Research Article
Effect of Temperature and pH on the Spectral and Conformational Distribution of Adsorbed Lysozyme
Ebonyi State University, Nigeria
C. E. Ekuma
Ebonyi State University, Nigeria
A. E. Umahi
Ebonyi State University, Nigeria
Protein adsorption plays a major role in a variety of important technological and biological processes (Denizli et al., 2000; Ghose and Chase, 2000; Hlady and Buijs, 1996; Shi and Ratner, 2000). Adsorption of proteins unto solid surfaces is a process that influences phenomena within many disciplines and as diverse as fouling of process equipment in the food industry, blockage of filtration membranes in bioseparation processes and regulation of immuno-response to foreign materials (Wahlgren and Arnebrant, 1990).
The understandings of the fundamental factors that determine protein adsorption are imperative to improve our ability to design biocompatible materials, biotechnological devices and coatings. There is no general rule for this but it is vital one understands the driving forces for the protein adsorption process and the effect of adsorption on the protein conformation and activity. Also, the understanding of the chemistry of the surfaces is of great importance for the adsorption kinetics as protein adsorption is a very important fundamental problem that involves large competing energy scales and conformational statistics that may result in reversible and irreversible processes (Fang and Igal, 2001).
The amphipathic nature of proteins, with their polar, non-polar and charged amino acids, also contributes to surface activity. Although hydrophilic polar and charged amino acids are generally located on the exterior of the molecule and hydrophobic residues on the interior, this is not absolute. Thus hydrophobic amino acids might be available on the protein surface for interaction with substrates. Additionally, unfolding of proteins can expose hydrophobic regions and allow interaction with the surface. One of the most important contributions to the understanding of the kinetics of protein adsorption is the Random Sequential Adsorption (RSA) model (Schaaf and Talbot, 1989). In this approach, the proteins are assumed to be rigid particles that interact only through excluded volume interactions. The particles are assumed to irreversibly adsorb to the surface and, thus, they do not have translational degrees of freedom or desorption on the surface. This model has been very useful in understanding why the kinetics of protein adsorption does not follow the Langmuir predictions. This model has been extended to consider conformational changes, desorption and the treatment of mixtures (Van Tassel et al., 1998). However, its major limitation is that it is hard to include detailed molecular information of the proteins and the formulation is based on a kinetic approach. Other models have been predicted like determination of adsorption kinetics by the diffusion of the proteins to the surface (Iordanskii et al., 1996) and that based on the minimization of free energy of the system, which provides the equilibrium state of the system and, thus, protein adsorption isotherms can be studied (Fang and Igal, 2001).
Dissolved protein molecules adsorb at almost all interfaces. There are several applications of this adsorption phenomenon, such as solid phase immunoassays for medical diagnostic tests, biosensors and immobilized enzymes in bioreactors. However, protein adsorption on interfaces have some undesirable consequences thus, an understanding of the protein adsorption process on a molecular level and a determination of the structure of protein adsorbates are necessary to optimize these applications or to prevent adsorbed proteins if they are undesired (Jackler et al., 2002).
The adsorption of proteins on surfaces is a complex phenomenon. The adsorbing particles are large and, thus, the surface-protein interactions are usually long range and the strength is many times the thermal energy. Furthermore, due to the large size and the shape of the particles, the interactions between the adsorbed particles on the surface are nontrivial and can be strongly influenced by the fact that the particles may undergo conformational changes upon adsorption (Fang and Igal, 2001; Nasir and McGuire, 1998; Norde and Giacomelli, 2000).
A common way of portraying the results of adsorption studies on solid surfaces is in the form of an adsorption isotherm, which gives the amount of adsorbed material per surface area at a constant temperature as a function of pressure or concentration in the bulk phase. The isotherm shows the extent of the adsorbed layer, that is, the net result of these two competing processes. The association of two freely translating and rotating molecules to form a complex, in the gas phase or in solution, generally involves a loss of entropy. Thus, the complex will survive as stable species only if its formation is favoured on enthalpic grounds. That is, the potential energy of the bound complex must be lower than that of the well-separated molecules. In solution it is the potential of mean force that must be lower (Ben-Tal et al., 2000).
For a complete characterization and prediction of protein adsorption, one seeks the information about adsorption isotherms, adsorption kinetics (in situ), conformation of adsorbed proteins (Andrade, 1985), number and character of surface bound protein segment and the physical parameters describing the adsorbed protein layer (Norde, 1986). All these information can be obtained using diverse protein adsorption experimental techniques. The combination of such information is a vital tool for predicting the mechanism of protein adsorption unto and desorption from surfaces of which one approach to this problem is the analysis of protein adsorption kinetics (József, 2002).
The polyelectrolytic nature of proteins enhances adsorption mainly via coulombic interaction between the charged surface and the oppositely charged amino acids (proteins). For instance, on a hydrophilic solid surface, the electrostatic interaction between a charged surface and an oppositely charged protein molecule is often the driving force for adsorption from solution unto solid surfaces. The stability of these electrostatic interactions within the adsorbed layer may be a major determining factor for amount adsorbed. A further driving force for protein adsorption is entropic changes usually associated with dehydration of the protein or a structural rearrangement of the protein molecules. Strong surface interactions may damage the original state of a protein molecule and this may lead to a loss of its coherent structure (denaturation). The surface and the relative stability of the protein structure determine the extent of such structural rearrangement on adsorption. Adsorption is an important feature of surface reactions, such as corrosion and heterogeneous catalyst and the property is also utilized in adsorption chromatography (Alan, 2000).
In this study, hen egg white lysozyme is used as a model protein. There are yardsticks for choosing this protein. For instance, it is a globular protein with well-defined molecular structure and its stability is likely to prevent complete unfolding at a surface. This suffices to say that the structure of lysozyme and the unfolding behavior of dissolved lysozyme are well characterized. Again, at moderate pH values, it has a high thermal stability, which has its origin in part from four disulphide bonds.
The adsorbents used for this study are silica particles, a non-synthetic surface and polystyrene particles, a synthetic surface. The equilibrium and non- equilibrium adsorptions of the protein were carried out. The effect of varying temperatures on the spectra distribution of adsorbed lysozyme was studied and an overall picture of protein adsorption kinetics carefully examined in the light of varying pH.
The main aim of this study is to investigate the relation between the thermodynamic stability and the degree of adsorption of hen egg white lysozyme at the silica/water and polystyrene/water interfaces as a consequence of maximum absorbance at wavelength, 750 nm, equilibrium concentration, amount adsorbed per 1 dm3 all at varying pH values; and to investigate the spectra distribution of the adsorbed lysozyme on both surfaces as a function of varying temperature. Instead of studying different proteins varying in stability, the stability of lysozyme was changed by varying the temperature, which has the advantage of keeping protein-surface interactions nearly unchanged. In addition, the contribution of adsorption-induced conformational changes to the driving kinetics for the adsorption of lysozyme was investigated. The outcome of this study is expected to be of uttermost importance in material selection for filters in the optical wavelength of the electromagnetic spectrum.
In addition to silica/water and polystyrene/water interfaces investigation, lysozyme adsorption studies have been carried out at the air/water and quartz/water interfaces by Vibrational Sum Frequency Spectroscopy (VSFS) and this showed evidence for water structural rearrangement at both interfaces upon lysozyme absorption (Gibum et al., 2002). The automated Rudolph thin film ellipsometry equipped with a thermostated curvette study showed that lysozyme adsorption from binary mixtures of oppositely charged proteins is affected by protein -protein interactions (Whalgren et al., 1992).
In this study, we have employed the optical spectroscopy to investigate the adsorption of lysozyme onto water/silica and water/poystyrene interfaces. This technique is particularly important because, it is capable of monitoring the restructuring of interfacial water molecules upon the surface conscription of a variety of adsorbates even in the presence of vast contribution of bulk water.
Materials/equipment: This study was carried out in Abakaliki, Ebonyi state, Nigeria between the month of August and September 2005.
Pure crystalline lysozyme (from chicken egg white, grade 1, product No L-6876, mol. Weight 14,000 g mol-1) used in this study is a product of Sigma chemical Co., USA. Chromatographic grade silica particles (mesh size 200 m) from Burgoyne Burbridges and Co., Mumbai, India with product No. 07079 and batch no 21920 was used without any further purification. Polystyrene; a general-purpose polystyrene grade G/26 and lot. No. 1211171 FKS B1 a product of Dongbu Hannong chemical Co. Ltd, Korea. The polystyrene was crushed with manual blender to smaller particles and sieved using the standard sieve plate to obtain samples of 200 m size. During crushing, the polystyrene was mixed with glucose to prevent coagulation of the fine particles and afterwards was washed with distilled water to separate the glucose and then dried at a temperature of 120°C. All inorganic salts used were BDH chemicals and were of analytical grade.
The digital pH meter CE HI 98127 used for the analysis is a product of Hanna instruments inc., Woonsocket, Rhode Island, 02895 USA, (PH, EC /TDS water proof family) which measures to accuracy of 0.1 with an inbuilt automatic temperature control that was standardized using standard buffer solutions (4.10, 7.10 and 9.18) respectively according to specifications. Visible spectrophotometer, 722S spectrophotometer; No. SFZ 1506010514 was used for the quantification of the lysozyme. The absorbances were taken and their concentration determined from the calibration curve. Digital analytic weighing balance, X21 0014 KERN 770 15,15402301, made in Germany, which measures to an accuracy of 0.0001g and Mechanical Shaker; versal shaker, type: LE-203/1 made in Hungary were the equipments and materials used throughout the course of this experimentation.
Sample preparation: Five different buffers of pH 5.0, 7.0, 10.0, 11.0 and 12.0 were prepared with each having ionic strength of 0.01.
| • | For pH 5.0, 8 mL of 0.1M solution of acetic acid was mixed with 35.2 mL of 0.1M Sodium acetate. |
| • | For pH 7.0, 100 mL of 0.05M solution of KH2PO4 was mixed with 55 mL of 0.05M NaOHF. |
| • | For pH 10.0, 160 mL of 0.025M Na2HPO4 was mixed with 5 mL of NaH2PO4. |
| • | For pH 11.0, 100 mL of 0.025M K2CO3 was mixed with 43 mL of 0.025M KHCO3. |
| • | For pH 12.0, 100 mL of 0.05M Na2HPO4 was mixed with 53.8 mL of 0.05M NaOH. |
Experimentation: The lysozyme (protein) to be tested was dissolved in the buffer used to prepare the test solution. Portions of this solution were diluted with the same buffer to obtain seven standard solutions having concentrations range of 0.1 to 1.4 g L-1, with the concentrations evenly spaced. The initial time of addition of the protein to the standard flasks was noted. Afterwards the flasks were shaken vigorously and, at different intervals of elapsed time, each of the flask was taken out of the shaker and the particles allowed to settle, filtered before withdrawing 1.0 mL of the protein solution. Using Folin-Ciocalteu reagent, the concentration of the protein solution after adsorption at a time t was determined spectrophotometrically in the light of Lowry method. The flasks were shaken for 16 h to attain equilibrium and at the end of this period, the lysozyme concentration (Cp) in the bulk solution (normally read off from the calibration curve) was determined by the spectrophotometry using the normal procedure.
To determine the amount of protein adsorbed at equilibrium, the flasks were shaken at constant temperature of 30°C for 16 h and then kept undisturbed for 4 h. This was done to ensure equilibrium attainment and for complete protein adsorption anticipated within this period. The procedure was also repeated at lower and higher temperatures of 20°C and 40°C at pH 7.0. The amounts adsorbed at different intervals of the elapsed time as a function of temperature were calculated.
To enable the study of the spectra distribution of the adsorbed lysozyme on both silica and polystyrene surfaces, the experiment was carried out at various pH and temperatures. Also, buffer solutions of varying pH, 5.0, 7.0, 10.0, 11.0 and 12.0 were prepared as discussed above to enable determination of the effect of pH. 0.4 g of each of the pure silica and polystyrene was added to each of the five flasks (double set) containing 10 cm3 of 7.143x10-5M lysozyme solutions at an ambient temperature of 30°C. The effect of temperature on the adsorption equilibrium of lysozyme was studied by varying the temperature from 20 to 50°C using the same initial concentration above and at pH 11.0; the isoelectric pH of lysozyme as to enable the study of the spectra distribution.
The present study was carried out at various pH values and temperatures. To determine the effect of pH, buffer solutions of varying pH values; 5.0, 7.0, 10.0, 11.0 and 12.0 were prepared as discussed earlier. The effect of temperature on the adsorption equilibrium of lysozyme was studied by varying the temperature from 20 to 50°C using the same initial concentration above and at pH 11.0, the isoelectric pH of lysozyme.
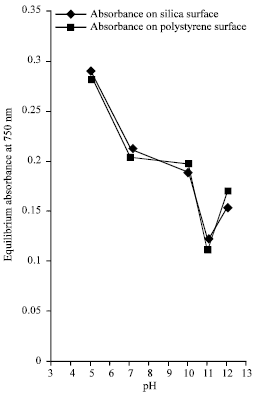
Figure 1 represents the graph of absorbance at wavelength, 750 nm against pH values for the two surfaces. Figure 2 is the graph of equilibrium concentration of lysozyme against pH values for the two surfaces.
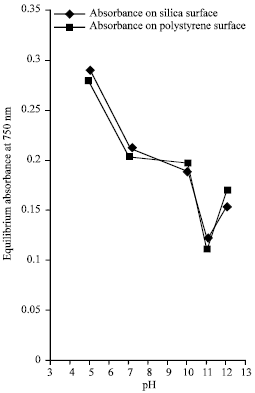 | |
| Fig. 1: | The graph of equilibrium absorbance at wavelength of 750 nm at varying pH values |
 | |
| Fig. 2: | The graph of equilibrium concentrationx10-5 (mol L-1) at varying pH values |
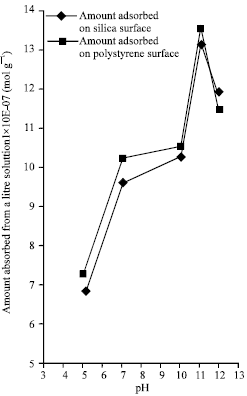
Figure 3 is the graph of adsorbed lysozyme per 1 dm3 against pH values for the two surfaces; Figure 4 represents the graph of adsorption density against pH values for the two surfaces; while Fig. (5-6) shows the graph of absorbance against maximum wavelengths for adsorbed lysozyme on silica and polystyrene surfaces.
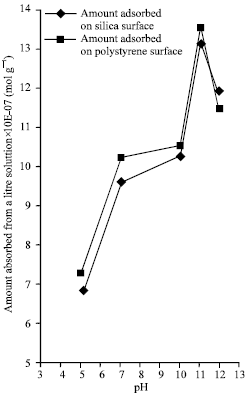 | |
| Fig. 3: | The graph of the amount of adsorbed lysozyme from 1 dm3 Solutionx10-7 (mol g-1) at varying pH values |
 | |
| Fig. 4: | The graph of the adsorption density (mol m-2) of adsorbed lysozyme at varying pH values |
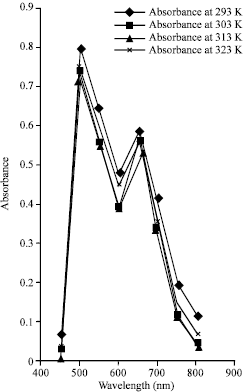 | |
| Fig. 5: | The graph of absorbance against maximum wavelengths for adsorbed lysozyme on silica surfaces |
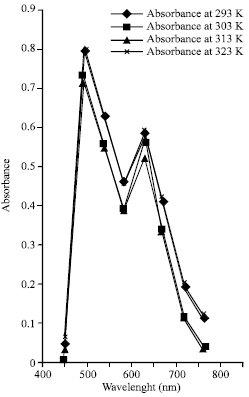 | |
| Fig. 6: | The graph of absorbance against maximum wavelengths for adsorbed lysozyme on polystyrene surfaces |
From Fig. 1 and 2, it can be inferred that the minimum equilibrium absorbance for silica was 0.123 and 0.114 for polystyrene at the same pH of 11.0 with increase on both side of the isoelectric pH while minimum equilibrium concentration was 1.856x10-5 mol L-1 for silica and 1.713x10-5 mol L-1 for polystyrene at the same isoelectric pH of lysozyme with increase on both side of the pH. The conclusion that can be drawn from this observation is that minimum equilibrium absorbance and minimum equilibrium concentration for silica and polystyrene interfaces occurs at the isoelectric pH of lysozyme. A cursory look at Fig. 3 show that maximum amount of adsorbed lysozyme of 13.203x10-7 mol g-1 for silica surface and 13.569x10-7 mol g-1 for polystyrene at the same pH of 11.0 which is the isoelectric pH of lysozyme. Also, it can be inferred from figure 4 that as the pH value increases, the adsorption densities of lysozyme on both surfaces increased rapidly reaching a maximum at 12.082x10-9 mol m-2 for silica and 5.879x10-9 mol m-2 for polystyrene at a pH 11.0; the isoelectric pH of lysozyme with a decrease on either side of the isoelectric pH for both surfaces. This behavior shows that lysozyme exhibits maximum adsorption in the neighborhood of its isoelectric pH. This is mainly due to entropic contributions originating from the dehydration of hydrophobic surface areas and adsorption-induced conformational changes of the protein.
Also it was observed that the low adsorbed amount onto silica at pH 5.0 and 7.0 is attributed to the net positive charges in silica at those pH values. The electrostatic repulsion between the charged lysozyme solution and the charged silica surface is the consequence of the decreased rate of adsorption.
In general, lysozyme exhibits a maximum adsorption density near or at the isoelectric pH.
The examination of the contribution of the adsorption-induced conformational changes to the driving kinetics for the adsorption of lysozyme indicates an endothermic and thus an entropy-driven adsorption process. Raising the temperature has the effect of destabilizing the native lysozyme structure. As a result, adsorption-induced conformational changes occur, which will serve as an entropic driving force for the adsorption process. Although dissolved lysozyme is unfolding at 70-78°C, pH 7 (Jackler et al., 2002), the structure profile of the lysozyme adsorbate at 30°C for silica surface appears to be similar to that at 30°C for polystyrene surface, which supports the conclusion that at the ambient temperature, lysozyme molecules must undergo major conformational changes upon adsorption. This finding is consistent with a recent fluorescence spectroscopy study, in which the temperature of unfolding of lysozyme was found to be decreased due to adsorption on silica particles (Czeslik and Winter, 2001).
From the trend of the observed parameters, it can be concluded that conformational changes in the adsorption of lysozyme onto Silica/Water and Polystyrene/Water interfaces occurs within the isoelectric point of lysozyme where major interaction involved in lysozyme (protein) adsorption is hydrophobicity, which may be due to entropic changes associated with dehydration of the protein or a structural rearrangement of the protein particles and adsorption-induced conformational changes of the protein. This correlates with the observation of Yoon et al. (1998), which showed that hydrophobic interaction is the most important aspect of protein adsorption and increased hydrophilicity results in a low level of adsorption. The slight increase in adsorption with increasing pH value on the polystyrene surfaces shows that the surface is inert. Evidently, silica adsorbed more lysozyme, with dimensions close to its pore size, than polystyrene, supporting its potential for size selective separations and possible application in protein separation. This is in agreement with the earlier study of Kisler et al. (2001) on Adsorption of Proteins on Mesoporous Molecular Sieves.
The investigation of the spectra distribution of the adsorbed lysozyme on silica interface showed markedly uniform disparity in absorbance within wavelengths (450 to 500 nm) for a given temperature. The maximum absorbance occurred at 0.8020 when the wavelength was 500 nm and decreased sharply to 0.4010 at wavelength of 600 nm. The absorbance had maximum value 0.5970 again at the wavelength 650 nm, after which it decreased to about 0.0410 at the wavelength of 800 nm. It can also be inferred that the adsorbed lysozyme on polystyrene surface showed the same behavior like that of silica, though; the maximum absorbance was more for that of silica. From this result, one can infer that these materials could be used as filter material within the optical window of the electromagnetic spectrum and possibly, as biosensors for biocompatible devices.
This study indicates that the combination of increased lysozyme binding capacity and improved stability upon extended contact (with aqueous solution) makes silica (a non-synthetic material) an effective adsorbent material for the removal of lysozyme from aqueous solution than polystyrene (synthetic material). The adsorption of lysozyme onto silica is found to be time, pH and temperature dependent. The maximum amount adsorbed, the minimum equilibrium absorbance, minimum equilibrium concentration and the amount adsorbed per dm3 of lysozyme occurs between the pH ranges of 10 to 12 for both surfaces which coincides with the isoelectric pH range of lysozyme. This behavior shows that lysozyme exhibits the above outlined behavior within the range of its isoelectric pH. The major interaction at this pH is hydrophobicity, which is due to entropic changes associated with the dehydration of the lysozyme molecule or a structural rearrangement of the protein.
The spectra distribution shows that the adsorbed lysozyme on silica and polystyrene surfaces could be used as filter materials in the optical region since maximum absorbance occurred around 500 nm a typical wavelength range of visible light.
The authors are most grateful to God for His mercies all these years and to their families for their patient during the course of this study.