Research Article
Growth Effect of Cinnamomum kanehirae Cuttings Associated with its Dark Septate Endophytes
Department of Forestry and Natural Resources, National Chiayi University, Chiayi, 60004 Taiwan, Republic of China
Cinnamomum kanehirae Hay., a member of Lauraceae is an endemic specie and one of the 5 precious hardwood trees in Taiwan1. Antrodia camphorata is a well-known medicinal polypore found only in C. kanehirae2. Thus, C. kanehirae has been widely felled and is now an endangered species3,4.
Cinnamomum kanehirae seeds have an entomophilous characteristic and cannot be easily collected, hence, cutting is the best silvicultural practice for the resource conservation of C. kanehirae2,3. Kao and Huang5 indicated that cutting C. kanehirae could promote the formation of adventitious roots and got the best rooting rate of up 80%. However, using cutting for forestations often fails. The major factor for such failure is the lack of axial roots6. Hence, increasing the survival rate of C. kanehirae plantation is a key point for its conservation.
The various forms of mycorrhizal associations are ubiquitous in nature and play important roles in plant nutrition and nutrient cycling. They also influence the structure and dynamics of the plant communities within which they exist7. Functions of mycorrizae are to promote growth8,9 and tolerance10,11 of the host plant. Mycorrizae can increase rooting, with increased plant rooting, the growth of the host plant is promoted, especially in cutting12-17. Scagel14,16 also demonstrated that cuttings inoculated with mycorrhizal fungi had greater root initiation than non-inoculated cuttings.
Although, there are many studies on the indigenous C. kanehirae of Taiwan, most focused on the physiological ecology, phylogenetic relationships18-20 and propagation of the genus3,5,21,22, little attention has been paid to C. kanehirae mycorrhizae4,20,23,24. Two kinds of mycorrhiza, Arbuscular Mycorrhizae (AM) and Dark Septate Endophyte (DSE) have recently been discovered in C. kanehirae20. The AM fungi are obligate biotrophs and cannot be cultured without plants25, but dark septate endophytes can be grown in solid or liquid media20. Lin et al.4, demonstrated that C. kanehirae cuttings could resynthesize with its root fungus endophytes to form the structure of dark septate endophyte. However, the benefits of these 2 endophytes associated with C. kanehirae cuttings remain unknown. The objective of this study was to investigate the growth efficiency and physiological characteristics of C. kanehirae and its dark septate endophytes.
Strains: Two dark septate endophytes were isolated previously from the roots of C. kanehirae (120°47’37.33" E, 23°28’16.12" N). The plantation was located in Dabang Township, Chiayi County, Taiwan20. Specimens of CkDB2 and CkDB5 were deposited at the Tree Mycorrhiza Laboratory of National Chiayi University and their Internal Transcribed Spacer (ITS) genomic sequences were deposited in the GenBank (CkDB2 and CkDB5 isolates, KT780305 and KT780306, respectively)4.
Cuttings: Two year-old cuttings of C. kanehirae were obtained from Jhongsing Nursery (Chiayi Forest District Office, Forest Bureau, Council of Agriculture, Executive Yuan, Chiayi, Taiwan) and transplanted in new pots (10 cm diameter) in a nursery substrate (peat and perlite 3:1 v/v, previously autoclave-sterilized at 121°C for 60 min).
Inoculation with endophytes: The inoculation was performed according to the method of Zhang et al.26 with modifications. In brief, the cuttings were transplanted into new pots with the inoculum (10 pieces) placed near the root. The three treatments (1 control and 2 inoculated treatments) each had 3 replicates. The cuttings were grown, watered and fertilized in a greenhouse. Standard fertility was given to each pot with 50 mL sterile Hoagland’s nutrient solution every week.
Staining of root: After 9 months of incubation, the cuttings were removed from the pots. The roots were washed thoroughly with tap water, cut into segments of approximately 1 cm long, cleaned in 10% (w/v) KOH and 3% H2O2 according to the degree of lignification of the roots and stained with trypan blue lactophenol. The method was modified according to that used by McGonigle et al.27. Observation was made under light microscopy (Olympus X5).
Plant growth responses: To measure the effects of stains CkDB2 and CkDB5 on growth performance of cuttings, the cuttings were carefully removed from their substrate after 9 months of incubation. Each treatment had 3 replicates.
The net growth of height and root collar diameter of all treatments were regularly recorded after the cuttings were transplanted. The last measurement was taken right before the harvest.
The plant dry weight (root, stem and leaf) was obtained by oven-drying at 80°C until constant weight. Leaf Area (LA) of fresh leaves was determined using a leaf area meter (LI-COR LI 3100). Leaf Area Ratio (LAR), Specific Leaf Area (SLA) and Leaf Weight Ratio (LWR) were calculated using leaf area and dry weights of harvested plants. The equations used are as follows:
 |
Plenchette et al.28 defined the Mycorrhizal Dependency (MD) as the degree to which a plant is dependent on the mycorrhizal condition, which is expressed as follows:
Chlorophyll concentration: The method was modified according to that used by Porra29. To measure chlorophyll concentration, the third and 4th fully-expanded leaves (counting from the apex) were collected (two leaves per plant, n = 3). Then, 0.05 g of each sample (fresh material) was grounded with 10 mL of 80% acetone. The suspension was filtered and measured by a spectrophotometer (spectrophotometer, Hitachi U-2000) in the wavelengths of 663 nm (Chlorophyll a: Chl a) and 645 nm (Chlorophyll b: Chl b).
| Chl a | = (12.7×D663-2.69×D645)×(V/1000W) |
| Chl b | = (22.9×D645-4.68×D663)×(V/1000W) |
| Chl a+b | = (20.2×D645+8.02×D663)×(V/1000W) |
| Dλ | = Stands for absorbency at the specific wavelength λ |
| V | = Volume of ground leaf-acetone liquid (mL) |
| W | = Volume of the fresh weight (g) |
Statistical analysis: Statistical analysis was performed using the software Statistical Package for the Social Sciences (SPSS 12.0) (Illinois, USA) for windows program. Means of 3 separate experiments±standard error (n = 3) were derived from all collected data. Differences among endophytes were analyzed by Turkey’s multiple range test at p≤0.05 significant level.
Morphology and colonization: After 9 months of incubation, all cuttings survived. However, the inoculated cuttings showed better growth than the control (Fig. 1).
In the observation of stained root, microsclerotia (Fig. 2a), a feature of dark septate endophyte were found in the root associations of CkDB2-inoculation. Microsclerotia were also found in the CkDB5-inoculation (Fig. 2b), but noting was founded in the control (Fig. 2c). Therefore, the 2 endophytes could be associated with C. kanehirae cuttings and the root associations had the feature of dark septate endophyte. Similar results were also obtained in other studies30-33.
Plant growth responses
Net growth of height and root collar diameter: Both net growth of height and root collar diameter were measured after 9 months of incubation (Table 1). The cuttings inoculated with strains CkDB2 and CkDB5 showed the greatest net height growth (10.33±0.95 and 11.73±1.04 cm, respectively), compared with the control (2.67±0.21 cm). The net height growth of all treatments were significantly higher than that of the control (p<0.05), but the average net growth of root collar diameter of all treatments showed no significant difference.
Plant dry mass: At harvest, there was significant difference in dry weight (root, leaf and total dry weight) of all treatments (Table 2). The CkDB2-inoculation and CkDB5-inoculation generated better root dry weight values (11.30±1.48 and 14.44±0.72 g, respectively) than the control (6.33±2.25 g), revealing statistically significant difference (p<0.05). The leaf dry weight and the total dry weight showed the similar results. The CkDB2-inoculation and CkDB5-inoculation produced better leaf dry weight values (6.44±1.34 and 7.32±0.65 g, respectively) than the control (2.72±0.31 g). The CkDB2-inoculation and CkDB5-inoculation also yielded better total dry weight values (26.07±3.92 and 30.23±2.16 g, respectively) than the control (13.94±3.58 g).
| Table 1: | Growth effect of Cinnamomum kanehirae cuttings with different treatments after 9 month incubation |
 | |
| All values were Means±Standard Deviation of three replicate cultures, values in the same column with different letters were different at 5% significant level | |
| Table 2: | Dry weight of Cinnamomum kanehirae cuttings with different treatments after 9 month incubation |
 | |
| All values were Means±Standard Deviation of three replicate cultures, values in the same column with different letters were different at 5% significant level | |
 | |
| Fig. 1(a-c): | Morphology of mycorrhizal synthesis of Cinnamomum kanehirae cuttings after 9 month incubation, (a) CkDB2-inoculation, (b) CkDB5-inoculation and (c) Control |
| Table 3: | Leaf area and leaf parameters of Cinnamomum kanehirae cuttings with different treatments after 9 month incubation |
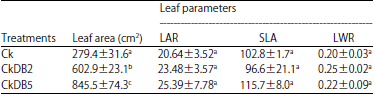 | |
| All values were Means±Standard Deviation of three replicate cultures, values in the same column with different letters were different at 5% significant level, LAR: Leaf area ratio, SLA: Specific leaf area and LWR: Leaf weight ratio | |
However, there were no significant differences in stem dry weight of the control, CkDB2 and CkDB5-inoculatin (4.90±1.90, 8.34±1.35 and 8.47±1.00 g, respectively).
Leaf area, leaf area ratio, specific leaf area and leaf weight ratio: The leaf area of cuttings was significantly higher when stimulated by inoculation (Table 3) (p<0.05). The CkDB2 and CkDB5-inoculation produced greater leaf area (602.9±23.1 and 845.5±74.3 cm2, respectively) than the control (279.4±31.6 cm2), but there were no significant differences in LAR, SLA and LWR.
Chlorophyll concentration: Chlorophyll concentration of all treatments were analyzed at harvest (Table 4). Both inoculations had significantly difference compared with the control (p<0.05). For Chl a concentration, CkDB2 and CkDB5-inoculation (0.50±0.17 and 0.54±0.04 mg g–1, respectively) had higher values than the control (0.23±0.04 mg g–1).
 | |
| Fig. 2(a-c): | Root structure of Cinnamomum kanehirae cuttings after 9 month incubation, (a) CkDB2-inoculation, (b) CkDB5-inoculation and (c) Control |
| Table 4: | Chlorophyll concentration in leaves of cuttings with different treatments after 9 months incubation |
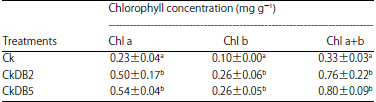 | |
| All values were Means±Standard Error of three replicate cultures, values in the same column with different letters were different at 5% significant level, Chl a: Chlorophyll a and Chl b: Chlorophyll b | |
Similar results were also observed in Chl b and Chl a+b concentration.
According to the above results, the 2 endophytes had positive effects on root growth of C. kanehirae cuttings. Many studies also demonstrated similar results13–17. Among these treatments, CkDB5-inoculation had increased root dry weight by more than 200% compared with the control. Similar results were also observed by Berta et al.34,35. Hence, CkDB5-inoculation had higher Mycorrhizal Dependency (MD) compared with CkDB2-inoculation (54 vs. 47%, respectively).
The mycorrhizal seedlings demonstrated that they could absorb more water and nutrients from the soil36-39 and had higher survival rate and growth rate of more than 100% compared with control seedlings34,35.
In plants, 90% of biomass (dry weight) came from photosynthesis40. While, previous findings indicated that inoculation had no positive effect on number and size of leaves17 and could not promote more efficient photosynthesis41,42. Zhang et al.26 demonstrated that number and area of leaves as well as chlorophyll concentration were all significantly affected by inoculation.
The results of this study confirmed the association of the two strains, CkDB2 and CkDB5 with the roots of C. kanehirae cuttings. Furthermore, these 2 endophytes could form microsclerotia of DSE with C. kanehirae cuttings. They also promoted increase in biomass, leaf area, chlorophyll concentration and net growth of height. Antrodia camphorata, restricting to the moribund heart wood of Stout camphor trees is a well-known medicinal polypore fund only in Cinnamomum kanehirae. Currently, C. kanehirae cuttings are used as a major propagation for plantation. However, these cuttings often fails to survive. The main reason for such failure to occur is due to the lack of axial roots. Hence, CkDB2 and CkDB5 can reasonably be inferred to have the potential to benefit C. kanehirae forestation, however CkDB5 has a slightly better performance in promoting growth of C. kanehirae cuttings.
The author appreciates the effort and assistance of the members of Tree Mycorrhiza Laboratory in National Chiayi University and Chiayi Forest District Office.