Research Article
Whey Induced-Modifications in Growth, Photosynthetic Characteristics, Protein patterns and Water Relations of Wheat Seedlings
Mansoura University, Mansoura, Egypt
A.H. Ibrahim
Seuz Canal University, North Sinai, Egypt
In most food production, by-products are inevitable. Traditionally such products have been considered as waste and dumped as sewage. Now, with more attention to environmental issues, such sewage is being put a multiplicity of uses (Jones et al.,1993; Robbins et al., 1999 and Schachermayr et al., 2000).
Cheese whey is produced in large quantities world-wide. One ton of cheese result in the production of about 8 tons of liquid whey (Abdel-Rahman and Abo-Hamed, 1992). However, little research has been conducted on the possibility of using whey as bio-fertilizer. In a study of the nutritive value of whey applied to plants grown in greenhouse on Miami silt loam, Cain (1953) found that oats which received whey at time of planting gave a greater yield than those which did not receive whey.
A study on the composition of whey and its effect on soil and the growth of alfaalfa, when applied at 4 rates (64, 128, 192 and 384 tons acre-1 week-1) for 3 weeks was carried out by Sharratt et al. (1959). They concluded that alfalfa can tolerate about 64 tons of whey a week without harm. They also found that number and size of soil aggregates increased with each treatment of whey up to 192-ton treatment. In addition, Sharratt et al. (1962) extended this study to include the effects of whey application on soil and corn growth. They found that all rates of whey application were effective in increasing corn yields in first and second season after application. Nitrate determinations in soil receiving whey indicated that the organic nitrogen in whey soil is readily converted to nitrates during the first and second season after application. Moreover, they concluded that whey had a beneficial effect on soil aggregation.
Recently, Jones et al. (1993) found that whey application, to sodic soil, lowered sodium adsorption ratio, exchangeable sodium percentage and saturation paste pH in soil surface. Furthermore, Prasad et al. (2000) studied the effect of cow’s whey on germination and enzyme activity of maize and cowpea seeds and found that presoaking of seeds in whey for 18 h increased germination rate and enzyme activity (amylase, proteinase and starch phosphorlase) of used seeds as compared with control.
On the light of the above limited reviews, it is of particular interest to investigate the effect of grains priming in different whey concentrations on growth vigor of wheat seedlings, photosynthetic pigments, 14C2 assimilation, nitrogen constituents, protein profile, ions content and some plant water relations.
A homogenous lot of wheat (Triticum aestivum L.cv. Giza 157) grains was selected. The grains were surface sterilized by soaking in 0.001 M HgCl2 solution for 3 min, washed thoroughly with distilled water and then soaked in the different concentrations of whey (100, 50 and 25% ) and in water (to serve as control) for about six hours at room temperature.
After soaking all the grains were thoroughly washed with water and then sown in earthern pots containing equal amounts of soil (clay, sand; 2/1 v/v). The pots, containing equal uniformed seedlings, were kept in greenhouse under a normal day/ night regime and irrigated with tap water when required. After two weeks from sowing, samples from each treatment were taken.
For estimation of leaf area and fresh and dry matter, ten samples were used and for the chemical analyses triplicate samples were taken from the 1st fully expanded leaf (from apex). Leaf lamina area was measured as 0.7 x length x breadth (Quarrie and Jones, 1979).
Extraction, clarification and determination of saccharides were carried out following the procedure of Younis et al. (1969).
Contents of chl a, chl b and carotenoids were estimated by the spectrophotometric method as recommended by Lichtenthaler (1987).
For determining 14C2 assimilation (Shaddad, 1979), 300 mg of leaf discs was introduced into a fixation apparatus designed to allow 14C2 to be photosynthesized by the discs. Aqueous solution of 14C-sodium carbonate of known ctivity (3.7 MBq cm3) was pipetted into the apparatus followed by 0.2 cm3 H2SO4 (10%). The evolved CO2 passed over and radioctivity of the green leaf discs was measured using a Packard Scintillation Counter.
For Hill reaction assay, detached leaves were ground using a chilled blender, in 50 mM MgCl2 and the resulting homogentate was suspended in 1 mM Na-Tricine, 10 mM NaCl and 10 mM MgCl2 solution and kept at –4.0 °C (Arnon, 1949). Hill reaction activity was determined as the rate of 2, 6 dichlorophenol indophenol (2, 6-DCPIP) photoreduction (Trebst, 1972) using a spectronic 21 D Spectrophotometer.
The method used for extraction and determination of total nitrogen, total soluble nitrogen and amino nitrogen was that of Yemm and Willis (1956) and as described by Haroun (1985).
For protein banding pattern, the electrophilic protein profile of wheat seedling were analysed by SDS-PAGE technique (Laemmli, 1970). Data were analysed and identified by gel documentation system (GDS) which compared polypeptide maps; molecular protein markers, percentages of band intensity, molecular weight of each polypeptide in relation to standard markers using Gel-Pro Analyzer version 3 Media Cyberne Tice Imaging Experts Software.
For ions analysis (K, Na, Ca, Mg and P), a known dry weight was digested by sulfuric acid- hydrogen peroxide method. K, Na, Ca, Mg concentrations were measured by atomic absorption spectrophotometer using a 2380 Perkin-Elmer machine. Total phosphorous content was determined by ammonium molybdate method as recommended by Rorison et al. (1993).
Total leaf conductance and transpiration rate of the 1st fully expanded leaf were measured using a Li -1600 M steady state Porometer. The atmospheric pressure and aperture area of the apparatus were adjusted to 101.3 kPa and 1 cm2 respectively as recommended by Ibrahim (1999).
Estimation of RWC was based on the methods described by Ritchie et al. (1990) and Pardossi et al. (1992).
Statistical analysis
Statistical analysis (ANOVA and correlation coefficients) were carried out using SPSS programme. A test for significant differences between means at P= 0.05 was performed using LSD test.
Chemical analysis of cheese-whey constitutions
Analysis of the chemical constitutions of the used cheese whey (Table 1) indicated that water content represent 89.6% while total solids and lactose are 10.4 and 5.9% respectively. These findings were not greatly different from the results of Abdel-Rahman and Abo-Hamed (1992) who reported that the water content and total solids of cheese whey were 88.7 and 11.3% respectively.
| Table 1: | Chemical analysis of cheese-whey constitutions |
 | |
Changes in growth criteria and photosynthetic pigments
The effect of grains soaking in cheese whey on growth criteria of wheat seedlings appeared to depend on the used concentration (Fig. 1). Whey treatment at 50% level significantly increased shoot length, shoot fresh and dry mases and total leaf area of wheat plants as compared with control plants. The other used whey concentrations (25 and 100 %) had no significant effect on growth criteria. This is compatible with the findings of Prasad et al. (2000) who found that the germination rate of presoaked maize and cowpea seeds in cow’s whey (for 18 h) was higher than control. In addition, Konar and Arioglu (1987) found that whey treatments after soybeans sowing greatly increased nodules number and seed yield. The increase in dry weight of wheat seedlings in response to whey treatment is attributed to the increase in the dry matter accumulation maintained by increasing photosynthetic output (Fig. 2) and protein synthesis (Fig. 4 and Table 2). The massive increase in the total cumulative leaf area production in response to 50% whey treatment (Fig. 1) could be probably due to the rapid rate of movement of nutrients and hormones transported with transpiration stream from the root to shoot and this hypothesis becomes clear particularly after looking to the stimulative effect of whey on rate of transpiration (Fig. 5). Moreover, the beneficial effect of whey on plant growth could be related to whey nutrient constituents e.g Ca, K, N and soluble sugars (Sharratt et al., 1962) or improved GA3 level in treated plants (Cihangir ad Aksz, 1996).
Grain presoaking in whey significantly improved total chlorophylls and total pigments content of wheat plants. As the concentration of whey increase there was a simultaneous increase in pigments contents of treated plants (Fig. 1). This increase may explained on the fact that whey induced gibberellin biosynthesis which inhibit chlorophyllase activity (Drazkiewicz, 1994 and Cihangir and Aksz, 1996) and/or increased rate of transpiration which might have increased the rate of translocation of minerals especially Mg (Fig. 5). All used concentrations of whey not virtually affected carotenoids concentrations of wheat plants. A non significant correlation (r= 0.43) was realized between shoot dry mass and total chlorophylls concentration and this indicated that only certain concentrations of whey activate the biochemical reactions of photosynthesis possibly via the effect on the expression of some genes.
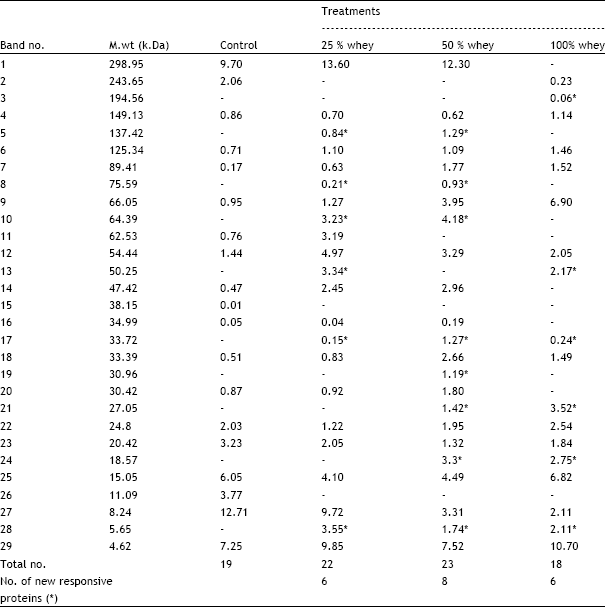
| Table 2: | Effect of different whey concentrations on molecular weight and relative concentrations of different types of protein bands of wheat seedlings |
 | |
Changes in Hill reaction and photosynthetic assimilation of 14CO2
Hill activity was improved in wheat seedlings in response to whey treatments and this effect increased with increasing whey concentration (Fig. 2). The stimulative effect of whey on Hill activity may probably due to the noticeable increase in chlorophyll biosynthesis as a result of whey treatment. In this respect, Aldesuquy and Gaber (1993) found that the increase in chlorophylls content was associated with an increase in Hill activity of Vicia faba plants under various conditions.
Whey treatment at 50% level significantly improved the soluble and insoluble fractions and consequently total 14C photoassimilates of wheat seedlings as compared with control plants (Fig. 2).
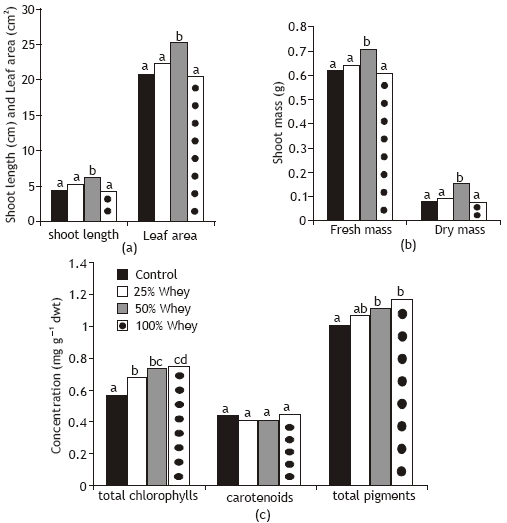 | |
| Fig. 1: | Effect of different whey concentrations on growth critaria and photosynthetic pigments content of wheat seedling * Bars in grouping leveled with the same letter(s) are not significantly different at P=0.05 |
These photoassimilates were positively correlated (r= 0.72-0.87; P < 0.05) with shoot dry weight. The increase in CO2 assimilation may result from changes of stomatal openings, biochemical reactions of photosynthesis or assimilates translocation (Wise et al., 1990 and Arnon and Gupta, 1995). The ratios of soluble to insoluble intermediates were reduced with whey treatments. A negative correlation (r= -0.79; P < 0.0 5) was detected between soluble/ insoluble ratio and shoot dry weight. These results indicated that grain priming in cheese-whey at 50% level increase the rate of transformation of soluble photosynthates to the insoluble ones and this change was in close parallel with the changes in leaf area and dry weight.
Changes of carbohydrates and nitrogen concentrations
As compared to control plants, reducing sugars, polysaccharides and total carbohydrates content of wheat shoot was increased in response to whey treatments particularly at 50% level (Fig. 3).
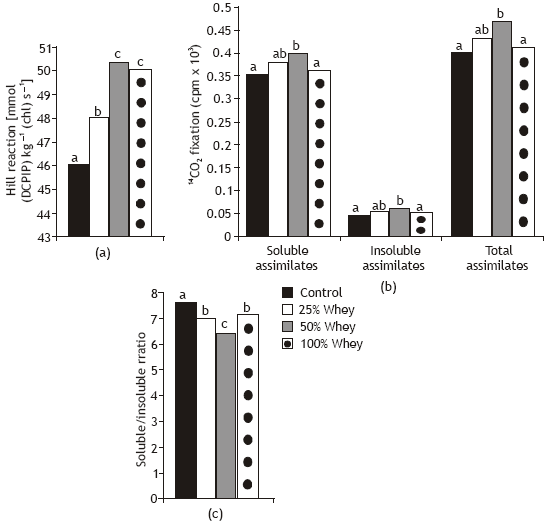 | |
| Fig. 2: | Effect of different whey concentrations on Hill reactions and 14CO2 photoassimilation of wheat seedlings |
On the other hand, whey treatments at 25 and 100% had no significant effect on carbohydrates content of wheat seedlings. The resulting increase in pigments formation in wheat leaves (Fig. 1) led to a consequent increase in the photosynthetic activity (Fig. 2) which in turn led to more accumulation of carbohydrates in the plant. This increase would be advantageous in terms of carbohydrate reserve for plant growth (Chaves et al., 1995).
Whey concentration at 50% significantly increased total N, total amino N and protein N concentration of wheat plants in relation to control plants. On the other hand, total soluble N was not affected by this whey treatment. These changes in nitrogen content may be attributed, in part, to an inhibition of proteolytic activity and, in part, to stimulation of protein synthesis (Fig. 4 and Table 2) in response to whey treatments. A positive correlations appeared between total carbohydrates and nitrogen content (r= 0.9; P < 0.01) and shoot dry weight.
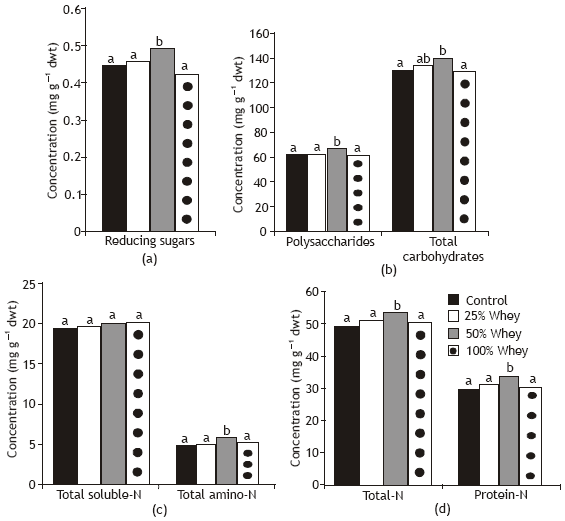 | |
| Fig. 3: | Effect of different whey concentrations on carbohydrates and nitrogent contents of wheat seedlings |
These results are in good conformity with the findings of Martin et al. (2002) who found that nitrogen and carbohydrate status play the predominant role in regulating various aspects of seedling growth including storage reserve mobilization and photosynthetic gene expression.
Changes in protein banding patterns
Scanning of the gel indicate the occurrence of 19 protein bands in control seedlings ranging from 4 to 298 kDa, whereas whey treatments at 25 and 50 % increased the protein bands to 22 and 23 bands respectively and grains soaking in 100% whey decreased the protein bands in wheat seedling to 18 bands (Fig. 4 and Table 2). Shoot dry weight was positively correlated (r= 0.76, P < 0.05) with the number of protein bands. Furthermore, whey at 25 and 50% levels increased the relative protein concentrations in wheat seedlings and the effect was more elicited with 47- 89 kDa polypeptides. This finding was consistent with those obtained by Sharratt et al. (1962) who found that whey treatment increase protein content of shelled corn from 8.5 to 11%.
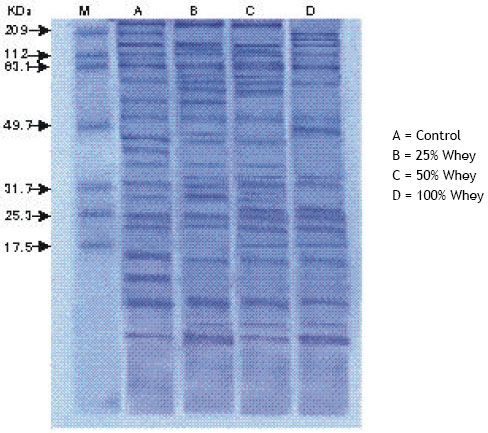 | |
| Fig. 4: | Effect of different whey concentrations on protein patterns of wheat seedlings |
On the other hand, grain priming in 100% whey reduced the relative protein concentration of wheat seedlings as compared with control seedlings and the effect was clear with 243-298 kDa polypeptides. The appearance of new protein bands with molecular weight 33 and 5 kDa in response to the three different whey concentrations could be considered as treatment-specific protein (King, 1991). Furthermore, the lower molecular weight protein (5kDa) are known to have a role during seed germination (Waters et al., 1996). These qualitative and quantitative changes in the pattern of protein synthesis with whey treatments suggested that new genes might be transcribed and others might be repressed or, at least, the expression of some genes were increased or decreased (Avise, 1994; El-Araby and Hegazi, 1999).
Changes in ions content and water relations
Grain priming with 50% whey significantly increased K, Ca and P concentrations and had non significant effect on Na concentration of wheat seedlings (Fig. 5). In this regard, Sharratt et al. (1962) documented that whey treatment increase nitrogen, phosphorous and potassium content of shelled corn. On the other hand, the effect of the other whey concentrations (25 and 100 %) generally was not significant for these ions. Magnesium concentration appeared to increase in wheat with increasing whey concentration as compared with control plants. These changes in magnesium content were consistent with the observed changes of chlorophylls concentration and could reflect the role of Mg ion in chlorophyll synthesis (Schoefs and Bertrand, 1997).
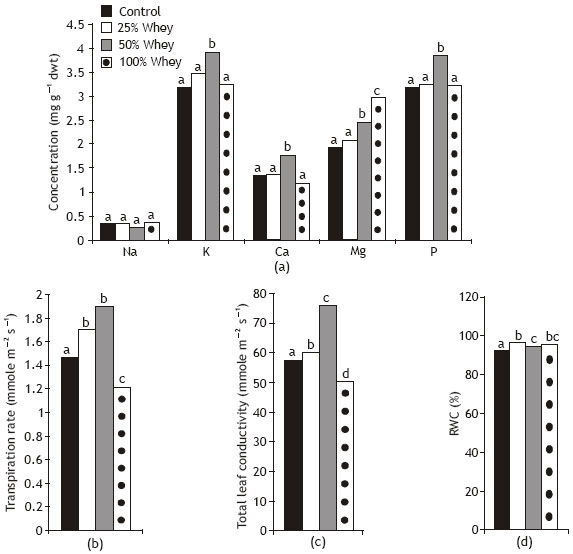 | |
| Fig. 5: | Effect of different whey concentrations on ions content and water relations of wheat seedlings |
Shoot dry weight was positively correlated with K, Ca and P content (r= 0.91-0.94; P < 0.01) and non- significantly correlated with Na and Mg content. The positive effect of whey on ions content of wheat seedlings may presumably be due to the increase in ATP content, activation of ATP ase of plasma membrane and consequently the stimulation of K carriers bound to plasma membrane (Dai et al., 1998).
Transpiration rate and total leaf conductivity of wheat seedling were significantly increased in response to whey treatments at 25 and 50 % levels (Fig. 5). On the other hand, grains soaking in 100% whey appeared to reduce these criteria in wheat plants in comparing with control plants. These results are in accord with the study of Abo-Hamed (1994) who found that grains soaking in 50 mM CaCl2 decreased stomatal resistance, while grains soaking in 100 mM CaCl2 increased the stomatal resistance of sorghum leaves when compared by grains soaking in distilled water. Shoot dry weight was positively correlated with transpiration rate (r= 0.79; P < 0.05) and total leaf conductivity (r= 0.94; P < 0.01) of wheat seedlings. The increase in transpiration rate of wheat plants would be advantageous for mineral nutrient uptake to compensate the requirements of increased growth (Kramer and Boyer, 1995). These changes in transpiration rate and total leaf conductivity may indicate that whey treatments regulate K+ and ABA concentration in guard cells, but the lack of data about the absolute values of K+ and ABA in the guard cells make this postulation not decisive.
Grains priming in cheese whey significantly increased relative water content (RWC) of whey seedlings. A non significant correlation was realized between RWC and shoot dry weight. This increase in seedlings water content was mainly due to increased water absorption that led to powerful water supply to shoot (Ritchie et al., 1990; Kramer and Boyer, 1995).
It is concluded from this study that soaking of wheat grains in cheese whey at 50% level improved seedlings growth vigour by inducing some modifications in the photosynthetic characteristics, carbohydrates, nutrient, protein patterns and water status of treated plants.