Research Article
Antioxidant and Antibacterial Activities of Vegetables and Fruits Commonly Consumed in Thailand
Department of Biotechnology, Faculty of Technology, Mahasarakham University, Maha Sarakham, 44150, Thailand
Plants are rich sources of several important bioactive compounds. Apart from medicinal herbs, some commonly consumed vegetables and fruits may also have important phytochemicals with pharmacological properties, for example, Brassica vegetables (Podsedek, 2007) and Lycium barbarum fruit (Goji) (Amagase and Farnsworth, 2011). Furthermore, some native vegetables, especially leafy green vegetables and fruits with dark colors have also been found to have bioactive compounds with antioxidant and antimicrobial activities (Andarwulan et al., 2010; Tan et al., 2011; Murillo et al., 2012). The antioxidant activities and bioactive compounds in dietary plants can provide additional health benefits for prevention of cardiovascular disease, cancer, neurological decline and diabetes (Wootton-Beard and Ryan, 2011). In Thailand, a variety of native vegetables and fruits that are used as daily food and seasoning are proven to have high antioxidant activities in addition to their nutritional values, for instance, Cassia siamea, Tagetes erecta, Antigonon leptopus, Gymnema inodorum, Piper sarmentosum, Mentha arvensis, Diospyros decandra, Terminalia chebula and Phyllanthus emblica (Chanwitheesuk et al., 2005; Kaisoon et al., 2011; Kubola et al., 2011). There are a large number of Thai vegetables and fruits that are attractive due to their health-related functional properties as well as their possible future application for prevention and therapeutic purposes. However, there are many of them that remain unexplored. Therefore, this study aims to evaluate the antioxidant and antibacterial activities of the vegetables and fruits commonly consumed in the Northeast region of Thailand.
Preparation of plant extracts: The vegetables and fruits that are commonly consumed in the Northeast region of Thailand were collect or purchased from Maha Sarakham provinces. The vegetables (their common and Thai names are written in the brackets, respectively) included Neptunia oleracea Lour. (water mimosa, Krashednam), Limnophila aromatica (Lamk.) Merr. (rice paddy herb, Kayang), Coccinia grandis (L.) J. Voigt (ivy gourd, Tamleung), Basella alba Linn. (Ceylon spinach, Plungkhaw), Sauropus androgynus (Linn.) Merr. (sweet leaf bush, Wanban) and Moringa oleifera Lam. (drumstick tree, Maroom). The fruits included Flacourtia indica (Burm. f.) Merr. (governor's plum, Takhobpa), Phyllanthus emblica Linn. (Indian gooseberry, Makhampom), Phyllanthus acidus (L.) Skeels. (star gooseberry, Mayom), Antidesma thwaitesianum Müll. Arg. (Mao, Mamaoluang), Ziziphus oenoplia (L.) Mill. (jackal jujube, Lepmeaw) and Terminalia chebula Retz. (black myrobalan, Smore). These vegetables (leaves, stems and branches) and fruits (peels without seeds) were air dried to have less than 10% humidity, ground into fine powder and extracted using distilled water (DW) and 80% ethanol (E80) as solvents at the ratio 1:5 w/v. The extraction process was performed for 12 h with agitation at 150 rpm at room temperature (28-30°C). The resulting solutions were centrifuged at 4000 rpm for 10 min and the supernatants were concentrated using vacuum rotary evaporator at 45°C. The percentage of extraction yield was calculated as following:
Antioxidant activity determination by DPPH scavenging assay: DPPH scavenging assay for evaluation of antioxidant activities was described previously (Butsat and Siriamornpun, 2010). Plant extract (100 μL) was mixed with freshly prepared 0.1 mM DPPH° solution (1.9 mL) and incubated at room temperature in the dark for 30 min and then measured for absorbance at 517 nm. BHA (butylated hydroxyanisole) was used as standard reagent. The levels of DPPH scavenging activities of the extracts were calculated and expressed as IC50 (mg mL-1). The experiments were performed in three replicates.
Antioxidant activity determination by ferric reducing ability of plasma (FRAP) assay: FRAP assay for determination of antioxidant activities was previously described by Butsat and Siriamornpun (2010). FRAP stock solution was prepared by mixing 300 mM acetate buffer (pH 3.6), 10 mM TPTZ solution (in 40 mM HCl) and 20 mM FeCl3.6H2O solution. FRAP solution (1.7 mL) must be prepared freshly and warmed at 37°C before adding to plant extract (300 μL). The mixture was incubated at room temperature for 1 h and then measured for absorbance at 593 nm. The standard curve was plotted using the result obtained from FeSO4 standard reagent. The antioxidant activities of the extracts were calculated and expressed as mol FeSO4 eq g-1 of dry weight of vegetable or fruit. The experiments were carried out in three replicates.
Total phenolic compound measurement: The level of total phenolic compounds was measured by Folin-Ciocalteu method as previously described (Butsat and Siriamornpun, 2010). Plant extract (200 μL) was mixed with Folin-Ciocalteu reagent (diluted 1:10 with sterile distilled water) (1 mL), shaken for 1 min and added with 10% Na2CO3 (800 μL). After that, the mixture was topped up with sterile distilled water to have total volume of 5 mL and incubated at room temperature in the dark for 2 h. Finally, the mixture was measured for absorbance at 760 nm. Gallic acid was used as standard reagent. The levels of total phenolic compounds of the extracts were calculated and expressed as mg gallic acid eq g-1 of dry weight of vegetable or fruit. The experiments were performed in three replicates.
Antibacterial activity evaluation by agar well diffusion assay: Agar well diffusion assay for determination of antibacterial activity was performed as described previously (Oke et al., 2009) with some modifications. Staphylococcus aureus, Straphylococcus epidermidis, Streptococcus pyogenes and Propionibacterium acnes were grown overnight at 37°C and adjusted to approximately 1x108 CFU mL-1. The bacterial culture (100 μL) was then spread onto Mueller Hinton Agar (MHA) plate and let to dry for 10 min before being punctured by 0.6 cm sterile cork borer. Plant extract (30 μL) was added to each agar well and the plate was incubated at 37°C overnight. The antibacterial activities of the extracts were measured by the diameter size of inhibition zone around the agar well.
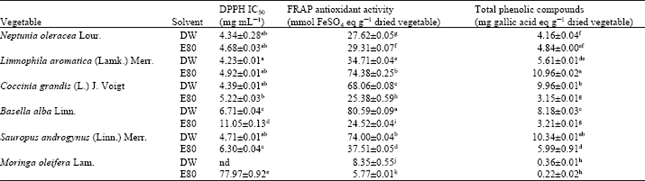
Levels of antioxidant activities and total phenolic compounds: The vegetables extracts were shown to have antioxidant activities measured by DPPH scavenging and FRAP assays (Table 1). The extracts of N. oleracea (extracted by DW and E80), L. aromatica (DW and E80), C. grandis (DW) and S. androgynus (DW) had significantly high antioxidant activities measured by DPPH scavenging assay. However, only the extracts of L. aromatica (E80) and S. androgynus (DW) that were also found to have significant levels of antioxidant activities measured by FRAP assays (74.38±0.25 and 74.00±0.04 mmol FeSO4 eq g-1 of dry weight of vegetable, respectively) and total phenolic compounds (10.96±0.02 and 10.34±0.01 mg gallic acid eq g-1 of dry weight of vegetable, respectively).
| Table 1: | Antioxidant activities and total phenolic compounds of vegetable extracts |
 | |
| Values (Mean±SD) in the same column with the same lower-case letter(s) are not significant different as determined by Duncan’s test at p<0.05, DW: Distilled water, E80: 80% ethanol, nd: Not detected | |
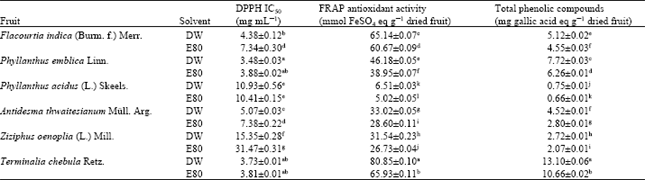
| Table 2: | Antioxidant activities and total phenolic compounds of fruit extracts |
 | |
| Values (Mean±SD) in the same column with the same lower-case letter(s) are not significant different as determined by Duncan’s test at p<0.05, DW: Distilled water, E80: 80% ethanol, nd: Not detected | |
Interestingly, B. alba extract (DW) that had the highest FRAP antioxidant activities (80.59±0.09 mmol FeSO4 eq g-1 of dry weight of vegetable) was found to have rather moderate levels of DPPH IC50 at 6.71±0.04 mg mL-1 and total phenolic compounds at 8.18±0.03 mg gallic acid eq g-1 of dry weight of vegetable.
The fruits extracts that were found to have significantly high levels of both antioxidant activities and total phenolic compounds are those derived from T. chebula (extracted by DW and E80) which giving DPPH IC50 at 3.73±0.01 and 3.81±0.01 mg mL-1, FRAP antioxidant activities at 80.85±0.10 and 65.93±0.11 mmol FeSO4 g-1 of dry weight of fruit and total phenolic compounds at 13.10±0.06 and 10.66±0.02 mg gallic acid eq g-1 of dry weight of fruit, respectively (Table 2). Also, P. emblica extracts (DW and E80) were shown to have significant levels of DPPH IC50 at 3.48±0.03 and 3.88±0.02 mg mL-1, respectively, but rather moderate levels of FRAP antioxidant activities and total phenolic compounds.
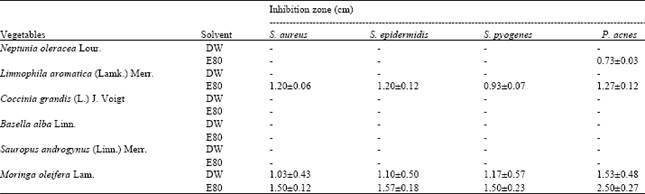
Antibacterial activities: For vegetable extracts, only M. oleifera and L. aromatica extracts were found to have antibacterial activities against all four tested bacteria (Table 3). The highest antibacterial activities was found in the E80 extract of M. oleifera that could inhibit S. aureus, S. epidermidis, S. pyogenes and P. acnes by producing inhibition zones with the diameters of 1.50±0.12, 1.57±0.18,1.50±0.23 and 2.50±0.27 cm, respectively, followed by the E80 extract of L. aromatica that produced inhibition zone at 1.20±0.06, 1.20±0.12, 0.93±0.07 and 1.27±0.12 cm, respectively. Moderate inhibition activities were found in the DW extract of M. oleifera which producing zone at 1.03±0.43, 1.10±0.50, 1.17±0.57 and 1.53±0.48 cm, respectively.
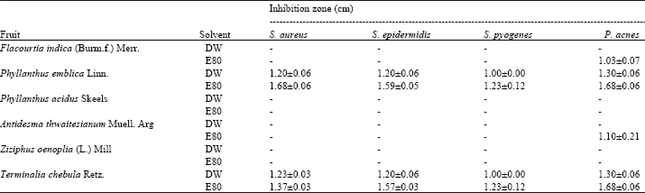
For fruit extracts, inhibition of all four tested bacteria were found when applying with the extracts of T. chebula (E80) that producing clear zones against S. aureus, S. epidermidis, S. pyogenes and P. acnes at 1.37±0.03, 1.57±0.03, 1.23±0.12 and 1.68±0.06, respectively (Table 4). Moreover, the extracts of P. emblica (E80) could also inhibit these four bacteria by inducing clear zones at 1.68±0.06, 1.59±0.05, 1.23±0.12 and 1.68±0.06, respectively.
Streptomycin (10 IU mL-1) was used as positive control group and inhibited S. aureus, S. epidermidis, S. pyogenes and P. acnes by producing clear zone at 3.47±0.15, 1.57±0.07, 1.83±0.03 and 1.63±0.07 cm, respectively. The solvents, DW and E80, were used as negative control groups and they did not show any inhibition against these four bacteria.
| Table 3: | Antibacterial activities of vegetable extracts |
 | |
| The size of cork borer is 0.6 cm | |
| Table 4: | Antibacterial activities of fruit extracts |
 | |
| The size of cork borer is 0.6 cm | |
In this study, the extracts of vegetables, L. aromatica, S. androgynus and B. alba, were shown to have good antioxidant activities. The phenolic compounds (especially flavonoids) and essential oils in the vegetables extracts have been indicated to strongly promote extracts’ antioxidant activities (Guimaraes et al., 2010). In this study, high levels of total phenolic compounds in the extract of S. androgynus (DW) could play an important role on promoting its antioxidant activities. The previous report suggested that the extract of Indonesian S. androgynus (extracted by 95% ethanol) was found to have high level of total phenolic compounds at 1.49±0.15 mg gallic acid eq g-1 fresh weight (Andarwulan et al., 2010). Moreover, the extract of L. aromatica (E80) was found to have strong aroma and thus could be assumed that it may contain high quantity of essential oil which may contribute to its high antioxidant activities. In addition, the ethanolic extract of B. alba leaves was reported to have robust antioxidant properties and provide strong protection against chemical carcinogenesis induced hepatocarcinogenesis in animal model (Reddy et al., 2008).
The extracts of T. chebula fruit were shown to have very strong antioxidant activities and high levels of total phenolic compounds. Recent reports have suggested that an antioxidant protein (~16 kDa) from T. chebula fruit has been successfully purified and characterized and it may be a major compound responsible for antioxidant activities of T. chebula fruit (Srivastava et al., 2012). Also, there are a number of previous reports that showed the strong antioxidant properties and high levels of phenolic compounds of the extracts derived from T. chebula fruit (Pfundstein et al., 2010; Saleem et al., 2002; Nampoothiri et al., 2011; Tubtimdee and Shotipruk, 2011). These results hence supported the findings of strong antioxidant activities and high levels of total phenolic compounds of T. chebula fruit extracts in this study. In addition, although P. emblica extracts was found to have slightly lower FRAP antioxidant activities and total phenolic compounds, they were found to have strong DPPH IC50 similar to previous reports (Liu et al., 2008a,b; Luo et al., 2009, 2011).
This study showed that the E80 extracts of M. oleifera, L. aromatica, T. chebula and P. emblica were found to have antibacterial activities against Staphylococcus aureus, Straphylococcus epidermidis, Streptococcus pyogenes and Propionibacterium acnes. As the negative control group using 80% ethanol alone showed no inhibition against these four bacteria, it is therefore unlikely that the residual ethanol in plant extracts could have any effect on their antibacterial activities. Furthermore, the antibacterial effects of these vegetable and fruits extracts have been previously reported. The extracts of M. oleifera were shown to inhibit Salmonella typhi, Pseudomonas aeruginosa and S. aureus (Doughari et al., 2007; Caceres et al., 1991). Also, the nanoparticles that applied with phenolic compounds (tannins) derived from T. chebula aqueous extract were shown to be effective at inhibiting S. aureus and Escherichia coli (Kumar et al., 2012). Moreover, the water extract of T. chebula was found to contain a heat stable agent(s) that could strongly inhibit Helicobacter pyroli (Malekzadeh et al., 2001). The extracts of P. emblica were also previously found to inhibit S. aureus due to a variety of its phenolic compounds (Mayachiew and Devahastin, 2008).
In this study, the antioxidant and antibacterial activities of Thai local vegetables and fruits have been illustrated. The results in this study may suggest the potentials of the extracts derived from S. androgynous, L. aromatica, B. alba, M. oleifera, T. chebula and P. emblica for application in cosmetic and/or therapeutic products due to their high levels of antioxidant activities and/or total phenolic compounds. These edible plants may be used as alternative to synthetic chemicals particularly because of their safety and ample amount in many local areas of Thailand.
This research is financially supported by the Mahasarakham University Research Fund and the Department of Biotechnology, Faculty of Technology, Mahasarakham University Thailand. Sincere thanks must go to Dr. Prapassorn Bussaman for useful idea and comments. Thanks to the Department of Biotechnology, MSU, for laboratory facility and Miss Jariya Chokchareon and Miss Supaksorn Srichat for laboratory assistances.