Research Article
A Review on Thornless Roses
SDU Faculty of Agriculture, Department of Horticulture Central Campus 32260 Isparta, Turkey
Roses (Rosa spp.) are one of the most important flower crops in the world (Short and Roberts, 1991). The rose is a member of Rosaceae family. Although the number of species in the genus is about 120, the direct ancestry of modern roses includes only 8-10 species. The basic chromosome number is n=7 and approximately half of the species are diploids (2n=14). There are also triploid (3n=21), tetraploid (4n=28), pentaploid (5n=35), hexaploid (6n=42) and two octoploid (8n=56) species (Stewart, 1969).
Roses species grow as shrubs, bushes and climbing plants. Most have ’thorns’. The term “thorn” is commonly used to refer to the hard multicellular epidermal appendages found on stems and leaves of plants. Since they have no vascular tissue and develop epidermally they are technically known as “prickles” (Nobbs, 1984 and Rosu et al., 1995). Thorns make roses difficult to grow and handle; retailers commonly “dethorn” roses prior to sale. Furthermore, in the industrial production of oils, essence, and other products for the pharmaceutical and cosmetic trade, thorns add to the difficulty of harvest. Therefore, a thornless rose would be preferred by many growers, merchandisers, retailers and by the public (Nobbs, 1984).
Researchers have used natural mutations, conventional breeding methods and chance seedlings to breed thornless roses. Spontaneous thornless mutants of rose have been described (Morey, 1969; Nobbs, 1984; Oliver, 1986; Druit and Shoup, 1991). Unfortunately, the best thornless mutants are sterile and/or have reverted to the thorny condition following temperature shock, freezing, or other environmental extremes (Nobbs, 1984). This instability suggests that many thornless roses are chimeras, consisting of mutant thornless tissue that grows together with normal thorny tissue (Nobbs, 1984 and Rosu et al., 1995). The instability could also be due to transposable elements. A tissue culture method to isolate pure thornless and chimeral plants, would allow differentiation between these two possibilities. If thornless mutant plants are chimeras, tissue culture could allow us to separate the pure thornless genotype from the thorny tissue growing with it. In this way the pure thornless roses will transmit thornless genes through a sexual cycle and geneticists will have access to new genes for thornlessness.
Chimeras: In Greek mythology, a chimera is a fire-breathing monster having the head and foreparts of a lion, the middle part of a goat and the hind parts of a dragon (Neilson- Jones, 1969). In plant science, the term chimera refers to a plant which has at least two genetically different cell layers or groups growing together as a result of mutation in the apical meristem. Mutations that occur within a plant often affect only a segment of the meristem and give rise to sectors of layers of the mutated tissue. A plant propagated from such a meristem is thus composed of two or more genetically distinct tissues (mutant cell groups) growing adjacent to each other in the same individual plant (Neilson-Jones, 1969 and Tilney-Bassett, 1986).
A chimera may develop in several different ways: spontaneous mutation, chemical mutagenesis and colchicine treatment, grafting, or by selecting among seedlings (Hartmann and Kester, 1990). Mutations are believed to be single cell events and may occur in any type of plant tissue. The location of a mutant cell is important for determining its effect on the chimeric plant. Those in the outer layers of the apical dome are more likely to be observed than those that develop deep in the body of the plant. Mutations may be detected visually chemically, morphologically or remain unknown. Mutations for fruit color, leaf shape or color, changes in fruit and leaf size, reversion from thorny state to thornless state are easy to distinguish visually. Other mutations such as change in resistance to severe winter or changes in tolerances to salt levels may never be observed except under a stress condition such as severe winter (Hartmann and Kester, 1983; Marcotrigiano, 1990).
Chimeras have been classified in several different ways (Tilney-Bassett, 1986). Plant chimeras can be classified according to their origin, induced by several factors, such as spontaneous mutations, chemical mutagens and colchicine treatments, grafting, inheritance (sorting out among seedlings) (Hartmann and Kester, 1975); mixing of different groups of cell in the callus cultures; or somatic hybridization of cells in protoplast fusion experiments.
Chimeras can be classified according to their behavior as species or graft chimeras, chromosomal chimeras, nuclear gene-differential chimeras or plastid gene-differential chimeras depending upon how they are expressed in vivo (Tilney-Bassett, 1986).
Chimeras can be classified into three distinct groups according to their structure; mericlinal chimeras, sectorial chimeras and periclinal chimeras based on the arrangement of their histogenic layers in the apical meristem (Hartmann and Kester, 1975 and Tilney-Bassett, 1986). The arrangement affects how they are expressed in vivo.
Sectorial chimeras: Sectorial chimeras are composed of two genetically different tissues situated side by side occupying distinct sectors of the stem. This type of chimera is often seen as distinct regions (sectors) on variegated plants (Hartmann and Kester, 1975).
The position of a lateral bud determines the segregation pattern of chimera. The location of the mutated cell in an apical meristem determines chimeral type. If a mutation occurs in a cell on one side of the apical meristem, only a portion of the stem will be affected yielding a sectorial chimera. Any lateral shoots developing from this mutated region of the stem might be completely mutant (e.g. thornless), on the other hand, a lateral shoot developing from the opposite side of the main stem will be parental type (e.g. thorny) (Hartmann and Kester, 1975).
Periclinal chimeras: The apical meristem of a periclinal chimera includes at least one complete layer of mutated cells. A chimera with a layer or two of mutated tissue that completely surrounds nonmutated tissue called periclinal chimera. The common name for a periclinal chimera is a “hand-in-glove” chimera since mutated tissue (the glove) completely masks internal regions (hand) (Dermen, 1960). Periclinal chimeras are relatively stable and are quite common. Their apical meristem includes one or more entire histogenic layers that differ genetically from the other/others. Many thornless blackberries (Rubus sp.) are good examples of this type of chimera (Harmann and Kester, 1975). ‘Thornless Evergreen’, for instance, consists of a layer of mutant epidermal (LI) tissue that surrounds a core of wild type thorny tissue (McPheeters and Skirvin, 1989).
Mericlinal chimeras: Mericlinal chimeras are similar to periclinal chimeras, a mutated tissue occupies only a small part of one of the histogenic layers and “does not extend completely around the shoot” (Hartmann and Kester, 1975 and Al-Wasel, 1994).
Thornlessness: Botanically all roses are thornless. The thorn-like structures of roses are properly called prickles, since they arise from single cells on the epidermis which divide repeatedly to form multicellular structures commonly known as “thorns”. In contrast, true thorns are modified stems or leaves and arise from deep within the tissues, so they possess vascular tissue (Rosu et al., 1995 and Nobbs, 1984).
In general, when a thorny plant mutates to the thornless condition, only the epidermal cells are affected. All internal tissues remain genetically thorny. Unless a plant loses its periclinal structure, the plant remains thornless, but shoots arising form the internal tissues, will be thorny. Shoots arising from the roots (root suckers) develop from the thorny internal tissues, root suckers will always have thorns (Skirvin, 1978).
Blackberries and raspberries (Rubus spp.) are members of the Rosaceae family and are close relatives of roses. Most blackberries are thorny (the British refer to this character as “spiny”) due to prickles which form on the epidermis. The actual number of prickles on a blackberry stem can be affected by light and, in general, plants grown in high light conditions have more thorns than those grown in the shade (Skirvin, 1978 and Peitersen, 1921). However, there are thornless brambles, most of which arose as sports (mutations) on thorny plants.
One of the most famous thornless sports of Rubus is the ‘Thornless Evergreen’ (‘TE’) blackberry which arose in 1926 as a sport of the original thorny ‘Evergreen’ parent. ‘TE’ is a periclinal (hand- in -glove) chimera in which the thornless epidermis surrounds a core of cells possessing the thorny genotype (McPheeters and Skirvin, 1983). Although ‘TE’ is thornless, it reverts to the thorny state when it forms root suckers. Thorny suckers originate from internal regions (LIII) of the plant; their phenotype represents the thorny genotype found at the core of the ‘TE’ sport. Furthermore, all of the offspring of ‘TE’ are thorny since its gametes develop from the internal (LII) regions of the plant. McPheeters and Skirvin (1989) succeeded in obtaining pure thornless shoots of TE in tissue culture. The pure thornless plants produced thornless root suckers and segregated for a dominant gene for thornlessness (Ste) (Hall et al., 1986 a, b and c). Among 1000 regenerants, one plant was found to be fully fertile and as good as the original parental clone; this plant was recently granted a plant patent as ‘Everthornless’ (McPheeters and Skirvin, 1983 or 1989).
Thornlessness in rose: The breeding of rose by conventional sexual methods is difficult, time consuming and expensive due to genetic incompatibilities, sterility, polyploidy and a relatively long generation cycle.
| Table 1: | Thornless roses that have been reported in the literature |
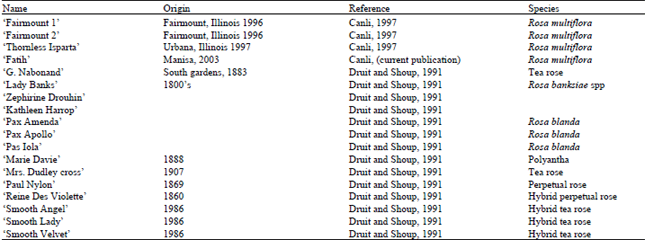 | |
Polyploidy is common in roses and is another drawback in rose breeding since gene segregation is complicated in polyploid species (Ben-Meir and Vainstein, 1994; Rajapakse et al., 1992; Hsia, 1995). In spite of these complications rose breeders, produce new cultivars at a remarkable rate. The introduction and development of germplasm for thornlessness could speed the introduction of the trait into roses by both conventional (sexual) and molecular means.
The most common types of thornless roses arose as natural mutations (sports) on thorny plants. Thornless sports or mutants of rose have been reported on many occasions (Table 1) for many years (Morey, 1969; Nobbs, 1984; Oliver, 1986; Druit and Shoup, 1991); but most have either been unstable for the thornless character (Nobbs, 1984 and Rosu et al.,1995); not true-to-type from seed or cuttings, or revert to the thorny state after a severe winter or other environmental stress (Nobbs, 1984; Oliver, 1986; Druitt and Shoup, 1991). Nobbs (1984) and Rosu et al. (1995) proposed that such unstable thornless roses are probably chimeras and consist of regions of both thornless and thorny tissue growing together.
Nobbs (1984) reports that there are degrees of thornlessness in roses that vary from complete thornlessness to thorny. However, completely thornless roses, which have no thorns on either stems or leaf midribs, are very rare. Roses with thornless stems and some thorn-like structures on petioles are more common than the completely thornless ones. Roses that have a few thorns on their shoots but not on the flowering stem, have been accepted as “thornless” by both consumers and growers (Nobbs, 1984).
Oliver (1986) reported finding a thornless chance seedling in 1963 which many years later reverted to a thorny condition. He assumed this reversion was a result of incomplete mutation. Later, he selected another 16 seedlings, two of which were absolutely free of thorns. Based on his experience, he did not expect them to revert to the thorny state.
Druitt and Shoup, two breeders from a central Texas nursery, released a report on thornless rose breeding in 1991. They stated that although there is little information on thornless rose breeding and genetics, they found more thornless roses than they had expected. They believe that a rose should only be classified as completely thornless if it has been grown at several locations for at least 15 years without thorns reoccurring.
Using this criteria on a collection Druitt and Shoup (1991) reported thornless roses in Texas, none of which has proved to be pure thornless. ‘G. Nabonand’, a tea rose found 1883 had been grown in their garden for three years without developing any prickles but they stated that the amount of time they had observed the plant was not long enough to be sure. Another old thornless variety is ‘Lady Banks’ (R. banksiae spp) which still produce few thorns after more than 150 years in southern gardens.
There are some relatively thornless roses that are commercially available: ‘Zephirine Drouhin’ a bourbon rose, ‘Kathleen Harrop’ a sport of ‘Zephirine Drouhin’, ‘Marie Davie’ a polyantha variety from 1888, ‘Mrs. Dudley cross= an old tea rose from 1907, ‘Paul Nylon’ perpetual rose from 1869 and another hybrid perpetual rose named ‘Reins Des Violette’ introduced in 1860 (Druitt and Shoup, 1991).
‘Smooth Angel’, ‘Smooth Lady’ and ‘Smooth Vavel’, Hybrid tea roses, were introduced in 1986 and described as "95 to 100 percent thorn free". However, these roses develop a few thorns when grown under various climates (Druitt and Shoup, 1991). Reported that a putative thornless rose they tested sometimes sprouted thorns or produced thorny canes. The frequency of reversion was affected by age, extreme temperature and change in soil make up.
‘Pax Amenda’, ‘Pax Apollo’ and ‘Pax Iola’ are named varieties bred from a thornless form of the American species R. blanda in South Dakota as thornless rootstocks. However, these also are not completely thornless (Druitt and Shoup, 1991).
Morey (1969) used mutation-inducing compounds to make many types of mutant rose. He reports that most of his mutants have been of no value for breeding because they were periclinal chimeras. The putative thornless roses reported by Nobbs (1984) and Rosu et al. (1995) probably are also chimeral. The situation remains unknown.
Rosu et al. (1995) modified the procedures used earlier by McPheeters and Skirvin (1983 and 1989) for chimeral blackberries to obtain putative pure thornless parent. Rosu et al. (1995) then studied methods to develop putative adventitious shoots from a chimeral thornless rose (R. multiflora thunb ex. J. Murr.). Since no thorns were observed on the stems of regenerants and three of the regenerants were classified as almost smooth, even on their petioles. While the parent plant produced some lateral branches with recurved thorns, the authors assumed they had succeeded in obtaining a pure thornless rose by separating the chimeral tissues.
Because the chimeral status of the regenerants was unknown, the authors tried to force adventitious shoots (suckers) from the roots of their putative chimeral plants without success (Rosu et al., 1995). The authors assumed that a periclinal chimeral thornless rose that possessed a thornless epidermis (LI) that overlayed a core of thorny cells (LII and LIII) should produce thorny, not thornless, root suckers. In this manner these authors planned to use suckers to distinguish chimeral and pure thornless plants, but they were not successful in forcing root suckers.
Three chimeral thornless rose named Thonless Isparta, Fairmount 1 and Fairmount 2. ‘Thornless Isparta’, a R. mutiflora, was found by Fatih A. Canli in a field close to Orchard Downs location of the University of Illinois at Urbana. Thornless Isparta arose as a sport on a thorny plant. He found about 10 to 12 completely thornless shoots coming out of a 10 to 15 year-old thorny rose bush. This cultivar was fertile and fruits contained many more fruit than either ‘FM1’ or ‘FM2’. ‘Thornless Isparta’ set one to eight seeds per fruit (average 3.03 seeds per fruit) and four out of ten seeds contained normally developed non absorbed embryos, indicating this plant a good parent to study the genetic basis of thornlessness.
‘Fairmount 1’ (‘FM1’) and ‘Fairmount 2’ (‘FM2’) are chimeral R. multiflora roses, were found by Dr. Philip Dziuk on his farm in Fairmount, Illinois.
In vitro regeneration and segregation methods to obtain pure thornless roses. He classified 59 plants as thornless among 930 plants after a year of growth. He also reported selecting 16 thornless roses from the seeds derived from segregating populations of parental clones of ‘Thornless Isparta’ and ‘FM1’. The thornless seedlings of ‘Thornless Isparta’ and ‘FM1’ should be pure thornless (thornless-thornless-thornless) and not chimeras since they developed sexually from a single cell.
Genetics of thornlessness in rose: The genetic basis of thornlessness in roses remains largely unknown because most thornless roses are infertile making genetic investigations and thornless cultivar improvement difficult (Nobbs, 1984). Thornlessness in the rose is due to cytoplasmic causes. He reports a cross between a thornless individual (parent not specified) and ‘Fragrant Cloud’. When the thornless seedling was used as the seed parent, it produced three thornless seedlings, but when ‘Fragrant Cloud’ was used as a seed parent, with the pollen from the thornless rose, all seven seedlings were thorny. This experiment is obviously too small to prove a point, but it suggests that the thornless character is cytoplasmically inherited and the thornless plant should be used as the seed parent (Nobbs, 1984).
Morey (1969) reported that “evidence suggested that the presence and amount of thorns, their size, shape and size classes are regulated by a wide assortment of genes, none of which have, to my knowledge, been subjected to critical study although the fact that the relative dominance of thornlessness seems to vary with different interspecific and intervarietal crosses.”
Although some thornless roses have little commercial value due to their instability, the use of tissue culture could allow us to separate the pure thornless genotype from the thorny tissue growing with it. In this way pure thornless forms of rose cultivars could be obtained and tested (Rosu et al., 1995). In addition, if we assume that the thornless character will pass through a sexual cycle, geneticists will have access to new genes for thornlessness. These can be used in traditional breeding cycles of crosses followed by selection or through genetic engineering efforts where the thornless gene could be introduced directly to outstanding thorny cultivars to yield thornless plants.
Most researchers have used natural mutations, conventional breeding methods and chance seedlings to breed thornless roses. These procedures are time consuming, expensive and the probability of obtaining a thornless rose is low. Thus, tissue culture techniques appear to be a good way to obtain pure thornless roses without losing the desirable characters of the chimeral parent.
Separation of chimeral plants into pure types: In vitro propagation has been accepted as rapid and reliable method to propagate many ornamental species. However, propagators and researchers have realized that clonal stability is not always the case and intraclonal variability has been observed in many crop species. Variation can be a serious problem to a propagator who requires extreme clonal stability, but such variation within a clone could facilitate the selection of unique forms of standard cultivars (Skirvin et al., 1994).
Rose shoots develop in vitro in two different ways, from pre-formed buds (axillary) or as adventitious shoots (not pre-formed). Most tissue culturistists agree that clonal stability is maximized when shoots develop from axillary bud cultures that have been proliferated at slow to moderate rates. Shoots derived adventitiously or from rapidly proliferating axillary bud cultures are the least stable and are most likely to show tissue culture-induced (somaclonal) variation. Unfortunately, the clonal stability of rose cultivars derived from axillary buds in vitro has not been thoroughly investigated (Skirvin et al., 1994).
There have been many rose plants obtained from in vitro cultures. In general, those from axillary buds are mostly fertile and identical to the parent (Barve et al., 1984); adventitious shoots are less stable (Lloyd et al., 1988, Skirvin et al., 1990). According to Dubois et al. (1988) no significant differences were observed among tissue cultures of single node-derived shoots with regard to lateral breaks, number of flower buds and number of petals. Martin et al. (1981) reported no variation among 2125 tissue culture rose plants growing for 3 years in a field. Somaclonal variation is most common among adventitious regenerants (Skirvin, 1978). Lloyd et al. (1988) reported significant variation among plants derived form callus of a rose species hybrid, R. persica x xanthina. Whether the variation among these plants was somaclonal (stable) or epigenetic was not reported by the authors (Skirvin et al., 1990). If stability is desired for propagation purpose, callus development should be minimized by avoiding media that induce callus (Skirvin et al., 1990).
There are a number of methods that have been used to either separate chimeras into pure forms or rearrange their histogenic layers. All of these methods basically involve the selection of desired types among a large population of individuals. In some cases, the mutant may segregate spontaneously, while in other cases separation occurs following wounding or mutagenesis. Segregation of different cell types from chimeras can occur in several ways: a) the result of changes in the histogenic layer composition b) selective growth of cells from one histogenic layer to produce an individual of pure type; c) differentiation of a single cell to form an entire organized shoot or plantlet of pure type, or d) mutation. Induction of rapid multiplication in shoot tips and adventitious shoot formation in vitro are the most common methods used to sperate chimeras into their consistuent genotypes.
Shoot tips: The first published report on rose shoot proliferation and rooting were made by Jacobs et al. (1968). Skirvin and Chu (1979) developed protocols for proliferation and rooting of R. hybrida. Similar reports were made by Hasegawa et al. (1979). At the same time Davies (1980) published another report on rose propagation in vitro. Since that time there have been many reports on the proliferation of roses from shoot tips and meristems in vitro (Pittet and Moncousin, 1981; Skirvin and Chu, 1979; Dubois et al., 1988; Skirvin et al., 1990). Dubois et al., (1988) published protocols for about 36 different “dwarf” roses.
Chimeral Yucca elephantipes shoot tips adventitious pure green plants that were non-chimeral (Pierik and Steegman, 1983). Hall et al. (1986a) succeeded in obtaining a pure ‘Thornless Loganberry’ ( Rubus loganococcus) from a periclinal parent type by culturing meristem tips.
Adventitious shoots: Several regeneration protocols have been published for adventitious shoots for Rosa sp. (De Wit et al., 1990; and Lloyd et al., 1988). Stems, leaves and roots could be induced to form adventitious shoots directly on MS with BA (8.9 μM for roots; 2.2 μM for leaves) or following a callus-induction stage on different media for leaves and roots [(MS + BA (13.3 μM) + NAA (1.1 μM)] or internodes [MS + BA (4.4 μM) + NAA (0.003 μM). Most of the plants rooted and about 90% survived acclimatization to a greenhouse.
Rout et al. (1991) reported shoot regeneration on half strength MS medium supplemented with BA (0.89 or 2.2 μM), NAA (5.4 μM) and 2,4-dichlorophenoxyacetic acid (2,4-D, 2.2 to 9.0 μM). They also reported somatic embryo formation on half strength MS with BA (2.2 FM), NAA (0.05 μM) and gibberellic acid (GA3, 0.3 FM) and L-proline (0 to 800 mg l–1). Other regeneration protocols have been published by Rosu et al. (1995), Hsia and Korban (1996), Lloyd et al. (1988) and De Wit et al. (1990).
McPheeters and Skirvin (1983; 1989) succeeded in obtaining pure thornless blackberries from chimeral ‘Thornless Evergreen’ (‘TE’) (Rubus lacinatus), a close relative of roses. ‘TE’ blackberry arose in 1926 as a sport of the original thorny ‘Evergreen’. ‘TE’ is a periclinal (hand- in -glove) chimera in which the thornless epidermis surrounds a core of cells possessing the thorny genotype (McPheeters and Skirvin, 1983). Although this plant is thornless, it reverts to the thorny state when it forms root suckers. Suckers form from internal regions (LIII) of the plant and their thorny phenotype represents the thorny genotype found at the core of the ‘TE’ sport. Furthermore, all of the offspring of ‘TE’ are thorny since its gametes develop from the internal (LII) regions of the plant. McPheeters and Skirvin (1989) succeeded in obtaining pure thornless shoots of ‘TE’ in tissue culture. The pure thornless plants produced thornless root suckers and its offspring segregated for a dominant gene for thornlessness (Ste) (Hall et al., 1986b). Among 1000 regenerants, one plant was found to be fully fertile and as good as the original parental clone; this plant was recently granted a plant patent as ‘Everthornless’.
The genetically thornless and thorny tissues were sperated randomly (McPheeters and Skirvin, 1983 ) and later by the controlled regeneration from the leaves (Norton, 1994).
The putative thornless roses reported by Nobbs (1984) and Rosu et al. (1995) probably are also chimeral. Rosu et al. (1995) modified the procedures used by McPheeters and Skirvin (1983, 1989) to obtain putative pure thornless R. multiflora thunb ex. J. Murr. rose. According to the authors, most of the parental plant's stems were thornless, but some lateral branches had recurved thorns. Since no thorns were observed on the stem of regenerants and three of the regenerants were classified as almost smooth, even on the petioles, these authors assumed they had succeeded in obtaining a pure thornless rose by separating the chimeral tissues.
Because the chimeral status of the parental plant was unknown, the authors tried to force adventitious shoots (suckers) from the roots of the putative chimeral plant without success (Rosu et al., 1995). The authors had assumed that a chimeral thornless rose that possessed a thornless epidermis (LI) that overlayed a core of thorny cells (LII and LIII) should produce thorny, not thornless, root suckers. They were not successful in forcing root suckers. The situation remains unknown.
Verifying the chimeral nature of a rose and distinguishing among types: Several authors have developed methods to assess the chimeral structure of known and unknown chimeras. Dermen (1960) demonstrated that the growing point of many rosaceous plants has an apical meristem arranged with three distinct cell layers (histogenic layers) that overlay each other. The outer layer (LI) is responsible for the development of the epidermal structures including prickles (“thorns”), the next layer in (LII) forms much of the body of the plant as well as its gametes and the innermost layer (LIII) contributes to the body of the plant and roots originate from this region. Dermen (1960) showed that one could determine the genotype of each layer and the chimeral status of a plant by histogenic analyses. The LI was assessed by its epidermal structure (in our case thornless or thorny); the LII was assessed by sexual crosses since gametes develop from this region; and the LIII was assessed by examining the phenotype of root suckers that developed adventitiously on roots.
McPheeters and Skirvin (1983, 1989) used this system to evaluate a large number of regenerants from ‘Thornless Evergreen’ (‘TE’) for pure thornless state. The original thornless parent was shown to be chimeral because it had a thornless epidermis (LI), but produced both thorny offspring (LII) and thorny root suckers (LIII) indicating its three histogenic layers to be arranged as thornless-thorny-thorny [LI, LII and LIII, respectively]. Thorny regenerants were no longer chimeral because they had a thorny epidermis (LI), produced thorny offspring (LII) and made thorny rootsuckers (LIII). Both pure thornless and chimeral thornless tissue culture regenerants had a thornless epidermis (LI), but only the pure thornless plants produced thornless root suckers. Later, the LII of the pure thornless plants was shown to segregate for the thornless character, indicating that it too was thornless (thornless, thornless, thornless) (Hall et al., 1986a).
Although thorny rose shoots can usually be distinguished from thornless ones, there is no rapid way to distinguish pure thornless and chimeral shoots because roses seldom sucker from shoots (Rosu et al., 1995). Therefore, to isolate a thornless rose, a method to assess the inner tissue (LII and LIII) of putative thornless roses must be developed. The isolation of a pure thornless rose will make it possible to study the genetics of the character and to use the gene(s) to breed a thornless rose. Eventually it may be possible to clone the thornlessness gene(s) and introduce them directly into outstanding rose clones and related species including the hybrid rose (R. hybrida). These gene(s) might also be useful to other thorny members of the rose family such as blackberries, raspberries and Pyracantha.