ABSTRACT
Fruit set in watermelon is influenced by weather conditions and can be improved by pollen collection and storage facilities. Watermelon had tricolpate pollen and was monosphonic in germination. Among the organic solvents tested, pentane, ethyl ether and acetone showed higher evaporation rate. Pentane (18125 pollen/flower) and benzene (16850 pollen/flower) solvents collected high number of pollens as compared to ethyl ether and ethyl acetate. Pollen germination on medium was 67% after collection in pentane but was less than control (87%). Other solvents showed very poor germination. Pollen incubated at 15, 20, 25, 30 and 35°C showed a linear increase in tube growth with the increase in temperature. Pollen stored at -10, 4 and 10°C room temperature for four weeks showed comparatively better germination of pollen stored at -10°C and germination decreased from 70 to 13.2% in 4th week of storage. Temperature 4°C was better (47.4%) for 1 week pollen storage.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.44.49
URL: https://scialert.net/abstract/?doi=ajps.2005.44.49
INTRODUCTION
Worldwide watermelon is the second largest vegetable after tomatoes and Korea ranks 16th in world’s watermelon production[1]. One of the characteristics of Korean watermelon culture is that most of the watermelons (72%) are being produced in protected cultivation (high tunnels) due to temperate climate and hand pollination is practiced. Korean watermelon growers produce three crops a year. Low temperature and high humidity during winter influence anther dehiscence and results in poor fruit set[2].
Triploid plants needs diploid pollen stimulus for fruit development. Similarly diploid parthenocarpic fruits can be produced by pollination with soft-X irradiated pollen[3] and hence pollen collection and storage before pollination is a general practice. The pistillate flower and the staminate flower just below it open the same day (Table 1). The anthers dehisced (Fig. 2a) when the corolla expands[4] , but the pollen remains on the anthers in sticky masses (Fig. 2b). The stigma is receptive throughout the day although most pollination takes place forenoon. Large, sticky pollen grains and an adhesive stigma signal is the necessity for active pollen transfer between flowers for pollination[5].
| Table 1: | The phenology of individual watermelon flowers during the days of receptivity* |
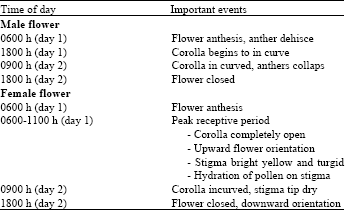 | |
| *Floral behavior was observed in greenhouse grown watermelon during spring | |
Pollen dehydration is essential for storage[6] and organic solvents make it possible to collect and store pollens for longer period of times. Although there have been several studies about pollen collection and storage in organic solvents[7,8] but few organic solvents have been tested for watermelon pollen. Shimizu[8] tested the utilization of 6 organic solvents for watermelon pollen storage and found ethyl acetate and ethyl ether the most suitable. The long-term pollen storage (beyond 1 month) showed more than 40% germination when stored in ethyl acetate at -20°C[9,10] but is genotype dependent[11,12]. Humidity becomes increasingly important at storage above 0°C[13].
Pollen germination and growth of pollen tubes are always necessary for fertilization, fruit development and seed formation. Studies on in vitro germination and pollen tube growth are very useful for explaining the lack of fertility[14,15] and are generally believed to provide the best estimate of pollen viability in vivo[16]. Nenadovic-Mratinic[17] found greatest pollen tube growth at 25°C. The objective of this study was to investigate different organic solvents for pollen collection and their effect on pollen germination, tube growth and storage on different temperatures.
MATERIALS AND METHODS
Seven organic solvents, toluene, chloroform, ethyl ether, ethyl acetate, pentane, acetone and benzene, were first tested for their rate of evaporation. Organic solvents, 10 mL each, were poured in test tubes and placed at room temperature in the laboratory. Evaporation rate, after 1 and 2 h, was recorded by measuring the remaining solvent with pipette. Four organic solvents (pentane, ethyl ether, ethyl acetate and benzene) were selected for further studies. The two higher evaporation rate solvents, pentane and ethyl ether, were selected on the basis of these studies, ethyl acetate was regarded well for watermelon pollen extraction and storage[9,10] and benzene showed maximum pollen germination in grapevine[18]. Male flowers were collected from greenhouse during 8.00-9.00 am to test pollen collection efficiency of these solvents. Anthers (5 flowers each) were removed from the flowers with forceps, submerged in 2 mL solvent and shake to down load the pollen in solvents. The number of pollen grains was counted by using haemocytometer under light microscope.
The germination of pollen grains extracted in these solvents was evaluated in vitro on a culture medium [417 mg L-1 Ca(NO3)2, 200 mg L-1 H3BO3, 100 mg L-1 KNO3, 217 mg L-1 MgSO4.7H2O, 10 g L-1 agar and 20 g L-1 sucrose]. Pollen was separated from solvents by vacuum drying and collected in an Eppendorf microtubes. Pollen was gently rolled with inoculating loop on medium dispensed in 50x12 mm petri dishes. Untreated flowers harvested from greenhouse were used as a control by removing their anthers with forceps and touching with the medium. Cultures were incubated at 25°C under dark. The number of germinated pollen grains from a randomly selected sample of at least 150 pollen grains was counted for each treatment after 2 h of incubation. Pollen germination was recorded by using light microscope at 40x magnifications.
During these investigations pentane solvent showed potential and was further used in pollen germination, tube growth and storage studies. Effect of temperature on pollen tube growth was recorded by incubating cultures on 15, 20, 25, 30 and 35°C. Pollen was cultured on same media and tube growth was measured after 1 h of incubation using Camscope with IT Pro Image Tracer software. For pollen storage studies, pollen grains were collected in pentane solvent and stored at -10, 4 and 10°C. Anthers stored at these temperatures were compared as control with pentane collected pollen for germination efficiency. Pollen was cultured in vitro on same media and incubated at 25°C under dark conditions. Germination was observed up to 4 weeks. Pollen collected in pentane and untreated were cultured to check day zero germination. The experiment was laid out according to completely randomized design with four replications. Data was subjected to comparative continuous descriptive and correlation analysis using Microsoft Excel ‘analyze it’ software.
RESULTS AND DISCUSSION
Evaporation rate of organic solvents: The evaporation rate was noted after 1 and 2 h (Table 2). Pentane had the highest evaporation rate (4.2/10 mL) followed by ethyl ether (4.1/10 mL) and acetone (2.8/10 mL) after 1 h. Toluene had the lowest evaporation rate (0.5/10 mL). The same order of evaporation rate was maintained even after 2 h. Higher evaporation rate of organic solvents is very important to dry pollen in short time before pollination. Shimizu[8] pointed out in his studies that lower evaporation rate results in loss of pollen viability. These organic solvents might cause any physiological or chemical damage to pollen (Table 2).
Pollen collection rate in organic solvents: Pentane, similar to its higher rate of evaporation, collected maximum number of pollen (18125 pollen/flower). Benzene was the second best for pollen collection (16850 pollen/flower). Ethyl acetate yielded the lowest number of pollen (7500 pollen/flower) (Fig. 1).
| Table 2: | Evaporation rate of different organic solvents used in watermelon pollen collection |
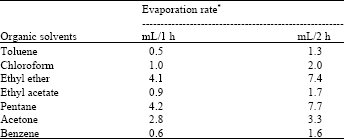 | |
| *A total of 10 mL solvent was used to record the evaporation rate | |
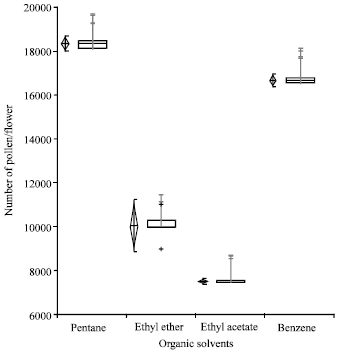 | |
| Fig. 1: | Pollen collection rate in different organic solvents The vertical diamond shows the mean and the requested confidence interval around the mean. The horizontal notched box shows the median and confidence interval around the median + indicates possible near outlier’s observations more than 1.5 IQRs (inter-quartile ranges). Gray vertical bars represent standard error |
Several scientists have discussed the usefulness of organic solvents in pollen collection[7,8,10,18]. Besides maximum pollen collection, the most important is the viability of collected pollen in solvents. Waki and Takeuchi[19] reported that washing flowers with toluene, sucrose solution or water yielded more pollen but the later two solvents reduced the germination rate considerably after drying. Kopp et al.[7] found only toluene and carbon tetrachloride effective for Salix pollen extraction among other tested solvents.
Pollen germination after collection in organic solvents: Pollen was tricolpate with spherical ornamentation (Fig. 2c). Some differences in their sizes were observed. The variations in pollen sizes are probably due to the tetrads that were formed at the end of microsporogenesis[20]. While the germination rates were high among the pollen grains of normal size, no or poor germination was observed in small pollen grains (Fig. 2d). Pollen germination was mainly monosphonic (Fig. 2e) but rarely disphonic (Fig. 2f). In some pollen grains cytoplasm appeared from two or three apertures. In certain cases, pollen tube growth in vitro stopped due to bursting.
The effect of organic solvents on pollen germination (Fig. 3) depicted the highest germination (87%) by fresh pollen as control followed by pentane (67%). Ethyl ether, ethyl acetate and benzene showed very poor (<2%) germination on culture media.
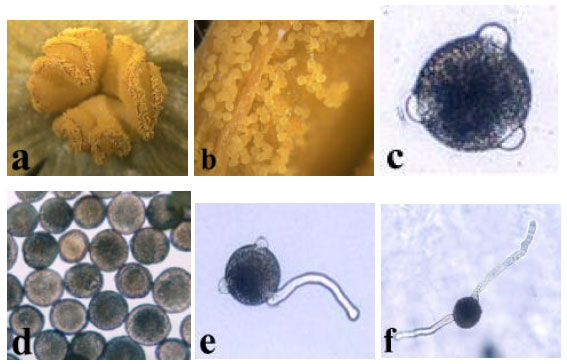 | |
| Fig. 2: | Pollen morphology and germination in vitro a) dehisced anther b) pollen on the anthers in sticky masses c) tricolpate pollen d) different sized pollen e) monosphonic germination f) disphonic germination |
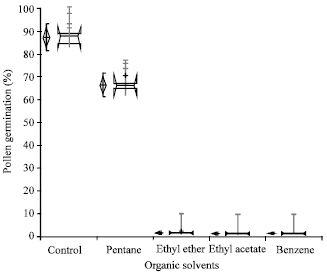 | |
| Fig. 3: | Effect of organic solvents on pollen germination The vertical diamond shows the mean and the requested confidence interval around the mean. The horizontal notched box shows the median and confidence interval around the median + indicates possible near outlier’s observations more than 1.5 IQRs (inter-quartile ranges). Gray vertical bars represent standard error |
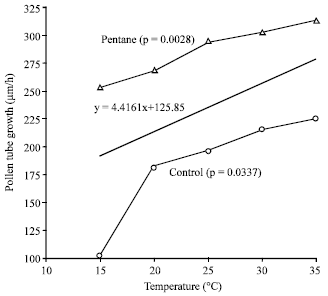 | |
| Fig. 4: | Correlation of temperature and pollen tube growth of untreated (control) and pentane extracted pollen |
This is in contradiction to previous investigations made by Shimizu[8], while testing the utilization of organic solvents for watermelon pollen storage found ethyl acetate and ethyl ether the most suitable but Agarwal[18] examined variable response of pollen stored in organic solvents. Pollen viability and membrane permeability is possibly affected by storage in organic solvents.
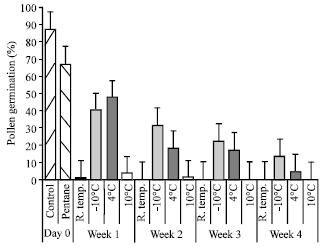 | |
| Fig. 5: | Effect of temperatures on pollen germination after collection in pentane. Thin vertical bars show SE |
Pollen grains stored in organic solvents with low dielectric constants (a measure of their non-polar nature) showed high scores of germinability than solvents with high dielectric constants (a measure of their polar nature)[18]. Jain and Shivanna[21] also correlated viability of pollen grains stored in organic solvents with integrity of pollen membrane. Kopp et al.[7] used different organic solvents for pollen extraction but these reduced pollen viability by 10-20% compared with untreated control whereas pollen extracted with acetonitrile, ethyl ether, chloroform or dichloroethane had germination percentages <5%. Immediate pollination is very important as after removal from solvents approximately 50% of pollen lost its germinability within 2 h at room temperature[8].
Effect of temperature on pollen tube growth: Pollen tube growth was compared between untreated pollen and pollen extracted in pentane solvent and recorded a linear correlation among temperature and tube growth. In control, it ranged 94.6 to 225.8 μm during 1 h of incubation at 15-35°C (Fig. 4). Low temperature, 15°C yielded minimum length of pollen tube both in control and pentane solvent (94.6 and 212 μm, respectively). Pollen tube growth increased with increase in temperature. Maximum pollen tube was recorded on 35°C in pentane collection and was 261.2 μm. Similarly Tuinstra and Wedel[16] examined that pollen tubes germinated at 10°C appeared to be shorter and less well developed than pollen tubes evaluated at higher incubation temperatures. Pollen tube growth in cherry was different depending on the incubation temperatures[17]. As temperature increased from 10 to 32°C, pollen tube elongation rate increased in snake melon (Cucumis melo var. flexuosus) but cucumber pollen tubes were only stimulated up to 21°C[22]. Pollen tube growth of pollen collected in pentane was higher than untreated control. Fresh pollen has higher moisture content[6]. Pollen extracted in organic solvents is dehydrated and when cultured on germination media, might be hydrate and germinate earlier than control. Hence longer pollen tubes were observed in pollen collected in pentane solvent. Pollen tube growth in cucurbits is also related to ovary and pollen size but we did not work out this relation.
Effect of storage temperature on pollen viability: Untreated pollen and pollen collected and stored at -10, 4 and 10°C were evaluated for pollen germination. Before storage, untreated pollen and pollen collected in pentane were cultured to record day zero germination. Untreated and pentane collected pollen exhibited 86 and 70% germination, respectively (Fig. 5). In first week of storage most of the untreated flowers spoiled and there was very little pollen germination. Pollen stored at -10 and 4°C maintained a moderate (>40%) germination percentage for 7 days. In 2nd week, pollen germination decreased to 31.3% at -10°C and this subsequently decreased to 22.2 and 13.2% in 3rd and 4th week, respectively. Other storage temperatures also showed a consistent decrease in pollen germination. Pollen stored at 10°C retained very poor germination (3.5%) and lost completely any viability after 7 days. Similarly van der Walt and Littlejohn[13] and Kopp et al.[7] reported low temperature storage more effective for long storage. Bomben et al.[23] mentioned a rapid and linear decrease in pollen germination stored at 4°C than at -18°C along with divergent response of species. Physiological considerations are also important during the course of pollen storage. Jauh[24] proposed that some proteins and mRNAs, which are necessary for pollen tube germination and growth, might be gradually degraded during long storage.
This study provides ease of handling stored pollen and had the promise to overcome the problem of pollination and fruit set due to indehiscence of anthers or non-viable pollen during winter. Pollen extraction with pentane and subsequent storage may reduce viability when compared with untreated pollen. However, crosses using pollen samples with only 10% viable pollen yielded large numbers of seeds[7]. Several researchers have reported pollen collection and storage in ethyl ether and ethyl acetate[8,10] but our attempts to germinate pollen in these solvents were unsuccessful or resulted in poor germination. In contrast, pentane was used first time in watermelon pollen collection and short-term storage and has the potential to use in future watermelon pollinations.
ACKNOWLEDGMENTS
We thanks Korean MAF and Korea Science and Engineering Foundation (Brain Pool Program) for providing funds to furnish this research.
REFERENCES
- Tuinstra, M.R. and J. Wedel, 2000. Estimation of pollen viability in grain sorghum. Crop Sci., 40: 968-970.
CrossRefDirect Link








