Research Article
Effect of Chitosan and Green Tea on the Quality of Washington Navel Orange During Cold Storage
Department of fruit Handling, Horticulture Research Institute, Agricultural Research Center, Giza, Egypt
Navel orange (Citrus sinensis, L. (Osbeck) is the most popular fruit crops among other citrus species in Egypt that represents near 65-70% of the total orange cultivated area. Citrus fruits suffer from relatively high loss during the harvesting and handling chain. Low temperature during storage and transport are used to extend postharvest life and for the purpose of insect disinfection, marketing fruits more prone to chilling injury. Development of postharvest rind disorders causes severe economic losses to the entire citrus industry worldwide. Chilling injury that occasionally develops in Navel orange following the cold disinfestations treatment required to export the fruit to fly-free zones where quarantine regulations operate.
Chitosan (poly-β-(1, 4)-D-glucosamine), is a natural carbohydrate polymer compound derived from chitin which has been found in a wide range of algae and is used in medical or industrial products as a bioactive material (Cho et al., 2008). It can be obtained from arthropod exoskeletons and crustacean shells (crabs and shrimp) either by chemical or microbiological processes. This antimicrobial biopolymer can also be obtained directly from the cell wall of some plant-pathogenic fungi (Devlieghere et al., 2004).
Chitosan is considered as an ideal preservative coating for fresh fruits and vegetables due to its film-forming and biochemical properties. It acts in three ways to enhance the storage life of fresh fruits and vegetables; it acts as an antifungal agent to control the postharvest fungal disease, forms a semi-permeable coating around plant tissues which modifies the internal atmosphere of the fruit and decreases the loss of moisture due to transpiration and induces defense mechanisms which delay ripening and lower the rate of respiration (Zhang and Quantick, 1997). Chitosan can be used for prolonging the storage life and preventing loss of the products after harvest.
Zhang et al. (2011) detected that chitosan maintained postharvest quality and beneficially influenced firmness, total soluble solid content, titratable acidity, ascorbic acid content and water content of citrus fruit after 56 days of storage. Also, it decreases the respiration rates, inhibits fungal development and delays ripening because of a reduction in the evolution of ethylene and carbon dioxide.
The most feasible hypothesis is a change in cell permeability due to interactions between the positively charged chitosan molecules and the negatively charged microbial cell membranes. This interaction leads to the leakage of proteinaceous and other intracellular constituents.
In general, it is known that the mode of chitosan action on phytopathogens fungi could develop in an extra level (plasma membrane) and intracellular level (penetration of chitosan on fungal cell) (Guo et al., 2008).
Green tea (Camellia sinensis), is a good source of polyphenolic compounds having strong antioxidant property which, proved to exhibit antimicrobial activity against some bacteria (Chan et al., 2007). The important polyphenolic compounds in tea leaves include catechin, theaflavins and thearubigins.
Green tea is proved to exhibit antimicrobial activity against some bacteria and has good antioxidant activity. The beneficial effects of the phenolic compounds are thought to result from their ability to scavenge reactive oxygen and nitrogen species (Gramza et al., 2006).
The purpose of this study is to study the effect of an environmentally antifungal active from chitosan coatings and or green tea extract as a natural antioxidant against the decay to extend fresh quality of Washington Navel orange and the ability of fruits to low cold stress as the optimum quarantine during cold storage.
Orange fruits were picked from 9 years old Washington Navel orange trees budded on Sour orange (Citrus aurantium) rootstock grown in a private orchard at Aga city, Dakahlia, Governorate. At both seasons, fruits were harvested at the first week of January, fruit chosen for this study was uniform in size, color and weight. They were picked early in the morning when yellow color covered greater of the fruit surface and the Soild Soluble Contents (SSC) percentage of 8 or higher (Kader, 1992).
Selected fruits were directly transported to the laboratory of post-harvest at Horticulture research Institute Mansoura, Egypt. Defective fruits were almost equal in size and apparently insect and pathogen injury free. All fruits were washed with tap water to remove the dust and foreign materials and then air-dried and a quick sorting was done to research fruit for any defects. At the beginning of the experiment, samples of 15 fruits were taken to determine the initial fruits properties and then received the following treatments:
| • | Dipping fruits in chitosan 2.0% for 5 min |
| • | Dipping fruits in green tea 2.0% for 5 min |
| • | Dipping fruits in chitosan 2.0% + green tea 2.0% for 5 min |
| • | Control (dipping fruits in tap water) for 5 min |
Chitosan crude stock solution was prepared by dissolving 2 g of chitosan powder in 100 mL of 2% acetic acid solution (2:98, v/v acetic acid: distilled water) according to Bautista-Banos et al. (2006).
Green tea water extract solution was prepared by mixing ground tea powder in distilled water (1:5 w/w) controlled at 90°C in an Erlenmeyer flask and stirred in a water bath shaking incubator (SW 23, Julabo Labortechnik GmbH, Seelbach, Germany) at 100 oscillation/min for 10 min (Siripatrawan and Harte, 2010). The water extract of green tea was filtered through Whatman No.1 filter paper.
The fruits were left to dry and then packed in one layer inside ventilated carton boxes each consists of 15 fruits (each box contains 4-5 kg). Nine boxes served for each treatment. The total number of boxes were 36 for all treatments after that stored for 60 days at 0±2°C with 90-95% R.H and then 3 carton boxes for each treatment were taken at 20 days intervals to determine the following fruits characteristics.
Loss in fruit weight: It was determined according to the following equation:
![]()
Decay: It was determined according to the following equation:
![]()
Chilling Injury (Cl): Fruit were evaluated for chilling injury severity according to the following scale: 0 = Normal (no pitting), 1 = Slight (a few scattered pits), 2 = Moderate (pitting covering up to 30% of the fruit surface) and 3 = Severe (extensive pitting covering >30% of the fruit surface). The chilling injury index was determined for each treatment by multiplying the number of fruit in each category by their score and then dividing this sum by the total number of fruit assessed (Ghasemnezhad et al., 2008).
Skin hue color (h°): Skin color was measured using a hand-held colorimeter (CR-10; Minolta Co., Ltd., Osaka, Japan) and Spectra-Match software, set to L*, a*, b* mode. The colorimeter has a beam diameter of 8 mm, three response detectors set at 0 viewing angle and a CIE standard illuminant C with diffuse illumination. This illuminant is accepted as having a spectral radiant power distribution closest to reflected diffuse daylight. Color changes were documented over the duration of the experiment. L* values indicate lightness (black [L* = 0] and white [L* = 100]), a* values indicate redness-greenness (red [a* = 100] and green [a*= {-100}]) b* values indicate yellowness-blueness (yellow [b* = 100] and blue [b* = {-100}]). A lower hue value indicates a redder product. The L*, a* and b* and h° values obtained from each fruit at the time of sampling represent average L*, a*, b* and h° values calculated from three separate light pulses from the colorimeter. The light pulses were timed to allow movement of the colorimeter to 3 locations on each fruit.
Color changes from green to yellow were indicated by calculating the hue angle (h°), from (a*, b*) using the methods described by McGuire (1992) as the following equation:
![]()
where, a is interval of colors between green (-) and red (+), b is interval of colors between blue (+) and yellow (-), h° is skin hue color
Juice weight%: It was obtained from the following equation:
![]()
Soluble Solids Content (SSC%): Soluble solids content in fruit juice was measured using a Carl-Zeiss hand refractometer according to AOAC (2005).
Titratable Acidity (TA%): It was determined in10 mL of fruit juice as a percentage of citric acid according to AOAC (2005).
SSC/acid ratio: It was calculated by dividing the value of SSC over the value of titratable acidity of each sample.
Ascorbic acid (mg/100 g fresh weight): Ascorbic acid (Vitamin C) was measured by the oxidation of ascorbic acid with 2, 6-dichlorophenol endophenol dye and the results were expressed as mg/100 g fresh weight according to Ranganna (1979).
Ascorbic Acid Oxidase (AAO) (U/mg protein/min): The oxidation of ascorbic acid was measured by changes in optical density at 265 nm in 30 sec. intervals for 5 min. Protein content of the extracts was determined according to Bradford (1976) using bovine albumin serum as a standard.
Peroxidase (POX) U/mg protein/min: The activity of POX was expressed as optical density per milligram of protein per minute. One unit of POX activity was taken as the changes of 1.0 unit of optical density per minute according to Urbanek et al. (1991).
Statistical analysis: Data of both seasons was analyzed using analysis of variance (ANOVA) technique. Differences among treatment means were statistically compared using the Least Significant Differences test (LSD) at p = 0.05 using the CoStat v6.4 program.
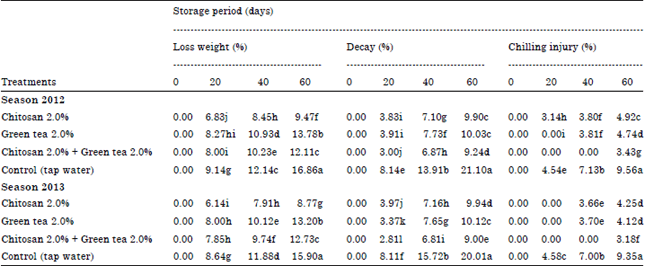
Loss in fruit weight percentage: Table 1 show that all dipping treatments with chitosan and/or green tea significantly reduced fruit weight loss than the control during both seasons under the study. However, control fruits lost 16.86 and 15.90% of their weight in the two seasons, respectively. The most effective treatment in both seasons were that of chitosan at 2% which recorded fewer weight losses values 9.47 and 8.77% , respectively than the other treatments used or the control.
| Table 1: | Effect of Post-harvest treatments on weight loss%, decay% and chilling injury% of Washington Navel orange fruits during cold storage 2012 and 2013 seasons |
 | |
| Means followed by the same letters are not significantly different by Duncan multiple range test at 0.05 levels | |
Decay percentage: Data in Table 1 reveals that all dipping treatments with chitosan and/or green tea significantly reduced percentage of decayed fruits than the control during both seasons under the study. In this respect, control fruits recorded higher decay percentage after 60 days which maintained 21.10 and 20.01% in both seasons, respectively. Moreover, chitosan at 2% + green tea at 2% gave lower decay percentage during both seasons which causes only 9.24 and 9.00% decayed fruits after 60 days of cold storage.
Chilling injury (Cl): Symptoms of Chilling Injury (Cl) in Washington Navel orange can be described small pitted areas and skin depressions irregularly distributed over the fruit surface. From Table 1 data presented that all applied treatments reduced chilling injury incidence compared to the control. At the end of the storage period, chitosan at 2% + green tea at 2% gave lower chilling injury indexes were 3.43 and 3.18%, compared with 9.56 and 9.35% in control fruits after 60 days of cold storage during both seasons, respectively.
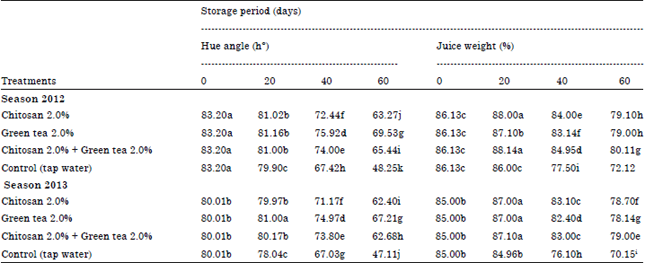
Skin hue color: From Table 2 data presented that all applied treatments were significantly delayed fruit color transition from green yellow to yellow during storage period compared with the untreated fruits in the two seasons.
The delay in color transition at orange skin was expressed as higher hue angle (h°). In control fruits, hue angle decreased rapidly during cold storage (48.25 and 47.11 h°). Moreover, green tea at 2% maintained transition of fruit color from green yellow to yellow than all treatments or the control after 60 days of cold storage in both season. The increment hue using these treatment reached about 69.53 and 67.21 h° after 60 days of cold storage, respectively during both seasons.
Juice weight%: The amount of juice present in a fruit is considered to be one of the most important qualities in juicy fruits like citrus. Data in Table 2 indicated that the percentage of juice weight gradually decreased from harvest till 60 days as a storage period advanced during cold storage.
| Table 2: | Effect of Post-harvest treatments on hue angle (h°) and juice weight (%) of Washington Navel orange fruits during cold storage 2012 and 2013 seasons |
 | |
| Means followed by the same letters are not significantly different by Duncan multiple range test at 0.05 levels | |
Treated fruits with chitosan at 2.0% + green tea at 2% were more effective in reducing the losses percent of juice weight significantly compared with all treatments applied or the control. The percent of juice weight due to this treatment was about 80.11 and 79.00% after 60 days during cold storage in both seasons.
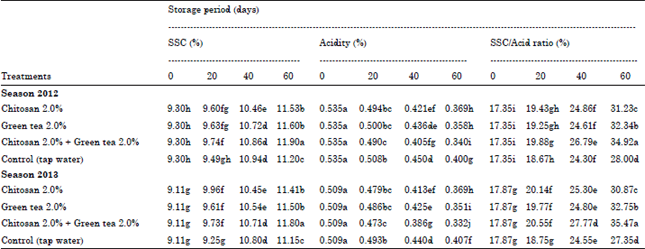
Soluble Solids Content (SSC%): Concerning to the effect on SSC, data from Table 3 showed that the longer the cold storage periods the higher SSC contents obtained in fruit juice of Washington Navel orange in both seasons regardless of the used treatments.
Since, all treatments gave somewhat higher values of SSC in fruit juice than the control fruits. Since, chitosan at 2% + green tea at 2% produced higher SSC values in fruit juice compared to other treatments conducted averaged 11.90 and 11.80% after 60 days of cold storage in both seasons, respectively.
The data also disclose that, control fruits gave less values of SSC which ranged from 11.20 and 11.15% after 60 days of cold storage in the both seasons under the study.
Titratable Acidity (TA): A gradual decrease in titratable acidity was found in all treatments used from harvest until 60 days through cold storage in both seasons. Data from Table 3 demonstrate that all treatments being used reduced the percent of total acidity in fruit juice than the control during both seasons. Control fruits retained higher significant percent of TA than all treatments being used attaining, 0.400 and 0.407% in both seasons, respectively. Commonly, minimum percent of TA was found in fruit treated with chitosan at 2.0% + green tea at 2.0% attained 0.340 and 0.332% during both seasons, respectively.
Soluble solids/acid ratio%: Considering the effect on SSC/acid ratio, data in Table 3 reveal that the values of SSC/acid ratio were progressively increased by the storage period advanced from harvest till 60 days at cold storage.
| Table 3: | Effect of post-harvest treatments on SSC%, acidity% and SSC/acid ratio of washington navel orange fruits during cold storage 2012 and 2013 seasons |
 | |
| Means followed by the same letters are not significantly different by duncan multiple range test at 0.05 levels | |
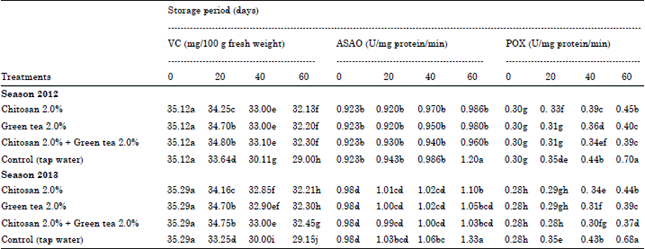
| Table 4: | Effect of post-harvest treatments on Vitamin C, ascorbic acid oxidase activity (ASAO) and peroxidase activity of washington navel orange fruits during cold storage 2012 and 2013 seasons |
 | |
| Means followed by the same letters are not significantly different by duncan multiple range test at 0.05 levels | |
With regard to the effect of these treatments on SSC/acid ratio the data reveal that, control treatment produced a lower value of SSC/acid ratio after 60 days of cold storage since the values averaged about 28.00 and 27.35% during the two seasons, respectively. Moreover, chitosan at 2.0% + green tea at 2.0% treatment gave higher values of SSC/acid ratio (34.92 and 35.47%) during the two seasons, respectively.
Vitamin C (VC) mg/100 g fresh weight: Data presented in Table 4 and Fig. 1 indicated that, Vitamin C contents of orange decreased gradually during cold storage. The reduction in ascorbic acid at control fruits ranged in the first season from 35.12-29.00 mg/100 g fresh weight (about 17.42%) and realized in the second one from 35.29-29.15 mg/100 g fresh weight (about 16.99% losses). This finding is correlated to the previously mentioned data about SSC/acid ratio, where these fruits recorded less fruit quality characters.
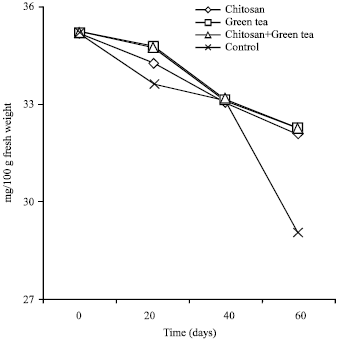 | |
| Fig. 1: | Ascorbic acid (Vitamin C) in Washington Navel orange fruits after 60 days of cold storage as a mean of 2012-2013 seasons |
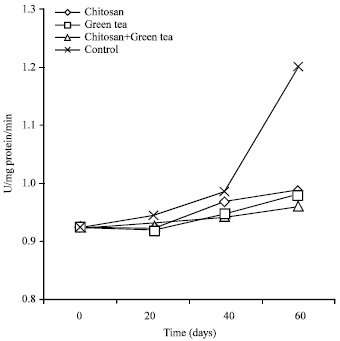 | |
| Fig. 2: | Ascorbic acid oxidase in Washington Navel orange fruits after 60 days of cold storage as a mean of 2012-2013 seasons |
Since, the high pronounced ascorbic acid values were found in fruit treated with chitosan at 2.0% + green tea at 2.0%, as the change in ascorbic acid content due to this treatment ranged from (0-60 days) 35.12-32.30 mg and from 35.29-32.45 mg, in both seasons, respectively (the percent of decline reached 8.04%).
Ascorbic Acid Oxidase (ASAO) (U/mg protein/min): From data presented in Table 4 and Fig. 2, ASAO activity in fruit juice of orange gradually increased during cold storage in both seasons.
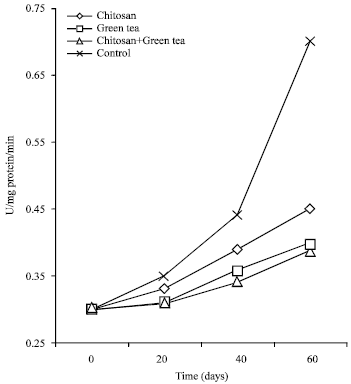 | |
| Fig. 3: | Peroxidase in Washington Navel orange fruits after 60 days of cold storage as a mean of 2012-2013 seasons |
Moreover, all the applied treatments reduced the activity of ASAO in fruit significantly than the control stored for 60 d ays at cold storage. Since, the values in control fruits were 1.20 and 1.33 U mg-1 in both seasons, respectively. Furthermore, chitosan at 2.0% + green tea at 2.0% treatment were more effective in reducing the activity of ASAO in orange fruits compared with all treatments used. The value of ASAO due to this treatment was about 0.960 and 1.03 U mg-1 after 60 days during cold storage in both seasons, respectively.
Peroxidase (POX) (U/mg protein/min): Data presented in Table 4 and Fig. 3 showed that, POX activity in fruit juice of orange gradually increased during cold storage in both seasons. Moreover, all the applied treatments reduced the activity of POX in fruit significantly than the control stored for 60 days at cold storage. Since, the percent in control fruits were 0.70 and 0.68 U mg-1 in both seasons, respectively. Furthermore, chitosan at 2.0% + green tea at 2.0% treatment were more effective in reducing the activity of POX in orange fruits compared with all treatments used. The value of POX due to this treatment was about 0.39 and 0.37 U mg-1 after 60 days during cold storage, respectively, in both seasons.
A novel approach to extend the post harvest shelf life is the use of edible coatings of natural antimicrobial compounds. Chitosan, the deactivated product of chitin, is one of a natural biopolymer with antifungal and eliciting properties, also, a common food additive with antifungal properties, able to reduce postharvest decay of fruits was reported by Romanazzia et al. (2007).
Coating fruits with chitosan reduced respiration rate and weight loss and partially inhibited decay of fruit during storage. The reduction in weight loss percentage and respiration rate by chitosan during cold storage could be due to chitosan enabled epidermal tissues to control water loss and reduce respiratory exchange (Du et al., 1998). Chitosan are barrier to moisture loss and therefore retarding dehydration of fruits (Zhang et al., 2011). Coating citrus fruit with chitosan was effective in controlling fruit decay caused by Penicillium digitatum Sacc. and Penicillium expansum Link (Chien et al., 2007) and rots including gray mould and blue mould caused by Botrytis cinerea.
Chitosan-treated strawberries have shown a range of changes that are related to a slowed ripening, such as decreased Titratable Acidity (TA) with delayed changes in Soluble Solids Content (SSC) and with reduced ethylene production (Mazaro et al., 2008).
Vitamin C contents of orange decreased gradually during cold storage. This finding could be attributed to the conversion of ascorbic acid to dehydro ascorbic acid and decreasing the active form of ascorbic (Hacisevki, 2009). Ascorbic acid is an important nutrient quality parameter and is very sensitive to degradation due to its oxidation compared to these nutrients during food processing and storage (Veltman et al., 2000). Ascorbic acid is also involved in the cell cycle and in other important enzyme reactions in plant tissue (i.e. ethylene biosynthesis).
It has been previously been reported that a loss of ascorbic acid in citrus fruit is not caused by storage temperature (Rapisarda et al., 2008) and that there was no loss of ascorbic acid during storage of oranges at low temperature. Chitosan may protect skin of navel orange fruits from decay, thus delay ripening and improve quality. So, tissues entering senescence or ripening have low biosynthesis of ascorbic acid. The decrease in ascorbic acid level was associated with reduced capability of preventing oxidative damage and with the incidence of disorders during storage (Lin et al., 2008).
Otherwise, ascorbic oxidase (ASAO) activity was very high and correlated to the decrease of vitamin C content. Nevertheless, such decrease did not exceed 20-30% of the initial ascorbic acid content (Ingallinera et al., 2005).
The peroxidases belong to a family of glycoproteins containing iron atoms as a prosthetic group and different quantities of carbohydrate residues. They are located mainly in the cell wall and in the cells vacuoles of plants. POX activities were low in harvested fruit and increased significantly during cold storage.
The increase in POX activity in non-treated fruit correlated with the chilling injury observed in these fruits. The results support the relationship between chilling sensitivity of the fruit skin and the induction of peroxidase enzyme during storage.
Chitosan contributes to induce systemic resistance in plants and this may be due to suppress the increase in activities of (POX) peroxidase (Dong et al., 2004).
The increase in POX activity closely correlated with the importance of chilling disorders in the fruit peel. Treatment with chitosan at 2.0% + green tea at 2.0% reduced Chilling Injury (CI) symptoms and reduced peroxidase (POX) activities. Also, peroxidase activity has antifungal effects and has been implicated in the defense responses to pathogens. The reduction in the activity of PPO and POX might be due to an indirect effect caused by feedback inhibition by the lack of phenolic compounds (substrate) (Masuda et al., 2003).
Green tea is an excellent source of polyphenols which are natural antioxidants that can be used as alternatives to synthetic antioxidants as they are typically less harmful than synthetic ones and appears to have an equivalent effect upon the inhibition of oxidation. These antioxidants which inhibit the oxidation of organic molecules, are very important, not only for living systems and their defense against oxidative stress but also for food preservation (Chan et al., 2007).
It is known that chilling injury is characterized by membrane disruption resulting in loss of tissue integrity accompanied by skin browning, this effect being attributed to the inhibition of polyphenoloxidase and peroxidase activities. Thus, green tea treatments were able to reduce these symptoms and in turn to increase the shelf life and marketability period. It has been proposed that green tea could act as anti senescence agent by enhancing fruit tolerance to low-temperature stress, the effect being attributed to the ability of polyphenols to inhibit oxidation (Gramza et al., 2006).
It could be concluded that, the combination treatment of chitosan (2.0%) and green tea (2.0%) was the most effective treatment in decreasing fruit decay, chilling injury, titratable acidity, activity of ascorbic acid oxidase (ASAO), perooxidase (POX) and losses of juice percentage. However, the same treatment maintain soluble solids content, SSC/acid ratio and ascorbic acid content.
So, application of chitosan and green tea are a promising potential in prolonging the storability of Washington Navel orange fruits, prevent the disease and could serve as an alternative to traditional chemical compounds to enhance the ability of fruits to low cold stress as the optimum quarantine during cold storage.