ABSTRACT
In the present study, the effect of different concentrations of salicylic acid which is an endogenous organic acid in plants and which is commonly cited as a hormone, on the growth and some other parameters of pepper (Capsicum annuum L. cv.) seedlings was investigated. The solutions were applied to the roots of seedlings using hydroponic method. In general, 1.5 mM concentration of salicylic acid had a stimulating effect while 5 and 10 mM concentrations had varying degrees of inhibitive effects on the seedlings. Although 0.3 mM SA application produced prominent results in the case of all parameters, the difference was not found statistically significant. The inhibitive effect produced by high SA was found much more dominant than the stimulating effect of low SA concentrations. Thus, it was established that SA had a bidirectional physiological effect on the seedlings in a concentration-dependent manner.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2011.300.304
URL: https://scialert.net/abstract/?doi=pjbs.2011.300.304
INTRODUCTION
Salicylic acid which is a secondary plant product, performs important actions in the growth and development processes of plants. It is a potent signaling molecule in plants and is involved in eliciting responses to biotic and abiotic stresses (Krantev et al., 2008). These actions include exercising a thermogenic effect (Ansari and Misra, 2007), increasing thermotolerance (Kaur et al., 2009), stimulating adventive root formation (Kling and Meyer, 1983), showing herbicidic effect (Shettel and Balke, 1983), reducing leaf shed (Ferrarese et al., 1996), providing resistance against pathogens (Alvarez, 2000), regulates ethylene biosynthesis (Huang et al., 1993; Srivastava and Dwivedi, 2000) and changing the quality and quantity of proteins (Doares et al., 1995). It has been claimed (Ray, 1986) that SA and similar phenolic compounds exercise their effect of providing resistance against different stress factors in plants (Szepesi et al., 2005; Yuan and Lin, 2008) by modifying the effects of abscisic acid and gibberellic acid (Apte and Laloraya, 1982) and cytokinins (Ray et al., 1983). These observations and reports on many other physiological effects brought about by SA invoked in several researchers the idea that this substance might be a new plant growth regulator (Hayat and Ahmad, 2007). It was purported that SA could exercise such an effect on NRA in the mediation of plant hormones (Schneider and Whitman, 1974). Fariduddin et al. (2003) reported increased NR activity due low concentrations of SA while higher concentrations were observed to be inhibitory to NR activity in Brassica juncea. A study by Singh et al. (2010) showed positive correlation between chlorophyll content and total nitrogen in cucumber cotyledons. Moreover, increase in nitrogen content and chlorophyll content at lower concentrations of SA indicates that this plays a regulatory role during the biosynthesis of active photosynthetic pigments. It is known at present that plants under stress age more rapidly than those under optimum conditions (Vaadia et al., 1961). Besides, cytokinins are known to have a delaying effect on proteolytic activity and leaf senescence (Nooden et al., 1997). It has been argued that the decline in plastid formation and chlorophyll-carotenoid synthesis in plants exposed to stress results from the accumulation of abscisic acid (Duysen and Freeman, 1976) or the decrease in cytokinins levels (Itai and Ben-Zioni, 1973). In another study carried out on discs of plucked rice leaves, ethylene biosynthesis was found to be inhibited in 2 h following SA administration (Huang et al., 1993). In still another study performed in spinach (Spinacea oleracea L.) suspension culture, ethylene accumulation inhibited chlorophyll production (Dalton and Street, 1976). Miguel-Chavez et al. (2003) observed that stem diameter and height of the plants increased by applying 10-10 and 10-8 M SA. Similarly, application of 10-8 and 10-6 M SA increased fresh stem weight, dry stem weight and root length. The commercially available form of salicylic acid is acetylsalicylic acid (ASA) (Mitchell and Broadhead, 1967). SA was observed to reduce leaf area (secondary leaf), root growth, as well as protein and chlorophyll (a+b) amount parallel to an increase in its concentration in barley plants which were developed from barley seeds germinated in SA solutions of varying concentrations and grown in SA-containing milieu (Pancheva et al., 1996). Khan et al. (2003) found that spraying minute concentrations (10-5 mol L-1) of SA and ASA on the leaves led to an increase in the overall photosynthetic yield of soybean and corn. It was reported in the same study that stomatal mobility and transpiration increased while chlorophyll amount remained unchanged. In studies where one-week-old corn and bean seedlings were ASA (50, 250, 1000 ppm) to the root (Canakci and Munzuroglu, 2002) or the leaf (Canakci and Munzuroglu, 2000) caused an increase in fresh weight loss and a decrease in transpiration in high concentrations. It has been reported in a study using discs obtained from primary leaves of one month old bean seedlings that chlorophyll a and b quantity decreased, carotenoid amount remained unaffected while fresh weight loss and protein destruction increased parallel to the increase in ASA concentration (100, 250, 500 ppm) (Canakci, 2003). ASA was found to lead to the closure of stoma pores in high concentrations (Larque-Saavedra, 1978). Environmental stress negatively affects the functions of the plasma membrane which can be measured as the level of lipid peroxidation. The amount of malondialdehyde (MDA) reflects the degree of lipid peroxidation. Study on the effect of SA in relation to oxidative stress indicate that the level of MDA in leaves remained unchanged (Gautam and Singh, 2009), decreased (Krantev et al., 2008) or increased (Popova et al., 2009) in association with toxic stress in relation to various SA concentrations. Under the circumstances, the objective of the present study is to examine the physiological and some biochemical effects of low and high concentrations of salicylic acid which builds resistance against biotic and abiotic stress in plants, on pepper seedlings in the absence of some other stress factor.
MATERIALS AND METHODS
Plant growth and treatment with salicylic acid solutions: The present study used 30-day-old seedlings produced from pepper (Capsicum annuum L. cv. Kapya) seeds were used as the vegetative material. Pepper seeds were obtained from May Seed Group. Seeds were first disinfected with 0.5% sodium hypochloride solution for 5 min and washed with deionised water. Then they were placed on double-folded filter paper wetted with 10 mL deionised water in petri dishes and germinated for 3 days in dark at 24-26°C temperature. Homogeneously germinated seeds were planted in plastic pots which were 25 cm-deep and 20 cm in diameter filled with leaf mould and sand (2:1 v/v). The plants were raised under natural light at 25±5°C and 60% humidity, in 16 h light/8 h dark photoperiod and were watered every other day. Of the 15-day-old seedlings, homogeneous ones were carefully removed from the raising environment and their roots were washed with tap water. These seedlings were placed in the channels of the sponge lids on the mouth of bottles of 150 mL volume and contained 100 mL Hoagland nutrient solution. Nutrient solutions were replaced every two days. When the seedlings were 30-day-old, their roots were treated for 16 h with 0, 0.3, 1.5, 5 and 10 mM salicylic acid test solutions prepared in deionised water using hydroponic method. The seedlings were studied after two days in terms of seedling and leaf length, seedling fresh and dry weight and pigment, protein and MDA content. While dissolving the crystal salicylic acid, 1 mL 95% ethyl alcohol was used (Shettel and Balke, 1983) and solution pH was set to 4.5-5 using 0.1 N NaOH (Larque-Saavedra, 1978). pH measurements were performed with JENWAY 3040 ion analysis using a pH-meter apparatus. Absorbance measurements were carried out using a UVA-093001 Heëlosá spectrophotometer. The growth parameters were determined in 10 seedlings for each group.
Tissue extraction and determination: One gram of fresh leaf obtained from seedlings which were treated with SA from the root was used in each group. For pigment analysis, the leaves were extracted and absorbance of these extracts was read against blind at 645 and 663 nm wavelengths. Quartz tubes of 1 cm3 volume were employed in the absorbance determinations. Chlorophyll (a+b) amounts were calculated from the absorbance values obtained (Witham et al., 1971). In order to establish the protein amount, fresh weights of seedlings to which SA was applied from the root were determined without delay and protein analysis was conducted. Protein extraction was performed according to Larson and Beevers (1965) method. The tubes containing the extracts were subjected to the procedures explained in the method (Lowry et al., 1951) and then, their absorbances were read against blind at 725 nm in the spectrometer, as described in the method. Standard curve prepared in advance were utilized to calculate the protein amount (mg g-1. fresh weight). A modified version of the method of Heath and Packer (1968) was used to determine the amount of lipid peroxidation (MDA) leaves.
Statistical analysis: All experiments were conducted in triplicate. The results were subjected to statistical analysis by measuring the standard deviation of the mean and using analysis of variance (SPSS 15.0 for Windows Evaluation, one-way ANOVA- Duncan test).
RESULTS
As understood from the results obtained after the treatment of pepper seedlings with SA for 16 h, 1.5 mM SA application encouraged increase in seedling length, relative to control group seedlings (13.58%) (p<0.05) but did not affect leaf length (p>0.05). Five mM SA application was observed to delay the increase in seedling length, in comparison to control group seedlings (16.46%) (p<0.05) but it did not affect leaf length (p>0.05). Ten mM SA application was shown to delay seedling length increase (21.76%) and leaf length increase (36.66%) (p>0.01) (Table 1).
The 1.5 mM SA application was found to have a positive effect on fresh weight increase (12.40%) (p<0.05) but not on dry weight increase (p>0.05). 5 mM SA application was established to exercise a negative effect on fresh weight increase (14.28%) and dry weight increase (18.48%) (p<0.05). 10 mM SA application negatively influenced fresh weight increase (25.93%) and dry weight increase (34.12%) (p<0.01) (Table 2).
The 1.5 mM SA application was established to bring about an increase in chlorophyll (a+b) (15.31%) and protein (16.64%) content (p<0.05). 5 Mm SA application was also found to cause reduction in chlorophyll (a+b) (13.01%) and protein (24.64%) (p<0.05) content.
| Table 1: | Seedling and leaf lengths found in pepper (Capsicum annuum L.) seedlings treated with SA for 16 h |
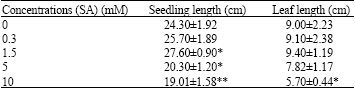 | |
| Significance level *: p<0.05, **: p<0.01 | |
| Table 2: | Fresh and dry weights found in pepper (Capsicum annuum L.) seedlings treated with SA for 16 h |
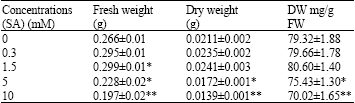 | |
| Significance level * :p<0.05, **: p<0.01 | |
| Table 3: | Chlorophyll (a+b), protein and MDA amounts found in pepper (Capsicum annuum L.) seedlings treated with SA for 16 h |
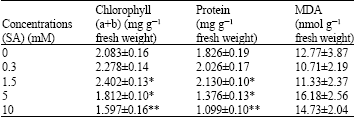 | |
| Significance level *: p<0.05, **: p<0.01 | |
Ten mM SA application was found to lead to significant reduction in chlorophyll (a+b) (23.33%) and protein (39.81%) (p<0.01) content (Table 3).
The 0.3 and 1.5 mM SA applications led to a decrease in the amount of MDA in comparison to the control but these data were found to be statistically insignificant (p>0.05). Similar to 5 and 10 mM SA applications led to a increase in the amount of MDA in comparison to the controls, these data were found statistically insignificant (p>0.05) (Table 3). Although 0.3 mM SA application also produced variations in all parameters, relative to the control group seedlings, the difference was not found to be statistically significant (p>0.05).
DISCUSSION
No studies on the effect of SA on pepper seedlings could be traced from the literature available. It is known that plants exposed to stress show reduction in growth parameters, reduced protein synthesis or increased protein destruction and earlier aging due to critical metabolic changes like chlorosis (Vaadia et al., 1961). Earlier scientists also could observe stress-associated changes in the case of high SA concentrations (Canakci and Munzuroglu, 2002). Low SA concentrations remained either ineffective or showed positive effects in terms of seedling growth (Khan et al., 2003). The inhibitory effect produced by high SA concentrations was found to be much more dominant than the stimulating effect of low SA concentrations. As we have already noted, lipid peroxidation is a central parameter that increases with environmental stress. The available data indicate that SA concentrations do not affect the amount of MDA (Gautam and Singh, 2009) on pepper seedlings.
The results brought about by high SA concentrations may be attributed to osmotic water correlations (Larque-Saavedra, 1978) intercellular space and stress due to a toxic effect. The decrease caused by high concentrations of SA in chlorophyll amount was claimed to have resulted from inhibition of chlorophyll biosynthesis, acceleration of chlorophyll destruction or both (Yang et al., 2002; Popova et al., 2009). It is known that SA and other salicylates have got an effect on the protein synthesis mechanism that could be described as a control mechanism (Fariduddin et al., 2003; Singh et al., 2010). It can be assumed that low SA concentrations inhibit ethylene biosynthesis (Huang et al., 1993; Srivastava and Dwivedi, 2000) while high SA concentrations stimulate it. The same mechanisms may be operating in opposite directions in a hormone-enzyme relation (Schneider and Whitman, 1974) depending on the SA concentration applied to the plant. Results showed that the toxic stress can be claimed for pepper seedlings (pigment and protein loss) but that oxidative stress is unclear (Gautam and Singh, 2009; Krantev et al., 2008; Popova et al., 2009).
CONCLUSION
Salicylic acid is an effective precursor molecule that builds resistance against abiotic and biotic stress in plants. It produces a similar effect depending on the concentration of the exogenously applied SA and the type of the plant. However when applied individually and at high concentrations, SA may be a toxic stress factor for the plant. In conclusion, these physiological effects of SA which is regarded a new growth regulator, should be considered at the hormonal dimension.
REFERENCES
- Ansari, M.S. and N. Misra, 2007. Miraculous role of salicylic acid in plant and animal system. Am. J. Plant Physiol., 2: 51-58.
CrossRefDirect Link - Apte, P.V. and M.M. Laloraya, 1982. Inhibitory action of phenolic compounds on abscisic acid induced abscission. J. Exp. Bot., 33: 826-830.
CrossRef - Canakci, S., 2003. Effects of Acetylsalicylic acid on fresh weight, pigment and protein content of bean leaf discs (Phaseolus vulgaris L.). Acta Biol. Hungarica, 54: 385-391.
Direct Link - Dalton, C.C. and H.E. Street, 1976. The role of the gas phase in the greening and growth of illuminated cell suspension cultures of Spinach (Spinacea olareacea L.). In vitro Cell. Dev. Biol. Plant, 12: 485-494.
CrossRef - Doares, S.H., J.N. Vesquez, A. Conconi and C.A. Ryan, 1995. Salicylic acid inhibition of proteinase inhibitors in tomato leaves induced by systemin and jasmonic acid. Plant Physiol., 108: 1741-1746.
Direct Link - Duysen, M.E. and T.P. Freeman, 1976. Promation of plastid pigment accumulation in water stressed what leaf sections by hormone treatment. Am. J. Bot., 63: 1134-1139.
Direct Link - Fariduddin, Q., S. Hayat and A. Ahmad, 2003. Salicylic acid influences net photosynthetic rate, carboxylation efficiency, nitrate reductase activity, and seed yield in Brassica juncea. Photosynthetica, 41: 281-284.
CrossRefDirect Link - Ferrarese, L., P. Moretto, L. Trainotti, N. Rascio and G. Casadoro, 1996. Cellulase involvement in the abscission of peach and pepper leaves is affected by Salicylic acid. J. Exp. Bot., 47: 251-257.
Direct Link - Gautam, S. and P.K. Singh, 2009. Salicylic acid-induced salinity tolerance in corn grown under NaCl stress. Acta Physiol. Plant., 31: 1185-1190.
CrossRef - Hayat, S. and A. Ahmad, 2007. Salicylic Acid: A Plant Hormone. Springer, Netherlands, ISBN-13: 9781402051838, Pages: 401.
Direct Link - Heath, R.L. and L. Packer, 1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys., 125: 189-198.
CrossRefPubMedDirect Link - Huang, Y.F., C.T. Chen and C.H. Kao, 1993. Salicylic acid inhibits the biosynthesis of ethylene in detached rice leaves. Plant Growth Regul., 12: 79-82.
CrossRefDirect Link - Itai, C. and A. Ben-Zioni, 1973. Short and long-term effects of high temperatures (47-49°C) on tobacco leaves, II. O2 uptake and amylolytic activity. Physiol. Plant, 28: 490-492.
CrossRef - Kaur, P., N. Ghai and M.K. Sangha, 2009. Induction of thermotolerance through heat acclimation and salicylic acid in Brassica species. Afr. J. Biotechnol., 8: 619-625.
Direct Link - Khan, W., B. Prithiviraj and D.L. Smith, 2003. Photosynthetic responses of corn and soybean to foliar application of salicylates. J. Plant Physiol., 160: 485-492.
Direct Link - Krantev, A., R. Yordanova, T. Janda, G. Szalai and L. Popova, 2008. Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J. Plant Physiol., 165: 920-931.
CrossRefPubMedDirect Link - Singh, P.K., V.K. Chaturvedi and B. Bose, 2010. Effects of salicylic acid on seedling growth and nitrogen metabolism in cucumber (Cucumis sativus L.). J. Stress Physiol. Biochem., 6: 103-113.
Direct Link - Larque-Saavedra, A., 1978. The antiranspirant effect of acetylsalcylic acid on Phaseolus vulgaris. Physiol. Plant., 43: 126-128.
CrossRefDirect Link - Larson, L.A. and H. Beevers, 1965. Amino acid metabolism in young pea seedlings. Plant Physiol., 40: 424-432.
Direct Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Mitchell, A.G. and J.F. Broadhead, 1967. Hydrolysis of solubilized Aspirin. J. Pharm. Sci., 56: 1261-1266.
CrossRef - Nooden, L.D., J.J. Guiamet and I. John, 1997. Senesence mechanisms. Physiol. Plant, 101: 746-753.
CrossRef - Pancheva, T.V., L.P. Popova and A.N. Uzunova, 1996. Effects of Salicylic acid on growth and photosynthesis in barley plants. J. Plant Physiol., 149: 57-63.
Direct Link - Ray, S.D., 1986. GA, ABA, phenol interaction in the control of growth: Phenolic compounds as effective modulators of GA-ABA interaction in radish seedlings. Biol. Planta, 28: 361-369.
CrossRefDirect Link - Ray, S.D., K.N. Guruprasad and M.M. Laloraya, 1983. Reversal of abscisic acid‐inhibited betacyanin synthesis by phenolic compounds in Amaranthus caudatus seedlings. Physiol. Plant., 58: 175-178.
CrossRefDirect Link - Miguel-Chavez, S.R., M. Gutierrez-Rodriguez and A. Larque-Saavedra, 2003. Salicylic acid increases the biomass accumulation of Pinus patula. Southern Applied For. J., 27: 52-54.
Direct Link - Schneider, E.A. and F. Wightman, 1974. Metabolism of auxin in higher plants. Annu. Rev. Plant Physiol., 25: 487-513.
CrossRef - Shettel, N.L. and N.E. Balke, 1983. Plant growth response to several allelopathic chemicals. Weed Sci., 31: 293-298.
Direct Link - Szepesi, A., J. Csiszar, S. Bajkan, K. Gemes and F. Horvath et al., 2005. Role of salicylic acid pre-treatment on the acclimation of tomato plants to salt- and osmotic stress. Acta Biol. Szegediensis, 49: 123-125.
Direct Link - Vaadia, Y., F.C. Raney and R.M. Hagan, 1961. Plant water deficits and physiological processes. Annu. Rev. Plant Physiol., 12: 265-292.
CrossRefDirect Link - Yang, C.M., C.N. Lee and C.H. Chou, 2002. Effects of three allelopathic phenolics on chlorophyll accumulation of rice (Oryza sativa) seedlings: I. Inhibition of supply-orientation. Bot. Bull. Acad. Sin., 43: 299-304.
Direct Link - Yuan, S. and H.H. Lin, 2008. Role of salicylic acid in plant abiotic stress. Z Naturforsch C., 63: 313-320.
PubMed - Alverez, M.E., 2000. Salicylic acid in the machinery of hypersensitive cell death and disease resistance. Plant Mol. Biol., 44: 429-442.
PubMedDirect Link








