Research Article
Comparison of Sunflower (Helianthus annuus L.) Hybrides by using Isozymes and Seed Storage Protein
Esfahan Payamenoor University, Esfahan, Iran
Fazilati Mohammad
Esfahan Payamenoor University, Esfahan, Iran
Seed identity and varietal purity testing are essential components of a modern and effective agricultural production system. Genetic purity of a seed sample defines the percentage of the sample that is not contaminated by seeds or genetic material belonging to other varieties or species. A combination of laboratory and field plot methods may be used to determine the cultivar trueness and genetic purity of the sample. Laboratory control is based on protein markers, isozymes, seed storage proteins and molecular markers. The development of isozyme and/or DNA databases is a prerequisite to varietal identification and protection. Isozyme marker loci have been available for use in quality assurance, variety identification, plant breeding, production and variety protection programs for more than three decades. They are especially useful in solving problems in seed science such as inadvertent mixing of hybrid seeds or lines, uncontrolled pollination, errors during multiplication, etc. (Geric et al., 1989; Zlokolica et al., 1996). Electrophoresis of seeds storage proteins (helianthinin) of sunflower shows promising results in genetic purity determination of sunflower hybrids and inbred lines (Anisimova and Gavrilyuk, 1989; Aksyonov, 2005a, b). Molecular markers offer a powerful supplement to the morphological and disease resistance data currently used for variety protection, cultivar classification schemes and estimation of the level of genetic diversity. Molecular markers have many advantages (Lombard et al., 2000) compared with morphological markers, resilient to environmental changes, nearly unlimited number and relative ease and rapidity of data collection. According to the Association of Official Seed Certifying Agencies (AOSCA, 2003), maximum limits for seed of other varieties or off-types found in seed lots range from 0 to 0.2% among different species, while the limits for certified seed range from 0.1-2% by weight. In practical terms, the maximum number of seeds of other varieties of the same crop permitted in 0.454 kg of certified seed is 6 for sunflower . Minimum genetic purity is 99.5% for female lines, 99.8% for male lines and 95% for sunflower hybrids (OECD Seed Scheme, 2006). According to the Official Gazette of SFRY (No. 47, 1987), genetic purity can be tested for scientific research purposes or for controversial cases. Hybrid seed is considered as satisfying if it contains no more than 5% of self-pollinated individuals. The aim of the present study was to compare two methods which are commonly used for estimation of genetic purity in sunflower hybrids, electrophoresis of isozymes and seed storage protein-helianthinin and to estimate the applicability of a modified method for preparation of storage proteins from sunflower seed.
Plant material: Three sunflower hybrids LottoR28, R101, Lotto3620, were analyzed in this study have been generated in National Institute of Genetics and Biotechnology, NIGEB, Iran. Preparation of helianthinin solution was modified from Samarah et al. (2006). Each seed was finely ground and protein extracted from flour by adding 1000 μL 50 mMm Tris-HCl buffer pH 7.4 containing 2.5% 2-mercaptoethanol and heated in 90°C for 10 min. After centrifugation at 3000 to 5000 rpm for 15 min, the supernatant was used for electrophoresis. Individual seeds were tested from each sample. Polypeptides were resolved by electrophoresis of proteins under denaturing (SDS) and reducing (2-mercaptoethanol) conditions in 12.5% PAGE using the method of Laemmli (1970). Electrophoresis was performed at an initial voltage of 50 mV, reduced to 25 V when the tracking dye reached the gel mold; analysis time was 16 h. The marker wide molecular weight range (17.200-77.0 kD) was used for determination of protein molecular weight in electrophoretograms. Proteins were simultaneously fixed and stained using a solution containing 0.24 g Coomassie Brilliant Blue R250 in 90 mL of a 1:1 (V/V) methanol: water and 10 mL of glacial acetic acid. Stem tissues of 5 day old seedlings homogenized in 50 mM TrisHCl, pH 6.8, in which 1% mercaptoetanol was added, were used for isozyme analysis. Isozyme systems Phosphohexose Isomerase (PHI), Phosphogluconate Dehydrogenase (PGD), Phosphoglucomutase (PGM), Malate Dehydrogenase (MDH), Isocitrate Dehydrogenase (IDH) and Acid Phosphatase (ACP) were analyzed according to Stuber et al. (1988).
RESULTS AND DISSCUSION
The main storage proteins in sunflower make up about 85% of the total protein content. Helianthinin has been reported to be present as a globular oligomeric protein with a molecular weight of 300-350 kDa (Sabir et al., 1973; Schwenke et al., 1979). It belongs to the cupin superfamily which is comprised of 11S and 7S seed storage proteins. The members of the 11S family include pea and broadbean legumins, rape cruciferin, rice glutelins, cotton β-globulins, soybean glycinins, sunflower helianthinin, etc. Comparison of the results of Lakemond et al. (2000), Lakemond (2001) and Gonzalez-Perez et al. (2004) clearly reveals many structural similarities of the soy 11S fraction (glycinin) and helianthinin. Starting from this fact, we used in our study a modified method for extraction of storage proteins from soybean (Samarah et al., 2006) (Fig. 1).
This method efficiently extracted proteins from sunflower seeds helianthinin showed subunit heterogeneity, in addition to the presence of multiple subunits within a single genotype, there were also differences in the SDS-PAGE patterns of helianthinin components between different cultivars (Raymond et al., 1994, 1995).
 | |
| Fig. 1: | Electrophoretogram of seed storage protein of hybrids, A: Ladder, B: Lotto3620, C: Lotto101 and D: LottoR28 |
The number of polymorphic zones corresponds to the number of subunits composed of the helianthinin molecule, controlled by Hel1, Hel2 and Hel3. The analysis of helianthinin revealed three zymogram patterns within and between the hybrids (Fig. 1 and 2).
In Fig. 1, there are two evident features: only the high molecular weight proteins (higher than 30 kDa) produced high intensity bands while the low molecular weight proteins were present in relatively low concentrations. All 6 analyzed enzymatic systems (MDH, PGM, PHI, PGD, IDH, ACP) were polymorphic, with two-or three-allele variants. The obtained differences in zymograms of the enzymes are useful for estimation of the genetic purity of sunflower hybrids. Similar results of polymorphism were reported by Chikkadevaiah and Nandini (2003). The comparative analysis of genetic purity level of the sunflower hybrids showed that the methods of electrophoresis of isozymes and seed storage proteins were in agreement in most cases. Differences in genetic purity level ranged from 1 to 5% in 81% of the samples. However, in the case of sample 1, the isozyme and seed storage protein analyses showed the values of 98 and 87%, respectively while in the case of sample 11, therespective values were 89 and 97% (Table 1).
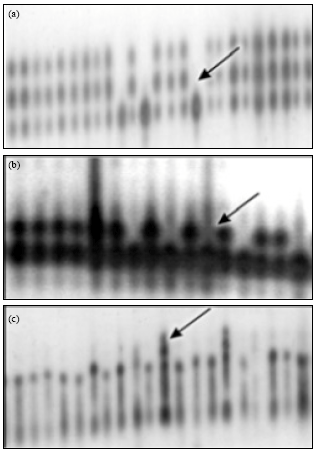 | |
| Fig. 2(a-c): | Isozyme patterns of sunflower hybrids(Helianthus annuus) (a) PHI, (b) PGM and (c) PGD |
| Table 1: | Comparative data of genetic purity level in sunflower hybrids measured on the basis of isozyme and seed storage protein analysis |
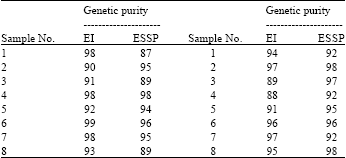 | |
| ESSP: Electrophoresis of seed storage protein, EI: Electrophoresis of isoenzymes | |
The main sources of error in the analytical methods used were sampling and reduction of samples in the laboratory. Both the sample size and sampling procedure dramatically impact the conclusions that may be drawn from any of these testing methods. Analytical results can also be influenced by type of sample, seedling or seed. In the isozyme analysis, seedlings of germinated seed were tested while in the analysis of seed storage proteins any form of seed sample was applicable. Reproducibility, sensitivity and specificity of results was critical for validation of the methods used.
In a study of sunflower genetic purity using field trials and electrophoresis of storage proteins (helianthinins), Aksyonov (2005a) concluded that albumin markers allowed to define the genetic homogenity level of hybrids at 80%. Our results showed that the level of genetic purity of sunflower hybrids could be accepted as satisfactory in most cases (OECD Seed Schemes, 2006). Seed storage proteins and isozymes cannot be used for genetic identification of sunflower hybrids due to their low polymorphism levels. Loss of genetic purity or varietal changes can occur due to various reasons: Handling methods, storage facilities, natural crosses, genetic mutations, random genetic drift and other selection factors. Therefore, no commercial seed is one hundred percent genetically or mechanically pure. The results on the genetic purity of sunflower seed obtained by Shabani et al. (2013) showed that increased attention should be paid to seed genetic purity and systematic control of genetic uniformity of parent components, lines, hybrids and varieties.
Seed certification programs and seed production companies are required to achieve various levels of seed purity for different species. The results of genetic purity level of the sunflower hybrids produced by the two widely used methods, seed storage proteins and isozyme, were in agreement in most cases. The differences ranged from 1 to 5% in 81% of the samples. The obtained results validate the two methods and could be useful for further study. The methods for genetic purity testing that are presently in use need to be standardized.
The authors acknowledged the support from National Institute of Genetic Engineering and Biotechnology (NIGEB), in Iran For providing the necessary research facilities.