Research Article
Ovicidal Activity of Acrolein Vapors to Indian Meal Moth Eggs of Various Ages
Department of Entomology, Faculty of Agriculture, Urmia University, P.O. Box 57135-165, Urmia, Iran
Control of stored-products pests has been one of the major tasks for conservators because the damage inflicted to foodstuff is irreversible. A number of insect species pose a potential threat to a variety of stored-products. The Indian meal moth, Plodia interpunctella (Hubner), is a cosmopolitan pest attacking a wide range of stored-products of different nutritional values and physical properties. This insect causes serious losses both in quantity and quality of stored foodstuffs (Johnson et al., 1992; Hyun and Ryoo, 2000). Fumigants are commonly applied for control of stored-products pests. Two of the commonly used fumigants are methyl bromide and phosphine. Methyl bromide is now under threat of withdrawal because it apparently depletes the Earth’s ozone layer (Dunkel and Sears, 1998; Leesch et al., 2000). Phosphine has been used in a variety of habitats for a long time (Rajendran and Muralidharan, 2001). Conventional use of phosphine has been frequent failure to control insects and certain insects have developed resistance to phosphine (Bell and Wilson, 1995). Moreover concerns about the further development of resistance to phosphine has made the search for new alternatives imperative (Leesch, 1995).
The recent emphasis on objectionable insecticide residue in foodstuffs has prompted considerable thought and research in the human health and the environment (Brewer et al., 1994). Any compound that can reduce the insecticide load in a particular storehouse with adequate effectiveness to control insects may be of utmost importance in stored-product insect control programs. The main challenge is now for alternative substances, which are inexpensive, convenient to use and without substantial disruption of the environment. According to these criteria acrolein as a potential insect control compound was selected for testing. Acrolein is colorless liquid, relatively no persistent and the half-life in aquatic systems ranges from less than one to ≈ four days (Bowmer and Higgins, 1976). Acrolein is not carcinogenic and shows little embryotoxic and teratogenic behavior (Ghilarducci and Tjeerdema, 1995). Because of its high toxicity to insects (Carroll et al., 1982) and fast acting characteristics, acrolein could be highly efficacious in fumigation systems.
Studies on different insect species agree that the egg is the most difficult stage to kill (Williams and Sprenkel, 1990). These authors also provided the indication of the tolerance changing of eggs to sulfuryl fluoride over longer periods in their experiments. Acrolein is not registered for use as a fumigant. Therefore, there is little published information about the toxicity of acrolein vapors.
The current study was undertaken to investigate the efficacy of acrolein at various temperatures and exposure times on different age groups of eggs of P. interpunctella.
P. interpunctella was collected from local stores and shops, in Urmia (37.39° N 45.4° E), a town in Iran in 2006-2007. Stock cultures were established and maintained in wide-mounted glass jars. Indian meal moth was reared on a 10:2:1 mix of wheat bran, glycerol and dried yeast powder at 27±1°C and 60±10% R.H. in a 16 h light, 8 h dark lighting regime. All insects were cultured under moderately crowded conditions to ensure proper development of the resultant insects. Insects were reared for two generations before commence of experiments.
Chemical: The test acrolein (2-propenal, acrylaldehyde) was 95% active ingredient with density of 0.8389 g mL-1 at 20°C and supplied by Merck-Schuchardt, Mûnchen, Germany. All doses used in this study are expressed as commercial formulations.
Preparation of eggs for experiments: Mixed sex adults were collected from stock cultures, anaesthetized with carbon dioxide before confining them in a plastic sieve by attaching a glass dish. Taping a moist cotton wool pad inside the dish provided drinking water. The sieves were placed over collecting dishes in the rearing room. The eggs laid in the dishes were kept in the same room to arrive at the age required for bioassay. Using a fine sable brush and a binocular microscope, eggs with known age were counted out in batches of 20 on to watch glasses. Eggs with obvious defects were avoided. To commence fumigation each watch glass was placed singly in 1150 mL glass jar, which served as the fumigation chamber. For experiments at 17±1°C the collected eggs were moved to 17±1°C acclimatize for the fumigation which was started following morning. For tests at 27±1°C there was no need for conditioning of eggs prior to fumigation.
Bioassay: Twenty eggs were prepared in each age group for exposure to each dosage. Preliminary dosage-mortality tests were done before each experiment to determine a range of dosages that would produce ca. 25-75% mortality (Robertson and Preisler, 1992). Five dosages between 2 and 32 mg L-1 were tested at 27±1°C and five dosages between 5 and 80 mg L-1 at 17±1°C. Age groups of eggs were fumigated for 24, 48 and 72 h in 1150 mL glass jars separately. The jars were capped with screwed lids. Blotting paper strips measuring 2x6 cm were attached to the lower side of each lid by adhesive plastic tape. The required amount of each dosage of acrolein was deposited on the blotting paper strip with an Oxford sampler through a 5 mm diam hole, located in the center of the lid. Immediately after the acrolein was pipetted, the hole in each lid was sealed with plastic tape. In each test, the control jar was treated identically except that no acrolein was deposited on the blotting paper. At the end of exposure, watch glasses were taken out and eggs were left to air off in the fumigation chamber for 2 h before returning to the rearing room. After returning to 27±1°C, eggs were checked for hatch from three days after exposure until no further hatch was observed and cumulative mortality rates were determined. Each test was replicated three times on three different days. Dosage-mortality data from the replicates were pooled and the dosage-mortality response was determined.
Data analysis: The median lethal dosage (LD50) and LD95 of acrolein in the term of mg L-1 was estimated by subjecting mortality data to the maximum likelihood program of probit analysis using SPSS (1993). This program has a provision for control mortality. Two age groups of eggs were considered significantly different in their susceptibility to acrolein if fiducial limits (95%) of LD50 of acrolein did not overlap.
Dosage-mortality values estimated from the probit analyses of different age groups of egg mortality are given in Table 1 to 8. In all experiments, acrolein was toxic to the tested eggs. The toxicity of acrolein is greatly increased at 27±1°C. Considerable variation among age groups was apparent in the slopes of regression lines for toxicity test, whether time or concentration was fixed in the tests.
Tests at 27±1°C: At 27±1°C eggs of P. interpunctella were much more susceptible than at 17±1°C. There was an inverse relationship between exposure time to acrolein and estimated LD50 values (Table 1- 4). At 27±1°C and 24 h exposure on the basis of LD50 values, eggs aged 1-2 days proved more tolerant than other age groups, followed by 0-1 day-old and 2-3 day-old and 3-4 day-old eggs. A similar trend was observed at 48 and 72 h exposure periods. There was a considerable overlap in 95% fiducial limits of some dosage-mortality regression lines. Therefore, no statistically significant difference between the estimated LD50 values was observed. Table 2 presents that at the LD50 level, the dosage of acrolein required for killing the most tolerant eggs (1-2 day-old) in the shortest exposure (24) h, was 3.80 mg L-1.
Tests at 17±1°C: At 17±1°C and 24 h exposure, the sensitivity order of the age groups of eggs to acrolein was measured as 1-2 day-old>0-1 day-old>2-3 day-old>3-4 day-old eggs (Table 5-8). At this temperature and 24 h exposure the dosage required to control 50% of 1-2 day-old eggs was 16.44 mg L-1 (Table 6). As at 27±1°C here was considerable overlap in 95% fiducial limits of dosage-mortality regression lines. Therefore, in such cases no statistically significant difference between the estimated LD50 values was observed.
| Table 1: | Toxicity of acrolein to 0-1 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 27±1°C in 1150 mL jars |
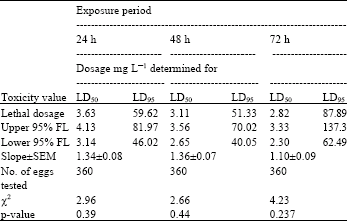 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 2, 4, 8, 16 and 32 mg L-1 | |
| Table 2: | Toxicity of acrolein to 1-2 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 27±1°C in 1150 mL jars |
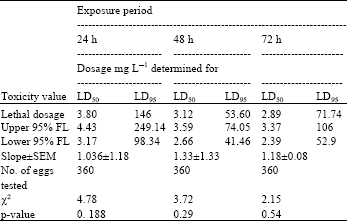 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 2, 4, 8, 16 and 32 mg L-1 | |
| Table 3: | Toxicity of acrolein to 2-3 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 27±1°C in 1150 mL jars |
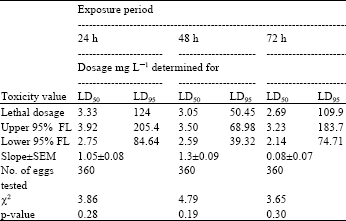 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 2, 4, 8, 16 and 32 mg L-1 | |
| Table 4: | Toxicity of acrolein to 3-4 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 27±1°C in 1150 mL jars |
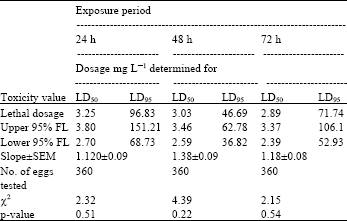 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are > 0.05 and the data fits regression model. Acrolein quantities used were 0, 2, 4, 8, 16 and 32 mg L-1 | |
| Table 5: | Toxicity of acrolein to 0-1 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 17±1°C in 1150 mL jars |
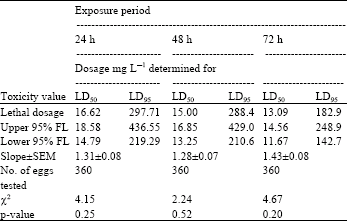 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 5, 10, 20, 40 and 80 mg L-1 | |
| Table 6: | Toxicity of acrolein to 1-2 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 17±1°C in 1150 mL jars |
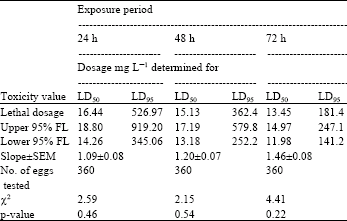 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 5, 10, 20, 40 and 80 mg L-1 | |
| Table 7: | Toxicity of acrolein to 2-3 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 17±1°C in 1150 mL jars |
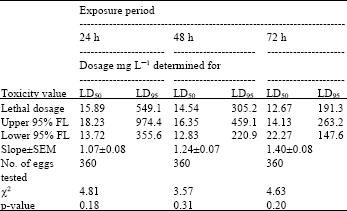 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 5, 10, 20, 40 and 80 mg L-1 | |
| Table 8: | Toxicity of acrolein to 3-4 day-old eggs of Plodia interpuctella exposed for 24, 48 and 72 h at 17±1°C in 1150 mL jars |
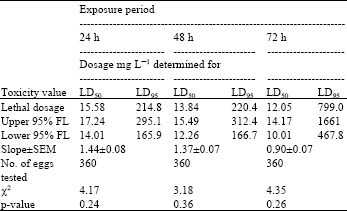 | |
| Three replicates (20 eggs per replicate) were tested in each of five acrolein dosages and control treatment. Pearson’s χ2 goodness-of-fit tests: all values of p are >0.05 and the data fits regression model. Acrolein quantities used were 0, 5, 10, 20, 40 and 80 mg L-1 | |
Control of stored-products pest insects is essential wherever foodstuffs quality is to be maintained. Fumigation is one of the most successful methods of rapidly controlling insect’s infesting stored-products. A good fumigant should have some characteristics consistent with the fumigation protocol, which ensures an appropriate level of insect control and produces the minimum of hazardous side effects (Bond, 1984). Unfortunately, the two available fumigants, methyl bromide and phosphine, fall short of this ideal (Casanova, 2002; Collins et al., 2002). In evaluating the effectiveness of a fumigant against an insect, it is essential that the dosage recommended be base on the most tolerant stage of the target insect to the fumigant. Studies on different insect species agree that the egg is the most difficult developmental stage to kill (Su and Scheffrahn, 1990; Drinkall et al., 1996). Williams and Sprenkel (1990) working with different age groups of Lyctus brunneus (Stephens) and Euvrilletta peltata (Harris) eggs reported that an intermediate age group proved less susceptibility to Sulfuryl Fluoride (SF) than younger or older eggs. In the present study the most tolerant eggs were aged 1-2 days and the dosage and exposure period required to achieve high mortality was dependent on the egg developmental stage. In 1-2 day aged group there was sufficient indication that longer exposure period achieved better kill than shorter ones of similar dosage. From this point of view results were in agreement with the findings of Su et al. (1989) who studied the toxicity of SF to Coptotermes formosanus Shiraki over varied exposure times. They reported that time and dosage was highly interchangeable but there was relatively advantage with longer exposure period.
In the present study, there was no evidence of a hatch delay longer than the time spent under vapors for eggs exposed at 27±1°C, indicating that some development must have occurred under fumigation. This finding would agree with the data collected by Bell (1976) who have demonstrated that the development of stored-products moth’s eggs may continue under fumigation period.
A new approach in fumigation research could be the use of less hazardous substances, which are more compatible with environment. The application of acrolein as an insect control material may be an appropriate approach to this objective. The half-life in aquatic systems ranges from less than one to ≈ four days (Bowmer and Higgins, 1976). Acrolein is not carcinogenic and shows little embryotoxic and teratogenic behavior (Ghilarducci and Tjeerdema, 1995). Acrolein is retained irreversibly in the respiratory tract after exposure by inhalation (Morris, 1996). Consequently, there is little, if any, distribution to other organs. Therefore, death from acrolein should be extremely uncommon under fumigation conditions. In the current study, acrolein vapor was toxic to all age groups of eggs of P. interpunctella. The mechanism(s) involved in the insecticidal action of acrolein vapors are not known.
Acrolein vapor is flammable, therefore in the application of very high doses of acrolein which are expected to produce volume of acrolein vapors in air near flammability range the risk of fire cannot be ruled out.
It is well established that a good fumigant must kill the most tolerant developmental stage of the target insect with acceptable dose in a short period of time. Since acrolein is highly toxic to the insects (Carroll et al., 1982) including P. interpunctella eggs and because methyl bromide may not be available for use as a fumigant in immediate future (Casanova, 2002), acrolein could be considered as a potential compound for fumigation.
The author is grateful to Abas Samdie and Mehdie Tajbakhsh sh (Urmia University) for useful comments on an earlier draft of the manuscript. Technical assistance of Hoshang Golizadehi, Asghr Saie and Hassan Kamyar (Agricultural Faculty, Urmia University) is gratefully acknowledged.