ABSTRACT
Arbuscular mycorrhizal fungal spores of Glomus spp., isolated from heavy metal-polluted and corresponding unpolluted soil obtained from Abou-Rawash farm, Giza (Egypt), were investigated in vitro to test their sensitivity to Zn, Cd and Pb, measured as an inhibition of the germination percentage and hyphal growth from germinated spores. The spores germination and hyphal growth for both isolates showed a variable responses to different concentrations of Zn, Cd and Pb. Only, Zn at low concentrations increased germination percentage and hyphal growth for both isolates spores compared to control (without metal) and this effect was more pronounced with the spores isolated from polluted soil. Germination of the spores isolated from polluted soil were more tolerant to Zn, Cd and Pb than the spores from unpolluted, with EC50 values of 76, 34 and 14 μ mol L‾1 for the former isolate and 30, 16 and 6 μmol L‾1 for the later to Zn, Cd and Pb respectively. Interaction between metals singly or in combination of 2 or 3 metals (at EC50 values) showed variation in sensitivity to Zn, Cd and Pb. The presence of Zn in the media significantly ameliorated toxicity of Cd and /or Pb in most circumstances as indicated by increasing hyphal growth of germinated spores. This effect was more pronounced with the spores isolated from polluted soil and in combination of 3 metal tested. It was concluded that, AM fungal spores from polluted and unpolluted soil differ in their heavy metals susceptibility to single or multiple metal exposure.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2003.1416.1422
URL: https://scialert.net/abstract/?doi=pjbs.2003.1416.1422
INTRODUCTION
In the natural environment, contamination by single pollutants rarely occurs. Man’s energy and chemical consumption, metal smelting, mining and manufacturing process more often result in environments contaminated with a mixture of potentially toxic metals. The main sources of the heavy metals contamination in the agricultural soil are fertilizer impurities and the use of refuge-derived compost and sewage disposal, municipal and industrial waste. Soil contamination with heavy metals has become a world problem, leading to losses in agricultural yield and hazardous health effects as they enter the food chain (Salt et al., 1995).
The toxicity of heavy metals in soil depends on their bioavailability, defined as their ability to be transferred from the soil to a living organism (Berthelin et al., 1995); metal bioavailability is a function not only of their total concentration but also of physic-chemical (as PH and organic matter) and biological (as biosorption and bioaccumulation) factors.
All organisms, including microorganisms, can achieve resistance to heavy metals by avoidance, when the organism is able to restrict metal uptake, or by tolerance, when the organism survives in the presence of high internal concentration (Turnau et al., 1996).
Arbuscular mycorrhizal (AM) fungi are abundant components of the soil biota in most terrestrial ecosystems. The influenced of AM fungi in plant nutrition is thought to be greater for elements such as phosphorus and heavy metals (Lambert et al., 1979). Heavy metals have been reported to inhibit spore germination and hyphal extension in vitro (McGee, 1987) and to reduce or completely eliminate AM colonization of plant roots in pot experiment (Chao and Wang, 1991). However, AM fungi occur in metal polluted soil, Gildom and Tinker (1983) demonstrated that the strain of Glomus mosseae isolated from heavy metals contaminated soil was much more tolerant to Zn and Cd than that from uncontaminated soil. These results are confirmed by Weissenhorn et al. (1993), who demonstrated that soils polluted by applications of heavy metals-contaminated sewage sludge and by atmospheric deposition from a smelter contained AM fungi more tolerant to Cd than reference strains isolated from noncontaminated soils. Griffioen et al. (1994) found tolerant AM fungal strains associated with Agrostis capillaries only at Zn/Cd contaminated sites, whereas no tolerant strains were detected at Cu contaminated sites. AM fungi spores isolated from zinc- polluted or cadmium-polluted soil proved more tolerant to cadmium than a laboratory reference strain Glomus mosseae regarding spore germination ability in sand amended with Cd solution (Weissenhorn et al., 1994).
The aim of this study was to investigate the response of two different local isolates spores of Glomus spp., one isolate obtained from polluted soil site and other from corresponding unpolluted site, to different doses of heavy metals, zinc, cadmium and lead with their interaction on germination and hyphal growth in vitro.
MATERIALS AND METHODS
Two isolate spores of arbuscular mycorrhizal (AM) fungus, Glomus spp. were used in this investigation. They were obtained from the rhizospheric soil of wiled plant Panicum turgidum Forssk grown in Abou-Rawash farm which is located in Giza, Egypt, as previously described by Shalaby et al. (1999). One isolate spores was obtained from heavy metal polluted soil (Spoll) irrigated with heavy metals-polluted sewage effluent for 10 years application, other isolate spores obtained from corresponding unpolluted soil (Sunpoll) irrigated with well water. AM fungi resting spores were isolated from rhizospheric soil in each site by wet sieving (63 sieve ) according to Gerdemann and Nicolson (1963). Only predominate, yellowish brown spores with the characteristic of Glomus spp. as described by Trappe (1982) were isolated under a binocular microscope and maintained in steril distilled water at 4-6 1C after surface sterilization in 3.5% calcium hypochlorite solution for 2 min and rinsing with sterilized distilled water several time and used within month. The soil samples were analysed according to Black et al.(1965) to determine physico-chemical composition and heavy metal content (Table 1).
Experiment 1
Effect of heavy metals treatments on the spores germination and hyphal growth: The spore germination and hyphal growth for each isolate were tested, on minimal (M) medium with 1% agar as described by Becard and Fortin (1988), for their sensitivity to zinc as (Zn SO4. 7H2O), cadmium as (Cd SO4. 8H2O) and lead as (Pb (NO3)2, which was added to the medium before autoclaving at 120 1C for 20 min and pH was adjusted to 6.5 using KOH. The final concentrations of each metal tested in minimal (M) agar medium were: 0, 5, 10, 20, 40, 80, 160 μmol L–1. For each treatment five replicates plates were prepared in petri-dish with diameter (16 cm). Each plate was inoculated with five spores which were placed at the vertices of an imaginary pentagon 6 cm far from edge. The Petri-dishes, were sealed with parafilm to prevent drying out and contamination, incubated at 30 1C in the dark.
| Table 1: | Characters of the soil from which AM Glomus spp spores were isolated |
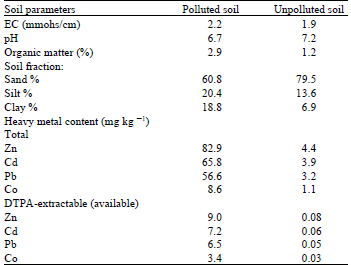 | |
| Table 2: | Graphically estimated EC50 values for scope germination as affected by Zn, Ca and Pb concentrations |
 | |
After incubation for 3 weeks, the plates were flooded with acid glycerol trypan blue (glycerol 500 ml; H2O, 45 ml; 1% Hcl, 50 ml; trypan blue, 0.5 g) for one hour, then examined under microscope (40x). Following two parameters were investigated:
(1) The spores germination percentage, (2) The hyphal length from germinated spore, estimated by a line- intersect method (Tennant, 1975). The effective concentration at which 50% of the spores that germinated without metal addition failed to germinate (EC50) was estimated graphically (Fig. 1)
Experiment 2
Effects of interactions between Zn, Cd and Pb on hyphal growth: The effect of heavy metals (at EC50 values) were investigated singly or in combination of 2 or 3 metals, on hyphal growth of isolates spores, using the methods of each isolate outlined above in experiment 1. The results were then expressed as a percentage of each individual metal treatment and displayed as histograms.
Data analysis: Data were presented as mean with standard errors (n=5). The mean effective concentrations at which 50% of the spores that germinated without metal addition failed to germinate (EC50) was estimated graphically.
Results: The spores of Glomus spp., isolated from polluted soil (Spoll) and corresponding unpolluted soil (Sunpoll) showed a variable responses for Zn, Cd or Pb treatments on spores germination percentage and hyphal growth from germinated spores (Fig. 1).
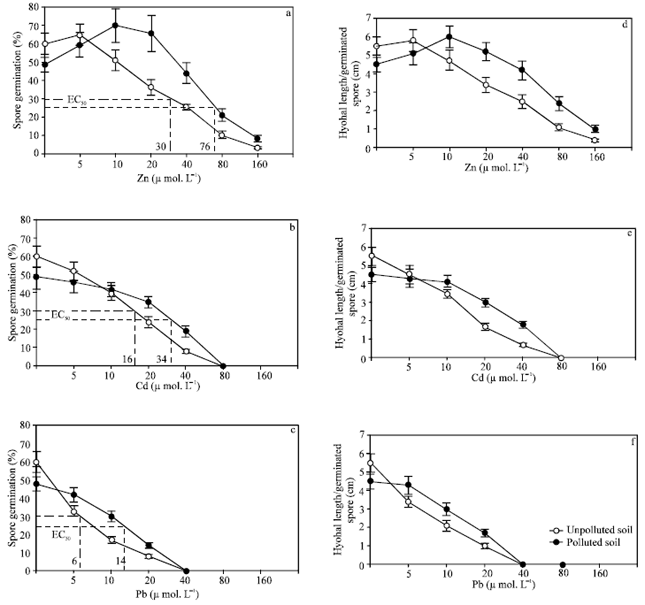 | |
| Fig. 1: | Effect of Za(a, d); Cd (b, e) and Pb (c, f) on spore germination and hyphal growth of two Glomus spp one isolated from unpolluted soil ( |
In control treatment (without heavy metal) and at low concentration of Zn and Cd (5 μmol L–1) the spore germination percentage and hyphal growth from germinated spore were higher for the spores isolated from unpolluted soil (Sunpoll) than spores isolated from polluted soil (Spoll). In contrast, with increasing heavy metal concentrations, percentage of the spore germination and hyphal growth of the spores isolated from (Spoll) were significantly higher than the spores isolated from (Sunpoll) in most treatments of three heavy metals tested (Fig. 1). Also, Zn at 5 μmol L–1 non-significantly increased germination percentage and hyphal growth for the spores isolated from unpolluted soil, while spores isolated from polluted soil showed highly significant increasing in germination percentage and hyphal growth with Zn treatments up to 20 μmol L–1 compared to control treatment, with less pronounced effect on hyphal growth (Fig. 1 a, d).
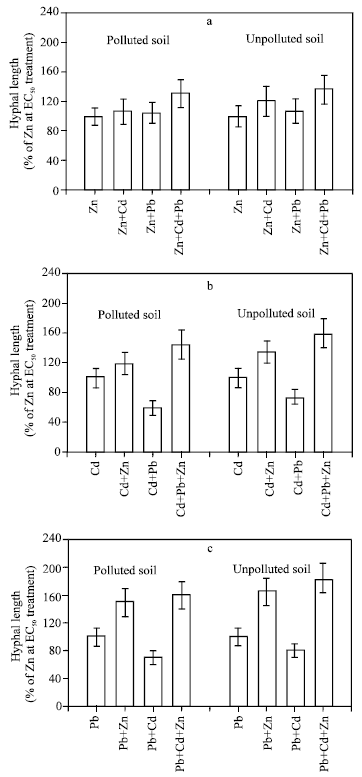 | |
| Fig. 2: | Interaction effects of (a) Zn, (b) Cd and © Pb at EC50 values on relative hyphal growth of Glomus spp spores isolated from polluted and unpolluted soils. Each column represents the means of 5 replicates plate (each with 5 spores), bars represents the standard error of mean. |
In the meantime, both isolates showed more toxic sensitivity to Pb than Cd, but they appeared more tolerant to Zn (Fig. 1a-f).
Each individual heavy metal treatment reduced germination percentage for both isolates spores by 50% of control treatment, as expected from the results of EC50 values graphically estimated (Fig. 1). The EC50 values for both isolates varied significantly for each heavy metal tested, 76, 30 μmol L–1 Zn more tolerant; 34, 16 μmol L–1 Cd less sensitive and 14, 6 mol Pb, more sensitive, for the spores isolated from polluted and corresponding unpolluted soil respectively. These results were confirmed with hyphal growth per germinated spore (Fig. 1 d-f).
The results of heavy metals interaction indicated a wide variation in response of the two isolate spores to heavy metals singly or in combination. In the meantime, the spores isolated from polluted soil (Spoll) was more tolerant to toxicity in all combination of metals treatments than the spores isolated from unpolluted soil (Sunpoll) (Fig. 2 a-c). Cd and Pb interacted with Zn singly (Zn + Cd), ( Zn + Pb) or in combination (Zn + Cd +Pb) to reduced Zn toxicity by increasing hyphal growth for both isolates spores compared to action of Zn alone (Fig. 2a). This effect was more pronounced with 3 heavy metals combination (Zn + Cd + Pb) which increase hyphal growth by 35 and 30% for the spores isolated from polluted and unpolluted soil respectively. Also, Zn ameliorated toxicity of Cd and/or Pb by increasing hyphal growth. Zn with Cd treatments increased relative hyphal growth by 20 and 35% compared to Cd treatment alone, while Zn with Pb increase hyphal growth by 50 and 65% compared to Pb treatment alone for the spore isolated from unpolluted and polluted soil respectively. In the meantime, Zn highly significantly ameliorated Cd and Pb toxicity in combination of 3 metals treatments (Zn + Cd +Pb) and hyphal growth increased by 45 and 60% compared to Cd treatment and by 60 and 85% compared to Pb treatment alone for spore isolated from unpolluted and polluted soil respectively. In contrast combination of Cd and Pb, (Cd + Pb) treatments, increased toxicity of Cd or Pb by decreasing hyphal growth by 25 and 40% compared to Cd treatment and by 20 and 30% compared to Pb treatment alone for the spores isolated from polluted and corresponding unpolluted soil (Fig. 2 a, b).
DISCUSSION
The spores, of AM fungus Glomus spp., isolated from polluted soil were more tolerant to heavy metals, Zn, Cd and Pb, compared to the spores isolated from corresponding unpolluted soil with over double magnitude EC50 values. In the meantime, germination percentage and hyphal growth of both isolates spores appeared more tolerant to Zn treatments than Cd while Pb was more toxic (Fig. 1). The isolation of metal-tolerant mycorrhizal fungi from polluted soil sites has been documented (Leyval and Weissenhorn, 1996). AM fungi, G. mosseae and G. macrocarpum, isolated from heavy metal contaminated soil showed higher percentage of spore germination in the presence of increasing doses of added Pb than a G. mosseae reference strain (Diaz et al., 1996).
The results showed that, the spores originally isolated from polluted soil differ in their metal susceptibility than the spores isolated from corresponding unpolluted. This variation may be due to ecophysiological adaptation of the spores isolated from polluted soil for heavy metals. The mechanism of adaptation to elevated heavy metal concentration are not known. However, regarding their relatively long generation time and the large number of nuclei in one spore, the observed rapid appearance of metal tolerance may be based on phenotypic plasticity rather than on selection of the tolerant genotypes (Weissenhorn et al., 1994). They suggested that, the spores of G. mosseae and G. etunicatum isolated from Cd polluted soil lost their tolerance to Cd and Zn compared to reference G. mosseae after one reproduction cycle (6 months) on metal-free substrate.
A number of studies had compared the response of AM fungus isolated from contaminated and uncontaminated soil sites in an attempt to relate metal sensitivity to the contamination of the soil origin. The results of investigations were contradictory. Weissenhorn et al. (1994) showed that, the spores of Glomus spp. isolated from polluted soil (long application of Zn polluted sewage sludge) culture exhibited a specific tolerance to Zn than unspecific tolerance isolate from other polluted soil (treated with Cd No3 for one year) culture to both Cd and Zn, despite the much higher Zn availability in sewage sludge polluted soil. Diaz et al. (1996) found that, sensitivity of two Glomus spp. isolates from different polluted soil sites was shown to vary significantly. In contrast, Weissenhorn et al. (1993) noted that, similar sensitivity (EC50 values) for Cd on Glomus spp. spores isolated from soil polluted with sewage sludge and from corresponding unpolluted soil. Willenborg et al. (1990) showed that, the susceptibility of ectomycorrhizal fungi to environmental stress factors, including heavy metals, varied from species to species but also from strain to strain within species. Also, other worker found wide variation in sensitivity of ectomycorrhizal fungi to Zn, with EC50 values ranging from 0.8-15300 m mol m–3 in agar medium and 135-3060 m mol m–3 in liquid medium (Denny and Wilkins, 1987; Tam, 1995).
Zn, but not Cd and Pb, particularly at low concentrations increased hyphal growth for germinated spores in both isolates and the effect was less pronounced than spore germination percentage (Fig. 1 a-d). This may be contributed to high affinity for Zn as essential element for fungal nutrition than for non essential Cd and Pb, where Zn is one constituent of Minimal (M) medium used for culturing AM fungi spores (Becard and Fortin, 1988). Weissenhorn et al. (1994) reported that, Zn tolerance was more pronounced at the level of spore germination than at the level of hyphal growth from germinated spores for AM fungi isolated from heavy metal-polluted and unpolluted soil. Smylla and Mroczkowska-Badner (1991) also observed higher Cd sensitivity of spore germination compared to hyphal growth from pre-germinated spores of Stredomyses they suggested that certain germination phases are particularly effected by competition between Cd and essential divalent cations or by inhibition of the enzyme activity. As known from higher plant, activities of stress related enzymes as oxidative enzymes, should be included as biomarkers in phytotoxicity of Cd contaminated soil (Lagriffoul et al., 1998). In the meantime, plant tolerated Cd toxicity by exhibited antioxidative defense mechanism by elevated antioxidative enzymes activities (Schickler and Caspi, 1999). Similar mechanisms may be assumed for AM fungi.
Interaction between heavy metals influence their relative toxicity to AM fungi. Inhibition of hyphal growth from germinated spores caused by multiple metals interaction can not be predicted from their individual toxicity and vary between two isolates. The most interactions of heavy metals reduced toxicity for both isolates compared to the toxicity of individual metals. Zn ameliorated toxicity of Cd and/or Pb for both isolates and was more pronounced with spores isolated from polluted soil (Fig. 2 a-c). This would suggest that soil contaminated by a mixture of metals might not be as to AM fungi as predicted from toxicity assessment of individual metals.
Amelioration of Cd toxicity with Zn interaction was recorded in ectomycorrhizal fungus Suillus variegatus where appeared to be highly sensitive to Cd in absence of Zn, with Cd EC50 value 0.8 m mol m–3, yet in the presence of 500 m mol m–3 Zn, the Cd EC50 increased significantly to 6.5 m mol m–3 (Hartley et al., 1997). Furthermore, Colpaert and Van Assche (1992) recorded amelioration by Zn to Cd toxicity on the ectomycorrhizal fungus Suillus bovinum where 765 m mol m–3 Zn reduced the toxicity of 8.9 to 89 m mol m–3 Cd. This amelioration by Zn was also recorded in the basidiomycete A. aegerita where increasing the concentration in agar media suppressed Cd translocation into the mycelium (Brunnert and Zadrazil, 1985). Metals tolerance interaction have also been documented in plants, while Zn was showen to interacted with Cd and Pb, both metals were competitively inhibited (Symeonidis and Karataglis, 1992).
There are a number of possible explanation for the interaction between Zn with Cd and/or Pb. The plasma membrane is the main selective barrier to influx / efflux of cations and anions and is the principal site of active transport (Meharg and Macnair, 1992). Ions being transported from external to the cytoplasm bind to transport sites in the plasma membrane. It is therefore, possible that competition between ions of the same valancy or size may occur, assuming that the number of binding sites is small in relation to the concentration of competing ions (Marschner 1995). It is possible, therefore, that the interaction of Zn with Cd and / or Pb would vary dependent on the concentration of metals involved. At relative high Zn concentrations amelioration of Cd and / or Pb toxicity for both isolates was significantly high in most treatments (Fig. 2), where the metals interaction takes place at EC50 value for each metal (EC50 values used were 76, 36 μmol L–1 Zn; 34, 16 μmol L–1 Cd and 14, 6 μmol L–1 Pb for spores isolated from polluted and unpolluted soil respectively). Also, as Zn is an essential element for fungal nutrition, divalent metal transporters might have a higher affinity for Zn than for the non-essential Cd and Pb. A second hypothesis could be that a physiological response in mycorrhizal fungi is induced by the presence of Zn, but not Cd and Pb. This response might result in decreased sensitivity to Zn and increased Cd and/or Pb sensitivity to mycorrhizal fungus. A range of mechanisms have been proposed by which mycorrhizal fungi protect themselves and their hosts against metal toxicity fall into four main categories: reduced influx; extracellular binding; complication with polyphosphate granules; and intracellular chelation (Hartley et al., 1997). These results indicated that, in most cases, AM fungal spores isolated from heavy metals polluted soil could be adapted to tolerate toxicity of heavy metals investigated as singly or in combination than the spore isolated from unpolluted soil.
Whilst single metal studies are essential for investigating the relationships between AM fungi and potentially toxic metals, it is likely that this investigation would have little practical relevance for predicting the effect of multiple metal exposure on AM fungi in contaminated soil. Potentially toxic metals are very rarely present singly in the environment but occur in mixture. Further work is required to examine the interactive effects of metals on AM fungi in symbiosis with host plants.
Acknowledgment
The author thank Prof. F. Gazzal, from the Soil Water and Environment Res. Inst., Agric. Res. Center, Giza, Egypt, for his help in soil analysis.
REFERENCES
- Diaz, G., C. Azcon-Aguilar and M. Honrubia, 1996. Influence of arbuscular mycorrhizae on heavy metal (Zn and Pb) uptake and growth of Lygeum spartum and Anthyllis cytisoides. Plant Soil, 180: 241-249.
Direct Link - Salt, D.E., M. Blaylock, N.P.B.A. Kumar, V. Dushenkov, B.D. Ensley, I. Chet and I. Raskin, 1995. Phytoremediation: A novel strategy for the removal of toxic metals from the environment using plants. Nat. Biotechnol., 13: 468-474.
CrossRefDirect Link - Schickler, H. and H. Caspi, 1999. Response of antioxidative enzymes to nickel and cadmium stress in hyperaccumulator plants of the genus Alyssum. Physiol. Plant, 105: 39-44.
CrossRef - Tennant, D., 1975. A test of a modified line intersect method of estimating root length. J. Ecol., 63: 995-1001.
CrossRefDirect Link - Weissenhorn, I., A., Glashoff, C. Leyval and J. Berthelin, 1994. Differential tolerance to Cd and Zn of Arbuscular Mycorrhizal (AM) fungal spores isolated from heavy metal-polluted and unpolluted soils. Plant Soil, 167: 189-196.
CrossRefDirect Link - Weissenhorn, I., C. Leyval and J. Berthelin, 1993. Cd-tolerant Arbuscular Mycorrhizal (AM) fungi from heavy-metal polluted soils. Plant Soil, 157: 247-256.
Direct Link - Gerdemann, J.W. and T.H. Nicolson, 1963. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Trans. Br. Mycol. Soc., 46: 235-244.
CrossRefDirect Link








