Research Article
Purification and Biochemical Characterization of Acid Phosphatase from Vigna aconitifolia
Department of Chemistry, College of Science, King Faisal University, Saudi Arabia
Acid phosphatases (EC 3.1.3.2) produced by both prokaryotic and eukaryotic cells and are presumed to convert organic phosphorus into available Pi (Ehsanpour and Amini, 2003; Amlabu et al., 2009). Phosphate is an important molecule for cellular growth that involved in many different biological reactions (Schachtman et al., 1998). The hydrolysis of phosphomonoesters by phosphatases in biological systems is an important process. This process is linked to energy metabolism, metabolic regulation and a wide variety of cellular signal transduction pathways (Allan et al., 1994). The role of acid phosphatase in phosphorus metabolism has been extensively studied in prokaryotic and eukaryotic systems (Duff et al., 1994). The physiological function of acid phosphatase is to provide inorganic phosphate for the cellular growing (Carswell et al., 1997).
The liberation of phosphate from phosphate ester is mainly affected by phosphatase (Bewley and Black, 1994). Deficiency of phosphorus in plants leads to metabolic disorders such as a decrease in photosynthesis, respiration and retardation of growth (Garcia-Sánchez et al., 1996). Phosphatases have been traditionally classified as being alkaline or Acid Phosphatase (AP) according to their optimum pH for catalytic activity above or below pH 7.0 (Barret-Lannard et al., 1982; Shahbazkia et al., 2009). Several interacellular acid phosphatases have been suggested to have a role in the hydrolysis of interacellular polyphosphates (Huber and Hamel, 1985).
Acid phosphatase was found to be localized in the cell walls of Pisum sativum (Olmos and Hellin, 1997), root cap of rice (Chen et al., 1992), phloem of Nicotiana tabacum (Bentwood and Cronshaw, 1975) and xylem of Phaseolus vulgaris (Charvat and Easu, 1974). The mechanism which regulates acid phosphatase distribution and activity is unclear, although their abundance may be regulated by the level of phosphate in the environment and their activity is clearly influenced by local polyelectrolytes (Tu et al., 1990). Acid phosphatase expresses its isozymes in many plants such as soybean seeds (Ferreira et al., 1998); Vigna sinensis (Biswas and Cundiff, 1991), tea leaves (Baker and Tadakazu, 1973) and lentil seeds (Bose and Taneja, 1998).
In the present investigation, we purified and characterized the AP from the shoot of Vigna aconitifolia as a step towards understanding its properties. The optimum pH and temperature were determined. Moreover, the catalytic active sites were characterized to get into the AP structural functional relationship.
Chemicals
All the chemicals obtained from Sigma and BDH chemicals LTD and stored as directed by supplier.
Plant Materials
The experimental plant used in this investigation was Vigna aconitifolia. Pure strain of seeds was used.
Seed Germination
Seeds of Vigna aconitifolia were sterilized with 0.1% sodium hypochlorite and soaked overnight in sterile water at room temperature. The wet seeds were germinated on filter papers moistened with distilled water in petri dishes until complete germination. The germinated seeds were sown in plastic pots containing 1 kg of oven dry soil. Then they were placed in a growth chamber maintained at 27/20°C light/dark (12 h) and grown for 10 days. At the end of the experiment, the shoot systems were washed with deionized water and blotted on paper towels.
Preparation of the Crude Extract of AP
Ten gram of collected shoots from 10 days old seedlings of Vigna aconitifolia were homogenized in a blender with 30 mL of 100 mM Tris-HCl (pH 5.0) containing 2 mM MgCl2 and 2% polyvinylpyrolidone (PVP). The homogenate was filtered through four layers of muslin and centrifuged at 5000 rpm for 20 min. The supernatant was filtered through a layer of absorbent cotton to get rid of fatty layer. The filtrate was used for AP purification.
Purification of AP
Ammionium sulphate were added and dissolved in a 30 mL of the crude extract, prepared as described above, to make a final concentration of 30% in the mixture. The extract was left for 1 h in a refrigerator at 4°C and then centrifuged at 10,000 rpm for 20 min in a refrigerated centrifuge (MLW WEB Medizintechnik, Leipzing GDR) at 4°C, to collect the precipitated proteins. To a 30 mL of the resulted supernatant ammonium sulphate were added in order to make a final concentration of 70%. Proteins having catalytic activity were precipitated and left to stand at 4°C for 2 h. The precipitated protein was collected by centrifugation at 10,000 rpm for 30 min at 4°C. The precipitated proteins were dissolved in 100 mM phosphate buffer (pH 5.4) and the catalytic activity was determined. The protein solution containing AP activity was applied to column (2.5x10) containing DEAE-cellulose. The enzyme was eluted by 100 mM phosphate buffer (pH 5.4) containing gradient 0.1- 0.5 M NaCl. The rate of elution was 30 mL h-1 at 4°C and 3 mL fractions were collected. The fractions with maximum AP activity were combined and applied to Sephadex G200 column (2x20 cm). The enzyme was eluted by 200 mM phosphate buffer (pH 5.4) containing gradient 0.1-0.4 M NaCl. The rate of elution was 20 mL h-1 at 4°C and 2 mL fractions were collected. The fractions with maximum activity were combined and used for subsequent kinetic studies.
Acid Phosphatase Assay
The assay method of Park and van Etten (1986) for AP was modified in the present study. The AP reaction buffer was consisted of 100 mM sodium acetate (pH 5.4), 10 mM MgCl2, 6 mM p-NPP as the substrate and protein extract. Assays were initiated by addition of substrate. The reaction was terminated by the addition of developing reagent. This reagent was prepared daily and consisted of 4 volumes freshly made 10% (w/v) ascorbic acid to 1 volume of 10 mM ammonium molybdate in 15 mM Zn-acetate (pH 5.0) solution. After addition of developing reagent, the samples were incubated for 30 min at 30°C and the A630. To calculate activities, a standard curve in the range of 1- 150 nmol Pi was constructed.
Protein Determination
Protein concentration was measured throughout the purification steps by the method of Lowry et al. (1951).
Table 1 shows that the protein content decreased progressively throughout the different steps of purification. It should be noticed that the enzyme activity in the crude extract was small and then increased gradually throughout the purification process. The final specific activity was 280 units mg-1 protein. At this final step of purification the enzyme activity was 60 fold purified. The partially purified enzyme from DEAE cellulose was applied to the column of Sephadex G200. The fractions were collected and the enzyme activity was measured in each fraction. The results are demonstrated in Fig. 1. It seems from the results that fraction no.6 contained the highest activity of AP, where the sharp peak appeared.
| Table 1: | Purification of AP from Vigna aconitifolia |
 | |
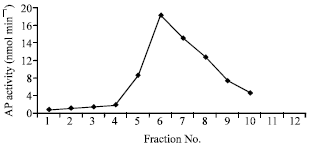 | |
| Fig. 1: | Elution profile for AP from sephadex G-200 chromatography |
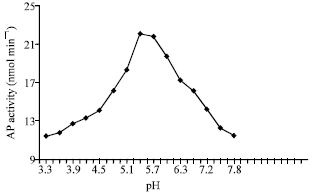 | |
| Fig. 2: | Effect of pH on AP activity |
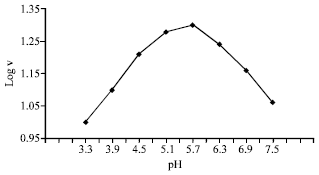 | |
| Fig. 3: | Determination of the apparent dissociation contant of the groups taking partin catalytic activity of AP |
On the study of pH effect on the enzyme activity, it was necessary to fix the other factors affecting the enzyme activity such as enzyme concentration, substrate concentration, MgCl2 concentration, as well as the temperature under which the reaction mixture was incubated. The results are illustrated graphically in Fig. 2. It appears from the figure that the enzyme activity increased gradually from pH 3.6 to 5.4 which seems to be the optimum value after which the enzyme activity started to decline. Plotting log V against pH as shown in Fig. 3, that there are two pKa values at 5.4 and 5.7 implicating the presence of histidyl and cysteine sulfhydryl groups (Price and Stevens, 1982).
It shows a considerable linear relationship was obtained at lower p-NPP concentrations where, a little increase at higher concentrations can be observed. Plotting the reciprocal of the reaction rate (V-1) against the reciprocal of p-NPP concentration (S-1) gave a straight line (Fig. 4). According to Lineweaver and Burk hypothesis, both Vmax and Km for AP were calculated. The values of the parameters values were 27.8 nmol min-1 and 133 mM. Plotting logS against logVo/Vmax-Vo gave straight line (Fig. 5) which is known as Hill coefficient (h) whose value was 2. The Hill coefficient (h) is equal to the binding sites.
The effect of other phosphate compounds, e.g., fructose-6-phosphate and sodium phytate, on the enzyme activity were also studied (Table 2). It is apparent from the results that p-nitrophenyl phosphate is the best substrate for AP. The activity of AP enzyme with fructose-6-phosphate was 44.3% of that detected with glucose while, sodium phytate expressed 67.1% of that measured with p-nitrophenyl phosphate as a substrate.
AP activity was measured at different periods of incubation (10-60 min). The results of these measurements are shown in Fig. 6. These results clearly show that the enzyme activity increased slowly from 10 up to 30 min at which highest enzyme activity was recorded and after which the activity declined gradually.
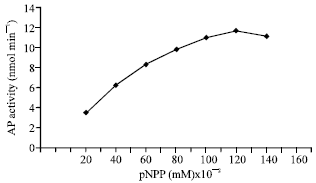 | |
| Fig. 4: | Effect of pNPP concentration on AP activity |
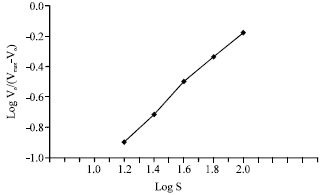 | |
| Fig. 5: | Hill plot for AP |
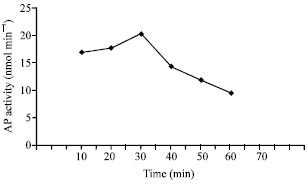 | |
| Fig. 6: | Effect of incubation time on AP activity |
| Table 2: | Effect of different substrates on AP activity |
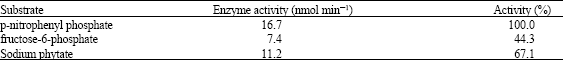 | |
Therefore, an incubation period of 30 min was chosen for AP mixtures throughout the present study.
The activity of AP was studied in a temperature range of 10-80°C. The results are illustrated in Fig. 7. The optimal temperature for AP was 30°C. Any further increase after the optimal temperature leads to an noticeable reduction in the activity. There is an apparent linearity between the enzyme activity and the temperature in the range 10-80°C and the highest enzyme activity is recorded at 30°C, which seems to be the optimum.
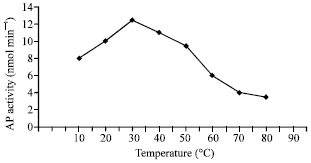 | |
| Fig. 7: | Effect of temperature on AP activity |
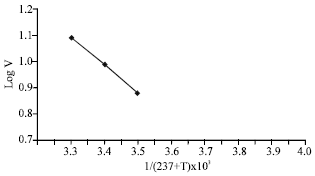 | |
| Fig. 8: | Arrhenius plot for AP |
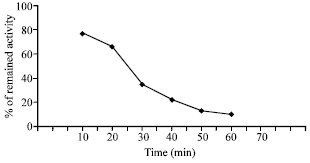 | |
| Fig. 9: | Rate of heat inactivation of AP |
After 30°C the activity declined gradually. When the logarithm of the reaction rate is plotted versus the reciprocal of the absolute temperature a straight line was obtained (Fig. 8) and the activation energy was calculated and its value was 0.44 kJ mol-1.
The rate of heat inactivation of AP activity was studied by plotting the percentage of the remaining activity against time at 50°C. The results are illustrated in Fig. 9. It could be considered that AP from Vigna aconitifolia was labile above 30°C and its activity declined after 10 min since the remaining activity was 76% which decreased to 66.3% after 20 min. After 50 min the enzyme activity was approximately abolished.
The enzyme activity was measured at different concentrations of the purified AP (Fig. 10) in volumes ranging from 0.1 to 0.7 mL of the enzyme solution.
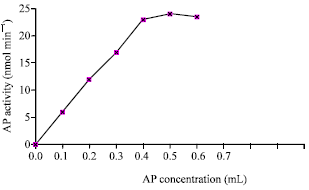 | |
| Fig. 10: | Effect of AP concentration on the reaction activity |
The results indicated that by increasing the enzyme concentration, there was a corresponding increase in the rate of reaction and a linear relationship was obtained.
The technique adopted for acid phosphatase purification from the Vigna aconitifolia shoot included precipitation of the crude extract by ammonium sulphate and using DEAE-cellulose and Sephadex G200 columns. The purification technique of acid phosphatase showed that the specific activity was 280 U mg-1 protein. This value is higher than that reported for AP from Stichopus japonicus and Trichoderma harzianum (Zhu et al., 2009; Leitao et al., 2010) and is similar to that reported for AP from tobacco cells (Okuda et al., 1987). However, this value was higher than those reported for the acid phosphatase from seeds of sunflower, maize endosperm cultures, seeds of Vigna sinensis (Park and van Etten, 1986; Miernyk, 1992; Tapan et al., 1996).
The optimum pH of the purified acid phosphatase was 5.4. This value is consistent with that reported for maize endosperm (Miernyk, 1992) and is higher than pH 5 reported for acid phosphatase from germinating seeds of Vigna sinensis (Biswas and Cundiff, 1991). Moreover, the obtained pH 5.4 for acid phosphatase from Vigna aconitifolia shoot is lower than the values obtained for the enzyme from lupin (Lupinus albus) seeds (Zheng and Duranti, 1995; Tapan et al., 1996); sunflower seeds (Park and van Etten, 1986); roots of Trifolium (Julie et al., 1999; Zhang and McManus, 2000) and cotton seedlings (Bhargava and Sacher, 1987).
Generally, enzymes are sensitive to changes in pH. The pH can influence the enzyme activity in a number of ways. Firstly, it can change the ionization of the enzyme substrate complex. Secondly, it can change the ionization of various groups of the enzyme molecule which may affect the affinity of the enzyme for the substrate. Thirdly, it changes the ionization of the substrate which may affect the binding of the substrate to the enzyme. Fourthly, at extreme pH it can brings about changes in protein structure (Palmer, 1985).
The present results showed that acid phosphatase has a broad substrate specificity with highest activities with p-nitrophenyl phosphate and fructose-6-phosphate while sodium phytate showed a little activities compared to p-nitrophenyl phosphate. These results are in harmony with the findings of other workers (Penheiter et al., 1997; Wannet et al., 2000). Plotting V-1 versus S-1 gave a linear relationship from which Km value for acid phosphatase was calculated. The obtained value of Km of acid phosphatase for p-nitrophenyl phosphate was 133 mM which is higher than 35 mM reported for the enzyme from maize endosperm (Miernyk, 1992).
The optimal incubation time for acid phosphatase was 30 min. Similar incubation time (30 min) was reported for acid phosphatase from tobacco cells (Okuda et al., 1987).
The optimal temperature of acid phosphatase was 30°C. A higher value was 37°C which was reported for the enzyme from cotton seedlings (Bhargava and Sachar, 1987).
Plotting 1/T+273 against log V resulted in a straight line in case of acid phosphatase which is in agreement with Arrhenius plot (Palmer, 1985). From this relationship the activation energy was calculated. The value of activation energy for acid phosphatase was 9.44 kJ mol-1.
In addition to the enzyme and substrate, many enzymes require, a non-protein substances for the catalytic reaction to proceed. These substances are known as cofactors and are usually known as activators, which probably affect the conformation of the enzyme producing a more catalytically active state, or interact with the substrate.
In conclusion, the characterized acid phosphatase from the shoot of Vigna aconitifolia would be useful for adapting organic phosphorus into available Pi that involved in many different biological reactions.