25 and TAM94L-3 germplasm lines of upland cotton with improved fiber length. Crop Sci., 43: 742-743.
Direct Link - Tyerman, S.D., C.M. Niemietz and H. Bramley, 2002. Plant aquaporins: Multifunctional water and solute channels with expanding roles. Plant Cell Environ., 25: 173-194.
PubMed - Xiong, L. and J.K. Zhu, 2002. Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ., 25: 131-139.
CrossRefPubMedDirect Link - Zhong, H. and A. Lauchli, 1993. Spatial and temporal aspects of growth in the primary root of cotton seedlings effects of NaCl and CaCl2. J. Exp. Bot., 44: 763-771.
Direct Link








