ABSTRACT
In this study Ca-oxalate crystals in vegetative and floral organs of Galanthus plicatus L., Galanthus gracilis L., Galanthus elwesii L. were investigated. The crystals were detected in all vegetative organs but they were only detected in tepal and ovary of floral organs. They were not observed in mature anthers and old adventative roots. Calcium oxalate crystals were of the raphide type.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.508.512
URL: https://scialert.net/abstract/?doi=ajps.2007.508.512
INTRODUCTION
Ca-oxalate crystals are inclusions (biominerals) that are often seen in higher plants (Esau, 1976; Franceschi and Horner, 1980) and in fungi (Arnott, 1995). The cells in a plant tissue that produce the crystals are generally referred to as crystal idioblasts (Foster, 1956; Arnott, 1982; Monje and Baran, 2002). Calcium oxalate occurs in two hydration states in plants, as the monohydrate (whewellite) or as the dihydrate (weddellite; Frey-Wyssling, 1981; Arnott, 1982). A number of crystal habits have been found for both hydration states: raphids, prisms, styloids, druses and crystal sand (Franceschi and Horner, 1980; Arnott, 1982). Both the chemical nature and the morphology of these crystals, as well as their localization within the plant body, could be specific for a given species (Monje and Baran, 2002). So they are used in plant taxonomy (Lersten, 1974). The Ca-oxalate crystals in the epiderma, mesophyll, cortex, phloem, xylem parenchyma and pericarp of various plants were observed by plant anatomists (Esau, 1976). Presence of the crystals in various tissues of laticifers (Dehgan and Craig, 1978), corm (Sunell and Healy, 1979) and seeds (Ilarslan et al., 1997) is known. In recent years it is also notable that these crystals were present in transitory floral organs such as stamens, gynoecia and petals (Meric and Dane, 2004). They are quite prevalent in floral organs of many taxa including Dilleniaceae, Liliaceae, Palmae, Malvaceae, Cunoniaceae, Euphorbiaceae (Tilton and Horner, 1980), Solanaceae (Horner and Wagner, 1992), Leguminosae (Buss and Lersten, 1972). The roles of Ca-oxalate crystals in plant growth and development remained unclear (Prychid and Rudall, 1999). Recently, detailed description of the calcium oxalate raphide crystal content of Leucojum aestivum L. in vegetative and generative tissues in different phases of its life cycle were studied (Ekici and Dane, 2007). In this study Ca-oxalate crystals of some Galanthus species which is sister genus with Leucojum were researched. The aim of this study is to contribute to the solutions of the problems about Ca-oxalate crystals and taxonomical problems of Amaryllidaceae.
MATERIALS AND METHODS
Studies of Ca-oxalate crystal types were made on plants from herbarium materials of Galanthus plicatus L. (EDTU 2055), Galanthus gracilis L. (EDTU 4723), Galanthus elwesii L. (EDTU 3101). Vouchers were kept in the herbarium of Biology Department of Trakya University (EDTU). Clearing-squash technique which developed by Herr (1971) was used to observe Ca-oxalate crystals in different tissues of Galanthus sp. Dry herbarium materials of Galanthus sp. were hydrated in warm water and then they were first pretreated in lactic acid (85%) for 24 h at room temperature. After this pretreatment materials were transferred directly to a recently developed clearing fluid composed of lactic acid (85%), chloral hydrate, phenol, clove oil and xylene (2:2:2:2:1, by weight) for 48 h at room temperature. Then they were taken on slides with a small amount of clearing fluid by means of a Pasteur pipette for microscopic examination (Herr, 1971). Some of the slides were coloured with 2% aceto-orcein. All preparations were examined under Olympus BH-2 microscope. Photos were taken with Olympus C-5060 digital camera.
RESULTS AND DISCUSSION
In this study Ca-oxalate crystals in vegetative and floral organs of Galanthus plicatus L., Galanthus gracilis L., Galanthus elwesii L. were investigated. Crystals were observed in three species of Galanthus in vegetative organs as young adventative roots, bulb membrane (Fig. 1a-c), flower stalk (Fig. 2a-c) and leaf cells (Fig. 3a-c). They were only observed in mature anthers and ovary (Fig. 4a-c) and tepal cells (Fig. 5a-c) of floral organs.
 | |
| Fig. 1: | Ca-Oxalate crystals in the bulb membrane cells of Galanthus sp. (a) G. elwesii (Coloured with 2% aceto-orcein); (b) G. gracilis and (c) G. plicatus. Scale bars = 40 μm |
Crystal content was dense in bulb membrane, flower stalks, leaves, tepals, ovary and young adventative roots, respectively. Calcium oxalate crystals were of the raphide type.
Physical and chemical conditions such as temperature, pressure, pH and ion concentration, may affect crystal growth, location and properties (Franceschi and Horner, 1980), however it is considered that crystal formation within the cell is under genetic control (Ilarslan et al., 2001).
 | |
| Fig. 2: | Ca-oxalate crystals in the flower stalk cells of Galanthus sp. (a) G. elwesii; (b) G. gracilis and (c) G. plicatus. Scale bars = 40 μm |
 | |
| Fig. 3: | Ca-Oxalate crystals in the leaf cells of Galanthus sp. (a) G. elwesii; (b) G. gracilis and (c) G. plicatus. Scale bars = 40 μm |
Although some species of monocotyledones have different crystal types in adjacent cells, a particular taxon can have specific crystal shape (Prychid and Rudall, 1999). According to Watson and Dallwitz (2005) mezophyll cells of leaves of Amaryllidaceae plants contain raphide crystals.
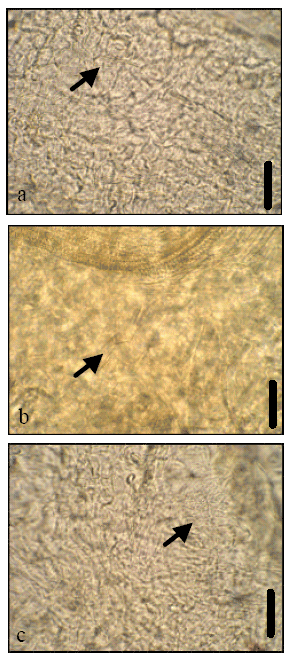 | |
| Fig. 4: | Ca-Oxalate crystals in the ovary cells of Galanthus sp. (a), G. elwesii (b) G. gracilis and (c) G. plicatus. Scale bars = 40 μm |
In this study, raphide crystals were observed in all vegetative and generative tissues except anthers and old adventative roots of Galanthus sp. at their flowering phases like L. aestivum which sister genus with Galanthus (Ekici and Dane, 2007). Their existence in meristematic cells has been demonstrated in several studies (Dane et al., 2000; Ekici and Dane, 2007; Horner and Wagner, 1980; Kausch and Horner, 1983; Seago and Marsh, 1989) as they were seen in young adventative roots of Galanthus sp.
 | |
| Fig. 5: | Ca-oxalate crystals in the tepal cells of Galanthus sp. (a) G. elwesii; (b) G. gracilis (Coloured with 2% aceto-orcein) and (c) G. plicatus. Scale bars = 20 μm |
These crystals were also seen in ovary wall of Triteleia (Themidaceae) (Berg, 2003) which is related taxon to Amaryllidaceae and Galanthus nivalis (Amaryllidaceae) (Chudzik and Sniezko, 2003), L. aestivum (Amaryllidaceae) (Ekici and Dane, 2007) like G. plicatus, G. gracilis and G. elwesii.
In conclusion, this study was determined CaOx crystals in G. plicatus, G. gracilis and G. elwesii, being members of Amaryllidaceae in floral stages of its development. Although crystals were seen in connective tissue of anthers of L. aestivum in pollen mitosis phase they were not seen in mature anthers of Galanthus species and L. aestivum. In further studies we will focus on crystal content of these taxa during their life cycles.
REFERENCES
- Berg, R.Y., 2003. Development of ovule, embryo sac and endosperm in Triteleia (Themidaceae) relative to taxonomy. Am. J. Bot., 90: 937-948.
Direct Link - Chudzik, B. and R. Sniezko, 2003. Calcium ion presence as a trait of receptivity in tenuinucellar ovules of Galanthus nivalis L. Acta. Biol. Cracoviensia Series Bot., 45: 133-141.
Direct Link - Dane, F., C. Meric and G. Huseyinova, 2000. Some Ultrastructural observations on calcium oxalate raphide crystal idioblasts and meristematic cells of the adventive root tips of Sternbergia lutea (L.) Ker-Gawl. ex Sprengel (Amaryllidaceae). Turk. J. Bot., 24: 71-80.
Direct Link - Dehgan, B. and M.E. Craig, 1978. Types of laticifers and crystals in Jatropha and their taxonomic implication. Am. J. Bot., 65: 345-352.
Direct Link - Foster, A.S., 1956. Plant idioblasts: Remarkable examples of cell specialization. Protoplasma, 46: 184-193.
CrossRef - Franceschi, V.R. and H.T. Horner, 1980. Calcium oxalate crystals in plants. Bot. Rev., 46: 361-427.
Direct Link - Frey-Wyssling, A., 1981. Crystallography of the two hydrates of crystalline calcium oxalate in plants. Am. J. Bot., 68: 130-141.
Direct Link - Herr, J.M., 1971. A new clearing-squash technique for the study of ovule development in angiosperms. Am. J. Bot., 58: 785-790.
Direct Link - Ilarslan, H., R.G. Palmer, J. Imsande and H.T. Horner, 1997. Quantitative determination of calcium oxalate in developing seeds of soybean (Leguminosae). Am. J. Bot., 84: 1042-1046.
Direct Link - Meric, C. and F. Dane, 2004. Calcium oxalate crystals in floral organs of Helianthus annuus L. Acta Biol. Szegediensis, 48: 19-23 (Asteraceae).
Direct Link - Monje, P.V. and E.J. Baran, 2002. Characterization of calcium oxalates generated as biominerals in cacti. Plant Physiol., 128: 707-713.
Direct Link - Prychid, C. and P. Rudall, 1999. Calcium oxalate crystals in monocotyledons: A review of their structure and systematics. Ann. Bot., 84: 725-739.
Direct Link








