Research Article
Role of Nitric Oxide, Glutathione and Sulfhydryl Groups in Zinc Homeostasis in Plants
Department of Botany, Faculty of Science, Suez Canal University, Ismailia 41522, Egypt
After iron, zinc is the most abundant intracellular metal. Virtually all intracellular zinc is associated with proteins (primarily via complex interactions with cysteines), where it is known to be an integral component of numerous metalloenzymes, structural proteins and transcription factors. Zn is an essential catalytic component of over 300 enzymes, including alkaline phosphatase, alcohol dehydrogenase, Cu-Zn superoxide dismutase and carbonic anhydrase. Zn also plays a critical structural role in many proteins (Schjerling and Holmberg, 1996; Bohm et al., 1997). Inside cells, Zn is neither oxidized nor reduced; Thus, the essential role of Zn in cells is based largely on its behavior as a divalentcation that has a strong tendency to form stable tetrahedral complexes (Berg and Shi, 1996).
Zn influx into the root symplasm has been hypothesized to occur as the free Zn2+ ion as well as in the form of Zn complexes with nonprotein amino acids known as phytosiderophores (Tagaki et al., 1984) or phytometallophores (Welch, 1993). Higher plants contain two major types of cysteine-rich, metal-binding peptides, the Metallothioneins (MTs) and the phytochelatins. MTs are gene-encoded polypeptides that are usually classified into two groups. Class 1 MTs possess cysteine residues that align with a mammalian (equine) renal MT; Class 2 MTs also possess similar cysteine clusters but these cannot be easily aligned with Class 1 MTs (Robinson et al., 1993; Prasad, 1999). MT genes have been identified in a range of higher plants (Prasad, 1999) including Arabidopsis where, in addition to Class 1 and Class 2 MT genes, MT3 and MT4 types have been recognized (Goldsbrough, 2000). Other species are also thought to contain an extensive MT gene family and more than one class of MT gene (Giritch et al., 1998), while expression studies have revealed tissue-specific patterns (Garcia-Hernandez et al., 1998; Charbonnel-Campaa et al., 2000; Goldsbrough, 2000). In plants, there is a lack of information concerning the metals likely to be bound by MTs, although Cu, Zn and Cd have been the most widely studied (Robinson et al., 1993; Goldsborough, 2000).
While protein bound cysteine might be used for redox-signaling and regulation of protein activity, this mechanism as such is insufficient to maintain the cellular redox balance as the abundance of many of the proteins might not be sufficiently high for effective redox control. At the same time cells would run a high risk of loosing important proteins through oxidation, if there were no other systems for redox buffering and reduction of oxidized protein cysteine thiols. Thus, in order to keep the internal medium in a reduced state, biological evolution invented a rather complex redox buffering system with the tripeptide glutathione (c-L-glutamyl-L-cysteinyl-glycine) as the most prominent ingredient. Glutathione is a redox active molecule that can be present in its reduced form (GSH) or the oxidized disulfide form (GSSG). The extent of disulfide bridge formation in proteins frequently tracks the oxidation state of the glutathione redox buffer (Schafer and Buettner, 2001). In particular the level and redox state of the cellular glutathione pool co-ordinates some of the key re-adjustments underlying the physiological and developmental plasticity in response to biotic and abiotic stress (Baena-Gonzalez et al., 2001; Noctor et al., 2002; Mou et al., 2003).
Multiple biological functions have been ascribed to NO as a molecule serving signaling or regulating tasks or acting as a cytotoxic molecule, depending on its mode of enzymatic synthesis, its local concentration and its chemical reactions with other molecules. Nanomolar concentrations of NO are synthesized by constitutively expressed NO Synthases (cNOS) in a tightly regulated and pulsative fashion, which typically serve to activate the guanylyl cyclase to synthesize the second messenger cGMP (Ignarro, 1999). In this case, the target of the radical NO is the heme group with its central iron atom. NO may react with molecular oxygen in a reaction mainly depending on the NO concentration to yield higher reactive nitrogen oxides (NOx such as N2O3, etc.), which display a much broader chemical reaction spectrum than NO itself (Wink and Mitchell, 1998). Among the amino acids present in proteins, preferentially cysteines are modified by NOx yielding S-nitrosothiols (Wink et al., 1994). Prominent targets within cells are proteins containing Fe-S or Zn-S clusters. Although Fe-S clusters are essential components of many active sites in enzymes, Zn-S clusters mainly serve as structural elements of proteins mediating specific DNA or RNA binding as well as protein-protein interactions.
In addition to affecting gene expression, S-nitrosation of zinc sulfur clusters can affect intracellular metal ion homeostasis. The chemical biology underlying this process as well as other redox sensitive aspects of zinc-sulfur complexes have recently been reviewed (Maret, 2004). Kroncke et al. (1994) originally showed that NO could S-nitrosate the major intracellular zinc-binding protein, MT (Kroncke et al., 1994) and cause the release of zinquin-detectable changes in free zinc (Berendji et al., 1997). Subsequently, Spahl et al. (2003) noted that iNOS-derived NO increased nuclear Zn and that this increase appeared to require the translocation of MT from cytoplasm to nucleus. Liu et al. (2001) and Chen et al. (2002) confirmed these observations and demonstrated that MT was the requisite target for NO resulting in such changes in free zinc. The metal status of MT was critical for resultant NO-mediated changes in that: (1) NO did not cause release of zinc in a cell in which most of the MT was in its apo form (St. Croix et al., 2002) and (2) Cu-MT was also nitrosated and depending upon the copper status and the amount of NO exposure, CuMT served as a copper chaperone for apo-ZnSOD (Liu et al., 2000) or a source of Fenton reactive copper (Liu et al., 2001). S-nitrosation: (1) requires the presence of molecular oxygen (Schwarz et al., 1995; Aravindakymar et al., 1999), (2) is modified by redox status of the environment (Khatai et al., 2004) and (3) is more facile for the MT-III than other isoforms of MT (Chen et al., 2002). Collectively, it is apparent that S-nitrosation of zinc sulfur clusters is an important component of NO signaling and that metallothoinein appears to be a critical link between NO and intracellular zinc homeostasis (Gow and Ischiropoulos, 2002).
On the basis of the chemical properties of NO, the present research was aimed to investigate whether NO is involved in zinc homeostasis in plants. It also aimed to study the involvement of GSH, SH groups and NO in mediating changes in zinc homeostasis.
Plant Material and Growth Conditions
Wheat seeds (Triticum aestivum L. cultivar giza-167) and bean seeds (Phaseolus vulgaris, cultivar Bronco were supplied by National Research Center (NRC), Dokki-Giza, Egypt. Seeds were surface sterilized in 1.8% (v/v) sodium hypochlorite and rinsed several times in distilled water. Seeds were germinated and grown in vermiculite watered with nutrient solution. The nutrient solution had the following composition: 5.25 mM KNO3, 7.75 mM Ca (NO3)2, 4.06 mM MgSO4 and 1.0 mM KH2PO4; Micronutrients: 46 μM H3BO4, 9.18 μM MnSO4, 40 μM Fe-EDTA, 9.0 μM CuSO4 and 2.0 μM Na2MoO4.
Zinc (ZnSO4) was supplied in different concentrations (0.5, 2.7 μM considered as zinc deficient concentrations, 5.4 as zinc sufficient concentration (control), Graziano et al. (2002), 10.8 and 21.6 μM as zinc toxic concentrations). The nutrient solution was adjusted to pH 5.5and renewed once a week. Plants were grown in open air at 14/10 h (25°C/22°C) day/night regime. The experiment was conducted at September 2005 in botany department garden. 21-d-old plants were harvested and prepared for analysis.
NO Treatments and Chemicals
Sodium Nitroprusside (SNP) was used as NO donors in a 100 μM concentration (Graziano et al., 2002); 100 μM Methylene Blue (MB) was used as NO scavengers which inhibit NO production and/or action (Cragan, 1999). The solutions were supplied to plants by irrigation once a week included in the nutrient solution.
Biomass Allocation and RGI%
The biomass allocation was estimated by dividing shoot or root fresh weight by total plant fresh weight. RGI% = average dry mass of treated seedling/average dry mass of control seedlings X 100.
Zinc Content Determination
The leaves and roots samples were mineralized by wet open digestion in HNO3: H2SO4: HClO4. Total zinc concentration in the digest was estimated by atomic absorption spectroscopy.
Protein Content
Total protein content was estimated spectrophotometrically according to Lowery et al. (1951).
NO Concentration
Nitric oxide concentration was directly measured in both wheat and bean leaves using the method of Ridnour et al. (2000). Assay mixture consisted of 100 μL tissue extract mixed with equal volume of Griess reagent (1% sulfanilamide, 0.1% naphthalethylenediamine dihydrochloride). The solution was immediately mixed by inversion incubated at room temperature for 3 min and then A496nm was measured. NO concentration of the solution was calculated using an extinction coefficient of 6600 M-1cm-1.
Total Sulphydryl Groups Content
Leaves of wheat and bean seedling were analyzed for reactive sulfhydryl groups by a modification of the methods reported by Bulaj et al. (1998) using sulfhydryl-reactive reagent 5,5’- Dithio-bis-2-itrobenzoic acid (DTNB). Reactive sulfhydryl groups in 2.0 mL aliquots of sulfhydryl buffer solution were assayed after collection, without delay, via spectrophotometry for A412. Sulfhydryl concentrations were calculated from standard curves prepared from Cys standards made immediately before the assay.
Glutathione Content
Total reduced glutathione content was determined spectrophotometrically following the method described by Griffith (1980).
Glutathione Reductase (GR) Activity
GR activity was determined as the oxidation of NADPH at 340 nm (extinction coefficient 6.2 mM cm-1) according to Donahue et al. (1997). Leaf samples of both plants (1 g) were homogenized in phosphate buffer (pH 7.6), 2 mM EDTA. The homogenate was centrifuged at 15000 g for 10 min and the supernatant was used for analyses. The assay mixture contained 0.1 mM buffer (pH 7.6), 2 mM EDTA, 0.15 mM nicotinamioleaolenine- dinucleotide phosphate (NADPH), 0.5 mM glutathione oxidised (GSSG) and 500 μl of the extract. The reaction was initiated by addition of NADPH and followed for 5 min at 25°C.
Cu/Zn SOD Activity
Cu/Zn SOD was measured by the photochemical method as described by Giannopolitis and Ries (1977). One unit of Cu/Zn SOD activity was defined as the amount of enzyme required to cause 50% inhibition of the rate of ρ-nitro blue tetrazolium chloride reduction at 560 nm.
Statistical Analysis
Analysis of variance (ANOVA) and student t-test were performed on all data using SPSS program ver. 11.0.
Shoot and Root Biomass Allocation and RGI%
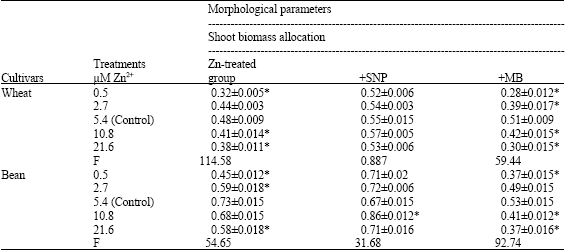
The results in Table 1 showed that zinc toxicity (10.8 and 21.6 μM) significantly decreased both shoot and root biomass allocation and RGI% in wheat and bean seedlings. SNP treatment significantly alleviated the adverse effect of deficient or toxic zinc concentration in both bean and wheat seedlings. On the other hand, application of MB (NO scavenger) increased the effect of zinc deficiency or toxicity by decreasing shoot and root biomass and RGI%.
The data of shoot and root biomass allocation and RGI% indicated that zinc concentration (10.8 and 21.6 μM) could be considered toxic to wheat and bean seedlings as they significantly reduced these parameters when compared to control concentration (5.4 μM).
Protein Content
The results in Figure (1a and b) showed that both zinc deficiency and toxicity induced significant decline in protein contents when compared to control (5.4 μM Zn concentration) in both plants under investigation. Application of SNP induced remarkable increase in protein content whereas treatments with MB significantly decreased protein contents at all zinc concentrations when compared to Zn-treated groups in both bean and wheat seedlings.
Zinc Concentration
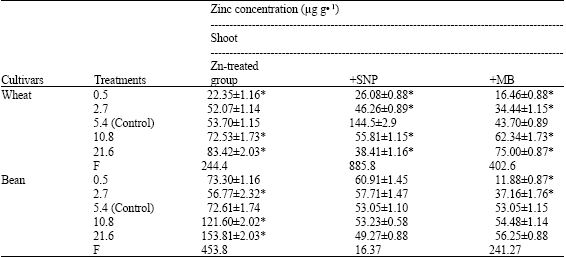
Zinc concentration in shoot and root extracts was increased with increasing zinc supply in irrigating nutrient solutions (Table 2). SNP application induced declines in zinc concentration at toxic zinc supply in shoot, root and total plant of wheat seedlings.
| Table 1: | Morphological parameters (shoot and root biomass allocation and RGI%) measured with fresh and dry weight of wheat and bean cultivars treated with different zinc concentrations alone or with either 100 μM Sodium Nitroprusside (SNP) as NO donor or 100 μM Methylene Blue (MB) as NO scavenger. Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Values are the means of three replicates±SE |
 | |
| Table 1: | Continued |
-2k7-59-75.gif) | |
| Table 1: | Continued |
-2k7-59-75.gif) | |
| *Significant differences as compared to control zinc concentration (5.4 μM) using student t-test (p<0.05) | |
| Table 2: | Zinc concentration expressed as μg g-1 dry weight, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Values are the means of three replicates±SE |
 | |
| Table 2: | Continued |
-2k7-59-75.gif) | |
| Table 2: | Continued |
-2k7-59-75.gif) | |
| *Significant differences as compared to control zinc concentration (5.4 μM) using student t-test (p<0.05) | |
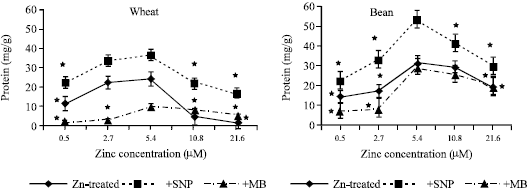 | |
| Fig. 1a and b: | Protein content expressed as μg g-1 fresh weight, in the leaf extract of wheat and bean cultivars treated with different zinc concentrations alone or with either 100 μM SNP (NO doner) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica±SE. *significant at p<0.01 when compared to control. *significant at p<0.01 when compared to control. *significant at p<0.01 when compared to control |
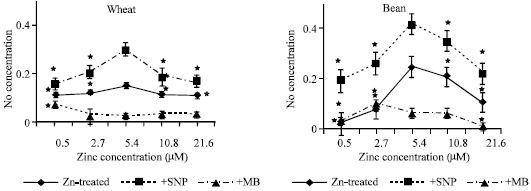 | |
| Fig. 2a and b: | Nitric oxide concentration expressed as μM/g fresh weight, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control |
In bean seedlings, SNP application at toxic zinc supply induced decline in zinc concentration in shoot and increase in zinc concentration in both root and total plant. On the other hand, MB application at zinc toxic supply induced increase in zinc concentration in shoot, root and total plant of both wheat and bean seedlings.
No Concentration
Application of SNP increased NO concentrations in both plant seedlings while MB induced a significant decline in NO concentrations (Fig. 2a and b).
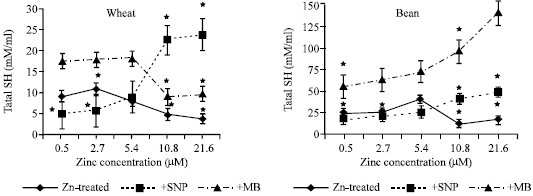 | |
| Fig. 3a and b: | Total SH expressed as mM/g fresh weight in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with e ither 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control |
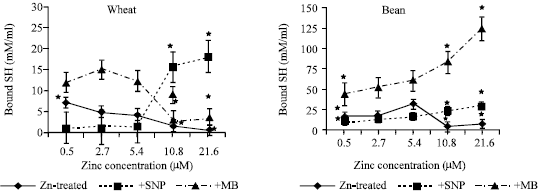 | |
| Fig. 4a and b: | Bound SH expressed as mM/g protein, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO doner) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control |
Total Sulphydryl Groups
The decrease in total SH (Fig. 3a and b) was observed at deficient zinc concentrations with SNP treated wheat and bean seedlings and the same zinc concentration significantly decreased free/total SH ratio (Fig. 5a and b). MB treated w heat seedlings induced a highly significant reduction of total SH with toxic zinc concentration. As zinc concentration increased, free/total SH ratio was increased in wheat seedlings treated with MB. It was also observed that bound SH exhibited the same trend as total SH (Fig. 4a and b). However, free/total SH ratio increased with zinc and MB application in wheat seedlings and declined at toxic zinc concentration in bean seedlings (Fig. 5a and b).
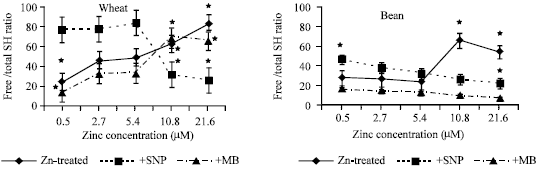 | |
| Fig. 5a and b: | Free/total SH ratio in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO doner) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control. *significant at p<0.01 when compared to control |
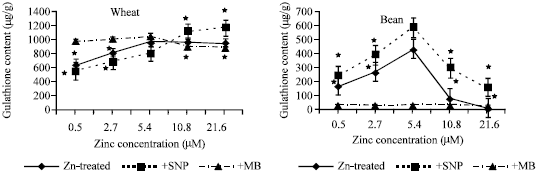 | |
| Fig. 6a and b: | Glutathione content expressed as μg g-1 fresh weight, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control |
Glutathione Content
In wheat seedlings (Fig. 6a), GSH content reached its lowest level at 0.5 Zn2+ supply then increased gradually with increasing Zn2+ concentration. SNP application (NO donor) decreased GSH content in Zn2+ insufficient concentration when compared to Zn-treated group seedlings while MB application significantly increased GSH content at (0.5, 2.7 and 5.4 μM) Zn2+ supplies. In bean seedlings,(Fig. 6b), SNP application increased GSH content at all zinc treatments when compared with Zn-treated group or MB treated seedlings.
Glutathione Reductase Activity
Different zinc concentrations had a non significant increase on GR activity in wheat seedlings (Fig. 7a) while the increase in GR activity was significantly recorded at toxic zinc concentrations in bean plants (Fig. 7b).
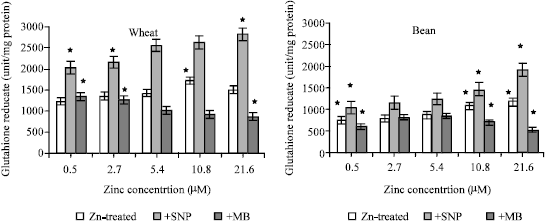 | |
| Fig. 7a and b: | Glutathione reductase activity expressed as unit/mg protein, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica ±SE. *significant at p<0.01 when compared to control |
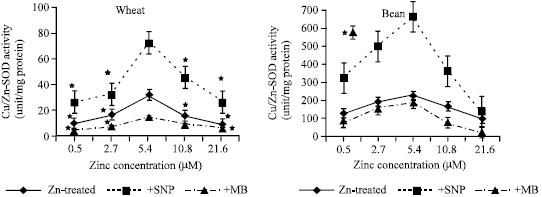 | |
| Fig. 8a and b: | SOD activity expressed as unit/mg protein, in the leaf extract of wheat and bean seedlings treated with different zinc concentrations alone or with either 100 μM SNP (NO donor) or 100 μM MB (NO scavenger). Zinc was supplied in different concentrations (0.5, 2.7, 5.4 (Control), 10.8 and 21.6 μM) as ZnSO4. SNP and MB solutions were supplied to plants by irrigation once a week included in the nutrient solution. Each point corresponds to the mean of two to four independent experiments. Data are the mean of three replica±SE. *significant at p<0.01 when compared to control |
On the other hand, SNP application induced a highly significant increase in GR activity at all zinc concentrations in wheat and bean seedlings Application of MB (NO scavenger) decreased GR activity with increasing zinc concentration. At toxic concentration of zinc (21.6 μM), GR activity declined with 19.4 and 38.1% at wheat and bean seedlings respectively when compared to that of control zinc concentration.
Cu/Zn SOD Activity
The results in Figure (8 a and b) indicated that Cu/Zn SOD activity significantly decreased at deficient and toxic zinc concentrations in both wheat and bean seedlings when compared to control (5.4 μM Zn) concentration. The decline in endogenous NO concentration (MB application) reduced Cu/Zn SOD activity at all zinc concentration as compared with Zn-treated seedlings. On the other hand, application of exogenous NO (SNP treatment) enhanced Cu/Zn SOD activity at all zinc concentrations when compared to Zn-treated or MB treated seedlings.
Under physiological conditions, the majority of Zn+2 within cells is complexed by proteins. To date, MT is the only protein that has been implicated in cellular Zn+2 storage and usually a significant percentage of the total cellular zinc is complexed by MT (Kägi and Schaffer, 1988). Several functions have been designated to MT (Palmiter, 1998). It has been shown to protect cells against oxidative damage and to exert chaperone-like activities donating Zn+2 to target apometalloproteins, e.g., zinc finger transcription factors. In addition, MT has been suggested to play a central role in heavy metal metabolism and detoxification and in the management of various forms of stresses (Davis and Cousins, 2000 and Maret, 2000).
Morphological results (Table 1) revealed that SNP application (NO donor) alleviated the adverse effect of low or high zinc concentrations by increasing shoot and root biomass allocation and RGI%. NO effect was the same in both wheat and bean seedlings. Growth inhibition is a general phenomenon associated with zinc toxicity (Collins, 1981). Zinc-deficiency stress usually involves peroxidative damage of crucial components of biomembranes, enzymes and thylakoids (Bray and Bettger, 1990) which are the major targets of toxic O2 radicals (Elstner, 1982). Metallothioneins (MTs), a cystein-rich (30% of all amino acid residues), heavy-metal-binding protein, is critical to intracellular Zn2+ homeostasis, as it has the ability to bind up to seven zinc atoms per MT molecule (Kägi and Schäffer, 1988). Due to the different metal affinities for zinc and cadmium in the two separate domains (Nettesheim et al.,1985), the β-domain has been implicated in zinc homeostasis and the tight binding of cadmium in the α-domain was proposed to be responsible for the role of MTs in heavy metal detoxification. In addition, it has been reported that MTs act as radical scavengers under oxidative stress (Sato and Bremner, 1993, Maret, 1995). Another possible key player in the role of MTs in signal transduction might be Nitric Oxide (NO), which was shown recently, both in vitro (Misra et al., 1996) and in vivo (Schwarz et al., 1995; Aravindakumar et al., 1999), to interact with MTs and thereby release bound zinc and cadmium. The importance of MTs in NO-induced changes in intracellular zinc homeostasis has been reported by St Croix et al. (2002). Under aerobic conditions, NO via thiolate-nitrosation induces the release of Zn2+ from the zinc-storing protein Metallothionein (MT) in vitro (Zangger et al., 2001). By using the fluorescent resonance energy transfer technique and cells transfected with MT fused to green fluorescent protein, NO was found to induce an intracellular conformational change of MT, suggesting intracellular Zn2+ release from this protein (Pearce et al., 2000). In addition, exogenously added NO has been shown to induce S-nitrosation of intracellular MT (Liu et al., 2001; Fabisiak et al., 2002). Moreover, nitrosative stress induces Zn2+ release in various types of cells predominantly within the cytoplasm (Tartler et al., 2000; St Croix et al., 2002).
Application of MB (NO) scavenger) proved that NO had a significant effect on increasing protein concentration at all zinc supplies (Fig. 1a and b). The data in Table (2) showed that SNP application increased total zinc concentration at zinc deficiency in wheat seedlings. This increase was concomitant with a highly increase in free SH and a highly decrease in total SH (Fig. 3a). These changes in free or total SH were also observed when MB was applied. Using 1H- and 113Cd-NMR spectroscopy, Zangger et al. (2001) have found that the exposure of MTs to NO leads to a selective release of all three metals from the N-terminal α-domain while leaving the four metals in the α-domain untouched. This finding indicates a possible regulatory role of NO in the specific redistribution of metal from MT. Specifically, the postulated function of Mts in heavy metal detoxification, which has been attributed to the α-domain, appears to remain unmodified by No. The results in Table (2) revealed that SNP decreased total zinc concentration at toxic zinc concentration in both wheat and bean seedlings. The NO effect was detectable in both seedlings shoots. It was also observed that the MB application (NO scavenger) reverse SNP effect at toxic zinc concentrations. Moreover, decreasing endogenous NO with MB application induced significant decrease of free/total SH ratio and increase in total SH at toxic zinc concentrations.
By binding sulfhydryl groups and phospholipids, Zn2+ stabilizes and protects cell membranes and enzyme proteins against oxidative attack of toxic O2 species (Willson, 1989; Bray and Bettger, 1990). Ernst et al. (1992) elaborated details on mechanism of heavy metal toxicity in plants at the cellular and organism level. He also reported that the plasma membrane was the cell compartment which regulated metal entry into the cell, in addition its proteins, especially the SH groups might be affected in their activity causing damage to membrane stability due to zinc toxicity. Cumming and Taylor (1990) described the signal transduction and acclimation mechanisms on metal tolerance in plants. They also indicated that the mechanism of metal tolerance were due to exclusion and external metal detoxification mechanism, internal metal detoxification mechanisms and multiple mechanisms and co-tolerances. The production of metal-chelating ligands high in thiol groups might also render co-tolerance to Zn2+.
SNP application (NO donor) increased total SH and decreased free/total SH ratio in wheat (Fig. 3a and 4a) at toxic zinc concentrations and this result was supported by MB application (decreasing endogenous NO). Moreover, the same NO effect was observed at all zinc concentrations in bean seedlings and this effect was reversed by MB application (Fig. 4a and 4b). Free cysteine with its free amino-, carboxyl- and thiol-groups is an effective chelating agent for metal ions with which it forms five- or six-membered ring structures (Taylor et al., 1966). In the presence of H2O2 transition metals reduced in this way can undergo a Fenton reaction whereby highly toxic hydroxyl radicals are formed (McAuliffe and Murray, 1972). Thus, homeostatic control of low cysteine levels becomes important for conferring resistance to oxidants. The intracellular concentration of cysteine needs to be maintained in the lower micro-molar range. Due to the resulting diminished ability to reduce transition metal ions, a cell can maintain millimolar concentrations of GSH and thus a concentration 10-50-fold higher than concentrations of free cysteine without triggering deleterious Fenton reactions. It could be suggested that NO generated by SNP mediated Zn release via nitrosylation of cysteine thiol groups could explain the increase in free/total SH ratio and the decrease in total and bound SH at zinc deficient condition. Oxidation of cysteine-SH by H2O2 leads to the sulfenic acid (Cys-SOH). While this first step of oxidation is reversible and can be explored for redox signaling purposes, further oxidation to cysteine sulfinic acid (Cys-SO2H) and cysteine sulfonic acid (Cys-SO3H) is irreversible and renders the protein labile to oxidative stress (Poole et al., 2004). The sulfhydryl groups of cysteine sulfinic acids can undergo reactions with other sulfhydryl groups to form disulfides or reactions with glutathione to form mixed disulfides. Concomitant changes in protein structure and function can lead to changes in metabolism and form the basis of cellular signaling events.
The data in Figure 6a and b showed that SNP application induced a highly significant increase in GSH concentration at all zinc concentration in bean seedlings while the increase was only observed in toxic zinc supply in wheat seedlings. The change in GSH concentration was correlated with GR activity in wheat plants at all zinc zupplies and in bean plant at deficient and normal zinc supplies only (Fig. 7a and b). Transition metals such as Zinc (Zn) are essential micronutrients for many physiological processes, but they become toxic at elevated levels. Zinc is one of the most abundant trace heavy metals present in agro-ecosystems. Di Baccio et al. (2005) found that total glutathione (GSH+GSSG) content was reduced with increasing Zn concentration, while the contribution of oxidized to total glutathione increased in Populus spp. The major low-molecular mass thiol compound in plants and animals is the tripeptide L-γ-glutamyl-L-cysteinyl-glycine also known as glutathione (GSH in reduced form and GSSG in its oxidized form). Glutathione has also been described as a ‘transport peptide’ in vivo for NO through the formation of S-NO groups (Jourd’heuil et al., 2000). The glutathione redox couple, a cellular redox buffer which maintains the given thiol/disulfide redox potential, has already been implicated in modulating the metal release from metallothionein in the absence of nitric oxide by Vallee and Maret (1993) and coworkers (Maret, 1995; Jiang et al., 1998; Jacob et al., 1998). These authors reported increased metal release in the presence of oxidized glutathione (GSSG) and even slightly tighter metal binding under the influence of reduced glutathione (GSH) (Jiang et al., 1998). Khatai et al. (2004) showed that metal release from MT2 by nitric oxide and peroxyinitrite suppressed by reduced but not oxidized glutathione.
Thus, in MT the protein plays a role in the biological function of zinc, a paradigm quite different from that in most other zinc proteins where zinc plays a role in the biological function of the protein. The tight binding of zinc to MT raises questions of how it is released and whether or not the release is controlled. In this regard, we have identified glutathione disulfide (GSSG) as a cellular ligand that reacts with MT and mobilizes zinc, resulting in the suggestion that the zinc content of MT is linked to the redox state of glutathione in the cell in such a manner that zinc remains bound to MT as long as high thiol reducing power prevails and is released once the redox balance becomes more oxidizing. In terms of molecular mechanism, GSH and GSSG could control the amount of free zinc available once it is released from MT, or bind to MT and affect its conformation and zinc binding. GSH binding would protect MT from the loss of Zn-2, thereby inhibiting its transfer, while providing a reactive thiol for the reaction with GSSG and resulting in zinc transfer through a process that is strictly proportional to and dependent on the concentration of GSSG. Hence, a system consisting of MT-GSH-GSSG provides a mechanism to control zinc transfer among a large variety of acceptors. SNP as (NO donor) increased SOD activities at all zinc concentrations when compared to control (Fig. 8a and b). This finding is supported with that of Zhou et al. (2005) who stated that NO donor treatment enhanced the activities of SOD, CAT and APX. Increasing SOD activity resulted in H2O2 production which may oxidize SH to form disulfide bond. The increase in GSH content in bean seedlings means that NO activate ascorbate-glutathione cycle which utilize the produced H2O2 and subsequently reduced SH oxidation. Moreover, increasing GSH at toxic zinc concentration suppresses zinc release from MT and this explains the reduced levels of zinc when SNP was applied. Jiang et al. (1998) have identified glutathione disulfide (GSSG) as a cellular ligand that reacts with MT and mobilizes zinc, resulting in the suggestion that the zinc content of MT is linked to the redox state of glutathione in the cell in such a manner that zinc remains bound to MT as long as high thiol reducing power prevails and is released once the redox balance becomes more oxidizing (Maret, 1994).
It could be concluded that nitric oxide plays a significant role in zinc homeostasis in wheat and bean seedlings subjected to different zinc concentration. The effect of nitric oxide was supported by using (MB) which scavenged the endogenous nitric oxide and reversed the action of NO in maintaining suitable zinc concentration for seedlings growth. At zinc deficient supplies (0.5, 2.7 μM) nitric oxide (as SNP application) decreased free/total SH and this mean that zinc is released from MT-rich-cysteine which chelate zinc. Moreover, GSH content was lower than that of Zn-treated seedlings. This decrease may be due to utilization of glutathione in detoxification of H2O2 at ascorbate-glutathione cycle or it may conjugate with free SH to suppress Fenton reaction. On the other hand, at toxic zinc supplies, the effect of NO was reversed (increasing total SH and GSH). The increase in GSH content suppresses metal release from MT and this is supported by the increase in total SH and decrease in free/total SH. The effect of nitric oxide was supported by the application of MB which reduces the endogenous nitric oxide.