Research Article
Antioxidant Activity of Silymarin in Microcystin-LR Cardiac and Pulmonary Induced Injuries on Mice
Faculty of Applied Medical Sciences, Taif University, Taif City, Saudi Arabia
LiveDNA: 966.33248
Cyanobacteria is a blue-green alga that is present in different water supplies as dams, lakes and blooms1. Microcystin is a cyclic heptapeptide consisting of variable amino acids2. Microcystin-LR which contains two amino acids leucine (L) and arginine (R) is the most microcystin associated with the incident of toxicity in different countries3. Humans can be exposed to microcystin-LR mainly through drinking water contaminated with cyanobacteria. The liver is the main organ that detoxified microcystin-LR and its by-products can be seen in urine4. In the liver, microcystin-LR causes severe damage characterized by hepatocyte destruction, hepatic hemorrhage, leading to shock and death5. In Saudi Arabia, cyanobacteria were found in permanent and semipermanent rainwater ponds in Asir region6. A previous study conducted on male mice that were administered a high dose of microcystin-LR found that mice liver had chronic inflammation characterized by hepatocyte degeneration with significant elevation of serum ALT and AST, reduction in gamma-glutamyl transferase (GGT) and total protein and albumin. These injuries caused by the inhibitory effect of microcystin-LR on protein phosphatases which disturbs intracellular signaling and cytoskeleton of these tissue7. Microcystin-LR toxicity on myocytes led to damage and death8. In the same vein, a previous study conducted on mice showed that intraperitoneally injection of these mice with a toxic dose of microcystin-LR caused pulmonary thrombosis9. Moreover, two English cases were complicated with pneumonia after exposure to microcystin-LR10. Silymarin is a poorly water-soluble flavonoid that has anti-oxidant, anti-inflammatory and anti-cancer effects11. Extracts of both fruits and seeds are the main source of Silymarin maianum12. Silymarin induces anti-oxidant activity either through slowing calcium metabolism preventing initiation step in auto-oxidation or inhibiting lipid peroxidation by induction of superoxide dismutase and glutathione peroxidase13. In addition to its antioxidation activity, silymarin inhibits toxin absorption by the cell5. During inflammation, silymarin has an anti-inflammatory effect by the regulation of inflammatory mediators. It also inhibits neutrophils migration factor and prostaglandins synthesis6. In an animal study, silymarin protects cardiac tissue against ischemic heart diseases14. A previous study showed that treatment of animals injected with bleomycin with silymarin induced recovery of pulmonary injuries15. This study aimed to evaluate the antioxidant activity of silymarin on heart and lung injuries induced by microcystin-LR in Balb/c mice.
Experimental methods: This study was carried out in the Faculty of Applied Medical Sciences at Taif University between August 2019 to December of the same year. Seventy-two male Balb/c mice 5-7 weeks of age weighing about 30 grams were grouped into 6 groups; the first group was the control group (C), which consisted of twelve mice fed with a normal diet without any supplementation. Whereas, the second and the third groups contained twelve mice each called the microcystin-LR control group (M6 and M12). The mice in these groups were injected with microcystin-LR 19 mg kg–1 b.wt. intraperitoneally. After six and twelve hours of injection, the mice in both M6 and M12 were killed. Twelve mice supplemented orally with 250 mg silymarin per kg body weight daily were classified as silymarin control group (S), then killed after ten days. The mice of the fifth and sixth groups were orally supplemented with 250 mg silymarin per kg body weight daily. Ten days later, each mouse in these groups was injected with 19 mg microcystin-LR per kg body weight intraperitoneally. Six hours later, the mice of the fifth group were killed (SM6), while the mice of the sixth group were killed after 12 h (SM12) of microcystin-LR injection. Immediately, a blood sample was collected into a plain tube from each mouse after death, then centrifuged. The serum was isolated and was stored at -20°C for ALT and AST estimation. After death, both heart and lungs were resected immediately from each mouse. In normal saline tubes containing heparin, both organs were perfused. Furthermore, it was homogenized with phosphate buffer saline with PH 7.2 using Ultra Turrax homogenizer, centrifuged at 3000 g for 30 min. Finally, the supernatant was removed and stored16 at -20°C.
Biochemical measurement: The chemicals were purchased from (Sigma-Aldrich, USA). Estimation of biochemical parameters levels was done by using a spectrophotometer (UV-1800, Shimadzu). The serum transaminases (ALT and AST) of each mouse were measured. Moreover, reduced glutathione level and protein phosphatase I activity were estimated in cardiac and lungs homogenates. Finally, protein carbonyl content protein, products of lipid peroxidation and toxic methylglyoxal were estimated in both heart and lungs homogenates16.
Statistical analysis: All data were presented by using mean ± or plus-minus standard deviation (SD). The one-way Analysis of Variance (ANOVA) was used in the comparison of each biochemical parameter between all 6 groups. The level of significance was set at p<0.05. SPSS software version 16 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis.
Data in Table 1 presented the serum ALT activity showed a highly statistical difference between six groups (p<0.01). The M12 had a higher ALT activity (1956.7±60.77) compared with the control group (427.9±2.31). Meanwhile, the SC had a lower activity (427.9±2.31). Furthermore, a highly significant statistical difference was observed in protein phosphatase I activity between all groups (p<0.01). The M12 group had a higher AST activity (2.923±0.007) compared with the control group (2.923±0.007). The SC group had a lower activity (0.405±0.006). Table 2 represents the PPI activity, LPO, GSH, CC and MG levels in the cardiac homogenate of 6 groups. A significant statistical difference in the PPI activity was discovered between all groups (p<0.05). The M12 group had a lower activity (3.11±0.077), while the SC group had a higher activity (5.84±0.010) compared with the control group (5.56±0.091). In addition, a significant statistical difference appeared in the GSH level between all groups (p<0.05) with a higher level in the SC group (2.00±0.070) while the lower level in the M12 group (0.775±0.017) compared with the control group (1.70±0.070 ). In the same homogenate, a significant statistical difference was found in the CC level between all groups (p<0.05). The M12 group had a higher level (0.468±0.027) while the SC had the lower level (0.180±0.007). Moreover, the MG level showed a significant statistical difference between the six groups (p<0.01). The M12 group had a higher level (4.130±0.021), while the SC group had a lower level (0.960±0.007). The activity of PPI and the levels of GSH, LPO, CC and MG of all groups in the lung homogenate are represented in Table 3. A significant statistical difference was found in the CC level between all groups with a higher level in the M12 (0.590±0.009) while the lower level in the SC (0.180±0.007) compared with the control group (0.260±0.005) (p<0.05). Finally, the PPI activity showed a significant statistical difference with the higher activity in the SC group (5.245±0.173) while the lower activity in the M12 group (2.900±0.056) compared with the control group (4.230±0.056) (p<0.05).
Cardiac diseases are still the most common cause of death all over the world. Oxidative stress plays an important role in cardiac disorders. Microcystin-LR is a toxic heptapeptide that affects different organs in the human body. It induces cell injury mainly through inhibition of serine/threonine protein phosphatase14. Microcystin-LR affects cardiomyocyte by inducing oxidative stress17. This stress either occurs due to the increasing production of reactive oxygen species (ROS) or decreasing antioxidant production18.
| Table 1: | Activities of Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in different groups |
**p<0.01, C: Control group, M6: Microcystin 6 hours group, M12: Microcystin 12 h group, SC: Silymarin control group, SM12: Silymarin microcystin 6 h group and silymarin microcystin 12 h group | |
| Table 2: | Biochemical parameters of cardiac homogenate exposed to microcystin-LR and Silymarin compared with control homogenate |
 | |
*p<0.05, **p<0.01, C: Control group, M6: Microcystin 6 h group, M12: Microcystin 12 h group, SC: Silymarin control group, SM12: Silymarin microcystin 6 h group and silymarin microcystin 12 h group | |
| Table 3: | Biochemical parameters of lung homogenate exposed to microcystin-LR and Silymarin compared with control homogenate |
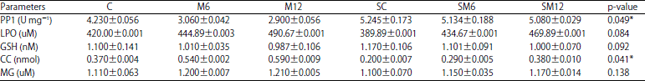 | |
*p<0.05, C: Control group, M6: Microcystin6 hours group, M12: Microcystin 12 h group, SC: Silymarin control group, SM12: Silymarin microcystin 6 h group and silymarin microcystin 12 h group | |
The increase of ROS is leading to the peroxidation of membrane lipids mainly polyunsaturated fatty acids19. In addition, microcystin-LR increases the level of toxic methylglyoxal by inhibiting glyoxalase I20. Previously, a histological study conducted on mice showed that long exposure of these mice to microcystin-LR induced enlargement of cardiomyocytes, loss of cells cross-striations, fibrosis and immune cells infiltration21. This study was carried out before on male Balb/c mice showed that injecting these mice with microcystin-LR induced methylglyoxal while it decreased reduced glutathione in the cardiac homogenate. Moreover, serum AST activity increased in these mice compared with those of the control group. Certainly, AST activity resulted from either hepatocyte or cardiomyocyte destruction. Silymarin is a polyphenolic flavonoid, isolated from Silybum marianum22. It performed a therapeutic effect in different tissues including cardiac, lungs and liver through its antioxidant activity23. Silymarin antioxidant activity was accomplished by its action as a scavenger of free radical, besides, it increased reduced glutathione level24. The results showed that silymarin dose decreased infarct size, lipid peroxide and transaminases level, however, it induced the activity of glutathione transferase and catalase25. In this study, silymarin decreased serum activities of AST and ALT nearly by two-fold in mice injected with microcystin-LR. The lipid peroxidation products level as a result of oxidative degradation of polyunsaturated fatty acids in cell membrane were reduced after silymarin administration26. Moreover, it induced the level of reduced glutathione which improved antioxidant activity against oxidative stress resulting from microcystin-LR toxic effect. In addition, protein carbonyl content and methylglyoxal resulting from microcystin-LR toxicity were reduced after the administration of silymarin. Also, it was found that silymarin induced protein phosphatase I activity involved in cell growth and differentiation26. Nearly, results of the present study may represent the protective effect of silymarin for cardiomyocytes either directly through induction of reduced glutathione level or indirectly through induction of protein phosphatase I involved in the growth of cardiomyocytes. As for lung tissue, a previous study found that microcystin-LR induced pulmonary injuries characterized by alveolar collapse, cell apoptosis, polymorphonuclear cell infiltration and inflammatory changes in rat. These changes were followed by the damaged lung tissues and impairment in its function26. In this study, intraperitoneal injection of Male Balb/c mice with microcystin-LR induced protein carbonyl content, however, it reduced protein phosphatase activity. Silymarin had a therapeutic effect on many pulmonary diseases including small cell carcinoma and lung ischemic diseases26. It played a significant role against pulmonary disorders either by its anti-inflammatory or antioxidant activities. Natural factor kaba B (NF-kB) induced inflammatory reaction in the lung by induction of chemokines, expression of pro-inflammatory cytokines and immune cells infiltration27. Silymarin acts as anti-inflammatory cytokine by inhibiting the inflammatory process through inhibition of NF-kB, tumor necrosis28 factor-α (TNF-α) and IL-6. Silymarin maintains a normal oxidant level by inducing the synthesis of superoxide dismutase catalase and precursor of reduced glutathione29. In this study, silymarin reduced both lipid peroxidation products and protein carbonyl content in lung homogenate resulting from lung oxidative stress and tissue damage. Moreover, it induced protein phosphatase activity responsible for lung tissue repair and cure. On the other hand, silymarin induced the reduced glutathione level responsible for antioxidant activity.
In conclusion, silymarin obtained a therapeutic role in cardiac and pulmonary injuries through its antioxidant property that may be resulted by the induction of reduced glutathione level or activation of protein phosphatase I. The discovery of an active substance in silymarin that is responsible for its antioxidant activity is recommended.
This study discovered the antioxidant activity of silymarin that can be beneficial for using this nontoxic compound to reduce the oxidative stress produced in some chronic diseases. This study will help the researchers to uncover the critical areas of medicinal plants that many researchers were not able to explore. Thus, a new theory on medicinal plants may be arrived at.
The author would like to thank all the staff at the animal house of the Deanship of Scientific Research, Taif University, SA for helping their help in this project.