ABSTRACT
Embryogeneic callus induction and subsequent differentiation is accomplished by application of suitable growth regulators and controlling conditions during culture. The growth regulator, 2,4-D is one of the most effective auxin for calli induction in sweet potato (Ipomoea batatas (L.) Lam.), however the optimum concentration varies with individual cultivars. The optimum 2,4-D level for the local sweet potato cultivars has not been determined yet it is important in multiplication of planting material and transformation procedures. To determine the optimum 2,4-D concentration that produced embryogenic calli early and in high frequencies for local Kenyan sweet potato cultivars Mugande, SPK004, Kemb10, Japon tresmesino and Zapallo, leaf explants were cultured in vitro on MS basal medium supplemented with six concentrations of 2,4-D (0, 0.5, 1.0, 2.0, 3.0 and 5.0 mg L-1), set as a factorial treatment arranged in a Completely Randomized Design (CRD), replicated three times. For each treatment four parameters were considered: Mean number of days taken to form calli, calli weight, calli diameter and calli strength. The results established significant (p<0.05) differences in cultivar responses to 2,4-D levels. Calli induction was effective when supplemented at low levels of 0.5 mg L-1 as high levels above 3.0 significantly reduced the quantity and quality of embryogenic calli that was formed. Low levels of 2,4-D should be used for local Kenyan sweet potato cultivars as the number of days taken to form calli reduced and calli incidences increased.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.484.489
URL: https://scialert.net/abstract/?doi=ajps.2007.484.489
INTRODUCTION
The sweet potato (Ipomoea batatas (L.) Lam) is an important food security crop in developing countries, with a high yield potential and excellent nutritive value in the storage roots (Burden, 2005). Lately, sweet potato has received increased attention because the crop can adapt to a wide range of environmental conditions and grow on soils with limited fertility and inadequate moisture (Bioethics, 2004). However, the improvement of the crop is vital for its agricultural productivity is adversely affected by viral infections and insect pests (Aritua et al., 1998).
Genetic improvement of sweet potato by conventional breeding is limited due to male sterility, incompatibility and hexaploid nature of the crop (Dhir et al., 1998). This has led to more emphasis on the use of biotechnological methods for genetic improvement in sweet potato, hence methods involving genetic transfer and somatic hybridization are attractive as they enable the direct introduction of desirable genes from other sources into the preadapted cultivars (Prakash, 1994). The adoption of genetic engineering techniques warrants optimization of a reproducible protocol that enables embryogenic calli formation and subsequent development of a reproducible plant regeneration system. This involves manipulation of in vitro tissue culture conditions with a sole aim of inducing calli to form early and in high frequencies of which plant growth regulators are fundamental (Panis et al., 2006).
The type and concentrations of plant growth regulators applied in culture medium were found to affect the successful induction of embryogeneic tissues (Al-Mazrooei et al., 1997). With the stimulus of endogenous growth substances or by addition of exogenous growth regulators to the nutrient medium, cell division, cell growth and tissue differentiation are induced (Tripathi and Tripathi, 2003). Several plant growth regulators have been used in sweet potato and among them 2, 4-Dichlorophenoxyacetic Acid (2,4-D) has been found to be the most effective auxin for embryogeneic calli induction and plant regeneration in sweet potato (Nguyen et al., 1997). Nevertheless the optimum amount required for embryogeneic callus induction and proliferation has not been studied for locally adaptable sweet potato cultivars. The purpose of this study was to determine the optimum 2,4-D concentration suitable for embryogenic callus induction in the local sweet potatoes in tissue culture.
MATERIALS AND METHODS
Study site: This experiment was carried out at the Biotechnology laboratory at the Kenya Agricultural Research Institute (K.A.R.I) Njoro, Nakuru District, Kenya under controlled temperature ranging from 27 to 29°C, 16/8 h photoperiod and light conditions of 700 μ mol m-2 sec-1.
Test cultivars: The sweet potato cultivars used were sourced from Centre Internationale de Potato (C.I.P) Nairobi, Kenya. The cultivars included SPK004, Kemb 10, Mugande, Japon tresmesino and Zapallo chosen based on their popularity, preferred taste by most farmers and their growth performance under local conditions. The cuttings obtained were grown in plastic pots in a green house to provide a readily available supply of stock plants. For constant availability of in vitro material during this study 5 cm shoot tips were collected as explants from grown cuttings and established in vitro for further multiplication.
Establishment of sweet potato material for in vitro micro propagation: Standard sterilisation procedures followed were as described by Pierik (1993). Shoot tips were inoculated onto multiplication media composed of 4.4 g L-1 Murashige and Skoog Basal Medium (MSBM) (Murashige and Skoog, 1962) supplemented with 100 mg L-1 Myo-inositol, 5 mg L-1 Gibberellic acid and 30 g L-1 Sucrose. The pH of the media was adjusted to 5.8 prior to adding Phytagel at a rate of 3 g L-1 (Zheng et al., 1996) and autoclaved at 121°C and 1.035 kPa for 15 min. Several cultures were established using only in vitro germinated plantlets till 100% aseptic material were obtained. Stock plantlets were maintained by continuous sub-cultures at 2-3 week intervals. The cultures of in vitro plants were maintained in a growth chamber set at a temperature of 27°C+0.5 for a 16 h photoperiod and under light intensity of 700 μ mol -m2 sec-1 provided by cool fluorescent tubes (Phillips).
In vitro establishment of callus culture: The source of plantlets used to set up callus cultures was from in vitro material established above. Equal amounts of media composed of half strength MSBM, supplemented with 100 mg L-1 Myo-inositol, 2 mg L-1 Thiamine-HCL, 40 g L-1 Sucrose, 10 m L-1 Vitamin stock solution (200 mg L-1 Calcium Pantothenate, 2 g L-1 Gibberelic acid, 10 g L-1 Ascorbic acid and 10 g L-1 Putrescine) and 3 g L-1 Phytagel was dispensed into six 500 mL volumetric conical flasks, autoclaved and cooled to 42°C (Zheng et al., 1996; Nguyen et al., 1997). For each cultivar, leaves from most recently expanded in vitro plantlets were cut into 50 pieces of 5 mm length. The cultures were transferred to a dark incubator set at a temperature of 29°C. Light was provided after six weeks in the dark and whole induced calli transferred to fresh media.
Data collection: To determine the optimum 2,4-D concentration for the local Kenyan sweet potato cultivars the number of days taken for calli to form, the colour of calli, calli weight and calli strength were observed and recorded.
Statistical analysis: The experimental design was a factorial treatment arranged in a completely random design with five levels of the factor cultivar and 6 levels of 2,4-D concentration, replicated three times and placed in an incubator. Calli strength was transformed by square root transformation (![]() ) to homogenise the variance before subjecting to ANOVA as suggested by Hills and Thomas (I978). Data was then subjected to Statistical Analysis Systems (SAS) (Proc:ANOVA) and where F-test was significant at 5% level, means were separated by Duncan’s Multiple Range Test (DMRT) (Hills and Thomas, I978; SAS Institute, 2001).
) to homogenise the variance before subjecting to ANOVA as suggested by Hills and Thomas (I978). Data was then subjected to Statistical Analysis Systems (SAS) (Proc:ANOVA) and where F-test was significant at 5% level, means were separated by Duncan’s Multiple Range Test (DMRT) (Hills and Thomas, I978; SAS Institute, 2001).
RESULTS
Days taken to form calli: Across most cultivars, the 2,4-D level that took the overall shortest time to form calli was attained at 0.5 mg L-1. However, shortest mean number of days taken to form calli in individual cultivars was obtained when the callus induction media was devoid of 2,4-D (39.9 days) followed by 0.5 mg L-1 2,4-D. These was obtained with the cultivar Mugande which gave a mean of 32 days (0.5 mg L-1) followed by the cultivar Kemb 10 with 33.3 days, SPK 004 with 36 days, when no 2,4-D was supplemented in the medium. J. tresmesino took 41 days with 1.0 mg L-1 and Zapallo took 52 days in medium with 0.5 mg L-1 2,4-D (Table 1). Differences in calli formation patterns were also observed among the cultivars. In Kemb 10, SPK004 and Mugande embryogenic calli formed in a circular pattern and spread out on the leaf surface while in the cultivars J. tresmesino and Zapallo calli formed mostly around the leaf edges but for all cultivars no calli developed below the leaf explant surfaces (Fig. 1).
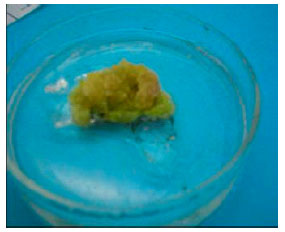 | |
| Embryogenic calli formed in a circular pattern from the cultivar Kemb 10 | |
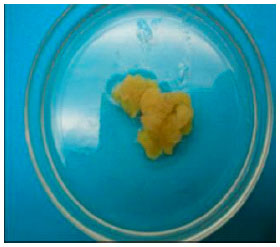 | |
| Embryogenic calli formed along the leaf edges from the cultivar Zapallo | |
| Fig. 1: | Selected calli formation patterns for the local Kenyan Sweet potato cultivars induced in vitro in tissue culture |
Calli weight: The incidence of calli formation and subsequent calli weight was dependent on the 2,4-D concentration supplemented in the callus induction medium and the test cultivars used. It was observed that using 0.5 mg L-1 2,4-D led to a significant increase in calli weight, while the lowest means were obtained at 1.0 mg L-1 followed by 3.0 and lastly 5.0 mg L-1 2,4-D (Fig. 2).
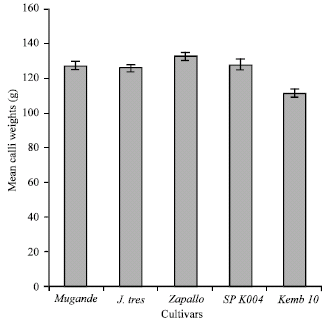 | |
| Fig. 2: | :Mean calli weights of sweet potato cultivars cultured on callus induction media composed of different 2,4-D levels |
Comparison of calli weights indicated genotype dependant responses to the 2,4-D added in the culture medium. The highest calli weights were attained with the cultivar Zapallo followed by SPK004 while the lowest overall mean calli weights were observed in Kemb10 (Table 2). Calli weights recorded progressively at 4, 6 and 8 weeks in culture showed a declining trend when the culture period was prolonged.
Calli strength: In all cultivars the most suitable calli strength was scored with the treatment devoid of 2,4-D followed by 0.5 and 1.0 mg L-1 2,4-D. The lowest means were attained at 2.0 mg L-1 2,4-D. Calli formed by the sweet potato cultivars were of different strengths and types (Fig. 3). Embryogeneic calli which were light yellow in colour were transferred to callus maturation medium for further development. The cultivar Mugande formed calli of favourable strength at both high and low 2,4-D levels contrary to the response in cultivars Zapallo, J. tresmesino and Kemb 10 which gave favourable calli strength at only low levels of 2,4-D.
| Table 1: | The interactive effect between sweet potato cultivars and 2,4-D conc. on mean number of days taken to form calli |
 | |
| Means within a column followed by the same letter are not significantly different at p = 0.05 based on Duncan’s Multiple Range Test (DMRT) | |
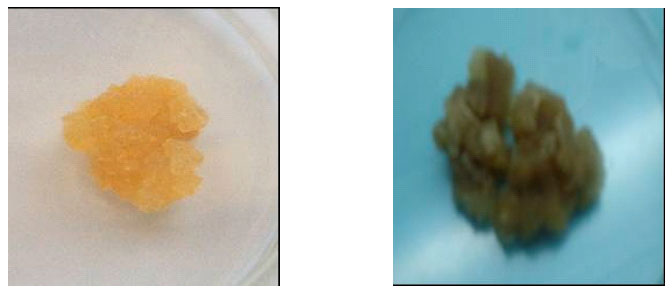 | ||
| Leaf explants from the cultivar Kemb10 at 4-6 weeks culture on callus initiation medium supplemented with 0.5 mg L-1 2,4-D | Leaf explants from the cultivar Kemb10 at 10 weeks culture after initiating callus cultures on 3.0 mg L-1 2,4-D | |
 | ||
| Dull brown non-embryogenic calli showing compact appearance with calli growing closely attached around the leaf explant | Firm, yellow and fully developed embryogenic callus, growing away from the explant | |
| Fig. 3: | Distinction of calli types formed from local sweet potato cultivars grown in vitro on callus induction medium at KARI-Njoro | |
| Table 2: | The interactive effect between the levels of 2,4-D and the 5 sweet potato cultivars on mean calli weight (g) |
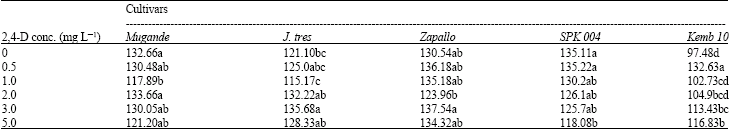 | |
| Means within a column followed by the same letter are not significantly different at p = 0.05 based on Duncan’s Multiple Range Test (DMRT) | |
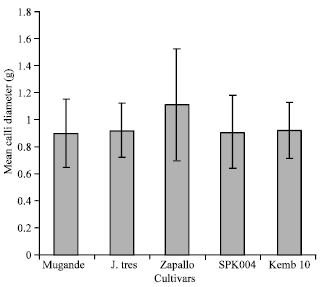 | |
| Fig. 4: | Mean calli diameter of sweet potato cultivars grown in vitro at KARI-Njoro |
Calli diameter: The highest mean calli diameter was recorded with the cultivar Kemb 10 at 0 mg L-1 2,4-D while the lowest calli weights were recorded in the cultivars Mugande and Kemb 10 at 3.0 mg L-1 2,4-D and Zapallo at 2.0 mg L-1 2,4-D. Increasing 2,4-D levels from 0.5 to 2.0 mg L-1 2,4-D significantly (p<0.05) reduced the calli diameter before an increase was observed to occur at 3.0 and 5.0 mg L-1 2,4-D (Fig. 4).
DISCUSSION
From the study, the level of 2,4-D supplemented in the culture medium significantly affected the incidences, type and quality of calli that was formed. A negative correlation was established between 2,4-D levels applied in the culture medium and calli incidences implying that increasing 2,4-D levels influenced the calli incidences from the local cultivars such that there was a significant decrease in the calli weights, calli strength, calli diameter and increased number of days taken to form calli. The optimum concentration of 2,4-D were identified to be effective when they were supplemented at low levels of 0, 0.5 or 1.0 mg L-1. These concentrations formed calli in high frequencies for all the local cultivars contrary to when high 2,4-D levels 2.0, 3.0 and 5.0 mg L-1 were used. Likewise, using the low levels of 2,4-D resulted in a decrease in the mean number of days taken to form calli and lower calli weights.
The disadvantage of increasing 2,4-D levels was evident from the negative correlation that was obtained between the 2,4-D concentration and number of days taken to form calli (r2 = -0.36, p = 0.0005). High concentration of 2,4-D levels may have caused inhibition of cell division within the explant. Radhakrishnan et al. (2001) reported that cells used up 2,4-D amounts as required and any excess began to actively show the herbicidal effects therefore slowing down the callus induction process. Nguyen et al. (1997) reported that cultivar differences played a significant role in determining the amount of calli that formed. Kemb 10 was more sensitive to an increase in the amount of 2,4-D compared to other cultivars.
Prolonging culture period for more than six weeks led to reduced calli weights in each treatment. This reduction in weight in the callus cultures of the local cultivars was probably due to exhaustion of nutrients in the medium by leaf explants during calli formation, therefore destabilizing callus culture proliferation. It could also have been due to moisture and aeration conditions during culture that influenced hormones to cause rapid cell re-adjustment during cell division as reported by Vasil and Vasil (1983). For successful induction of callus to occur, chemical signals are sent to the cells of leaf explants that are to form calli to counteract and adjust to the 2,4-D levels within the medium as reported by Sachs (1993).
Differences in the sensitivities of 2,4-D levels among the cultivars were critical for this study as optimal calli induction determined the strength of calli. Silvertand et al. (1996) also reported similar results in leek (Allium ampeloprassum L.) that higher 2,4-D levels gave a watery and soft callus while low 2,4-D gave compact embryogenic callus. In the local I. batatas cultivars used in this research, the most suitable calli strength was scored with the treatment devoid of 2,4-D followed by 0.5 and 1.0 mg L-1 2,4-D. This was probably because calli formed at high 2,4-D levels were small, undeveloped and took long to form. Light yellow to white callus was the most suitable calli type for this study because these calli were easier to isolate from medium and transfer whole without contamination or callus breaking. Small, undeveloped, soft dispersed callus having a yellowish-brown colour was observed on the media surface a finding in agreement with studies by Nguyen et al. (1997), who reported that these calli hardly developed and were discarded. Early and timely transfers had to be done in this study as prolonged cultures negatively affected callus proliferation and subsequent calli weight.
Depending on the 2,4-D level supplemented on the culture medium and the test cultivar, different types of calli type and strength developed. Significant differences in cultivar responses to the 2,4-D treatments supplemented in culture medium on the variables above was established. The cultivar Zapallo was most responsive in all the variables mentioned as it gave higher ratings when compared to other cultivars. Therefore the order of ease of regeneration was Zapallo followed by SPK004, Mugande, Japon tresmesino and finally Kemb 10.
ACKNOWLEDGMENT
The authors wish to thank the Kenya Agricultural Research Institute (KARI)-Njoro and International Atomic Energy Agency (IAEA) for providing funds and facilities that supported this research, Centre Internationale de Potato (C.I.P) Nairobi, Kenya for providing planting material and all who contributed towards the success of this research.
REFERENCES
- Al-Mazrooei, S., M.H. Bhatti, G.C. Henshaw, N.J. Taylor and D. Blakeslew, 1997. Optimisation of somatic embryogenesis in fourteen cultivars of sweet potato (Ipomoea batatas (L) LAM). Plant Cell Rep., 16: 710-714.
Direct Link - Aritua, V., E. Adipala, E.E. Carey and R.W. Gibson, 1998. Incidences of sweet potato virus disease: Virus resistance of sweet potato grown in Uganda. Ann. Applied Biol., 132: 399-411.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Sachs, T., 1993. The role of auxin in the polar organization of apical meristems. Aust. J. Plant Physiol., 20: 531-533.
CrossRefDirect Link - Silvertand, B., A. Rooyen, P. Van Lavrjsen, A.M. Harlem and E. Van Jacobsen, 1996. Plant regeneration via organogenesis and somatic embryogenesis in callus cultures derived from mature zygotic embryos of leek (Allium ampeloprassum L.). Euphytica, 71: 261-270.
Direct Link - Tripathi, L. and J.N. Tripathi, 2003. Role of biotechnology in medicinal plants. Trop. J. Pharm. Res., 2: 243-253.
Direct Link








